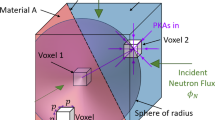
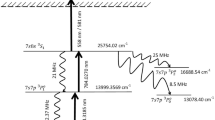
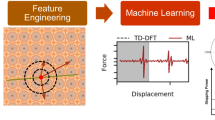
Abstract
I SHOULD like to comment on the assumption as to the atomic weight of RaG made by Dr. King in his letter in NATURE of May 6, p. 582. Instead of taking this as known, and calculating the atomic weight of radium by taking account of the α-particle, energy and electron masses, it appears to me that the reverse process would be more justified. It is true that there is an unexplained discrepancy in the difference between the atomic weights of uranium and radium, but we have no reason to doubt the value of the atomic weight of radium obtained by Hönigschmid. As his radium was obtained from Joachimstal pitchblende, which contains practically no thorium, there is no liability to disturbance from the presence of isotopes of radium in his material. On the other hand, the theoretical atomic weight of RaG was obtained by subtracting the mass of five α-particles from the atomic weight of Ra. That the lowest value (206.046) found by Hönigschmid for lead from Morogoro pitchblende agrees so nearly with the theoretical value (205.93), shows only that the amount of original lead present in this material was very small, although, of course, it gave splendid confirmation to the theory of isotopy. We cannot say, however, that we are dealing with pure RaG, even though we know that the amount of admixed isotopes must be very small.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
LAWSON, R. A Proposed Laboratory Test of the Theory of Relativity. Nature 109, 613 (1922). https://doi.org/10.1038/109613c0
Issue date:
DOI: https://doi.org/10.1038/109613c0