Abstract
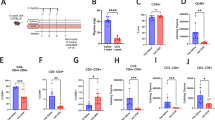
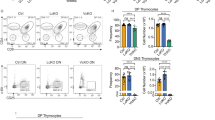
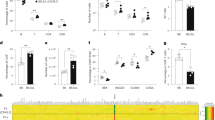
The Fas death receptor is a cell surface molecule involved in apoptosis as well as in proliferative or activating signals of many cells types, including T lymphocytes. Using quantitative real-time reverse transcription–PCR analysis, we confirm that expression of this gene is scarcely perceptible in thymic lymphomas induced by γ-irradiation in C57BL/6J mice. Notably, we also demonstrate for the first time that Fas expression is significantly upregulated in vivo both after single high dose of radiation and in thymic lymphoma-free mice. In addition, we determined its levels of expression in five mouse strains exhibiting different degrees of susceptibility (SPRET/Ei, SEG/Pas, BALB/cJ, C57BL/6J and RF/J). Interestingly, we found the highest levels of expression in SPRET/Ei and SEG/Pas strains (both derived from the Mus spretus species), which are known to have the most resistant phenotype, and the lowest levels in the most susceptible strains C57BL/6J and RF/J. DNA sequencing of the Fas promoter in all five strains showed many polymorphisms that can be classified into three functional haplotypes by using luciferase assays: (1) C57BL/6J and RF/J, (2) BALB/cJ and (3) SPRET/Ei and SEG/Pas. Promoter activities in response to single high doses of radiation correlated well with the levels of Fas expression and are consistent with the degree of strain susceptibility.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Accession codes
Accessions
GenBank/EMBL/DDBJ
References
Adachi M, Suematsu S, Suda T, Watanabe D, Fukuyama H, Ogasawara J et al. (1996). Proc Natls Acad Sci USA 93: 2131–2136.
Booker JK, Reap EA, Cohen PL . (1998). J Immunol 161: 4536–4541.
Castro JE, Listman JA, Jacobson BA, Wang Y, López PA, Ju S et al. (1996). Immunity 5: 617–627.
Chan H, Bartos DP, Owen-Schaub LB . (1999). Mol Cell Biol 19: 2098–2108.
DeRyckere D, Mann DL, DeGregory J . (2003). J Immunol 171: 802–811.
Feuk L, Prince JA, Blennow K, Brookes AJ . (2003). Hum Mutat 21: 53–60.
Fleck M, Zhou T, Tatsuta T, Yang P, Wang P, Mountz JD . (1998). J Immunol 160: 3766–3775.
Hettmann T, DiDonato J, Karin M, Leiden JM . (1999). J Exp Med 189: 145–157.
Hettmann T, Leiden JM . (2000). J Immunol 165: 5004–5010.
Hoogendoorn B, Coleman SL, Guy CA, Smith K, Bowen T, Buckland PR et al. (2003). Hum Mol Genet 12: 2249–2254.
Kishimoto H, Surh CD, Sprent J . (1998). J Exp Med 187: 1427–1438.
Krammer PH . (2000). Nature 407: 789–795.
Kurasawa K, Hashimoto Y, Iwamoto I . (1999). Cell Immunol 194: 127–135.
Lai HC, Sytwu HK, Sun CA, Yu MH, Yu CP, Liu HS et al. (2003). Int J Cancer 103: 221–225.
Maas K, Westfall M, Pietenpol J, Olsen NJ, Aune T . (2005). Arthritis Rheumatism 52: 1047–1057.
Meléndez B, Santos J, Fernández-Piqueras J . (1999). Oncogene 18: 4166–4169.
Muschen M, Warskulat U, Beckann MW . (2000). J Mol Med 78: 312–325.
Nagata S, Suda T . (1995). Immunol Today 16: 39–43.
Nishimura Y, Ishii A, Kobayashi Y, Yamasaki Y, Yonehara S . (1995). J Immunol 154: 4395–4403.
Ogasawara J, Suda T, Nagata S . (1995). J Exp Med 181: 485–491.
Oukka M, Ho C, de la Brousse FC, Hoey T, Grusby MJ, Glimcher LH . (1998). Immunity 9: 295–304.
Pinkoski MJ, Green DR . (1999). Cell Death Differ 6: 1174–1181.
Rathmell JC, Thompson CB . (2002). Cell 109: S97–S107.
Reap EA, Roof K, Maynor K, Borrero M, Booker J, Cohen PL . (1997). Proc Natl Acad Sci USA 94: 5750–5755.
Santos J, Herranz M, Fernández M, Vaquero C, López P, Fernández-Piqueras J . (2001). Oncogene 20: 2186–2189.
Santos J, Montagutelli X, Acevedo A, López P, Vaquero C, Fernández M et al. (2002). Oncogene 21: 6680–6683.
Santos J, Pérez de Castro I, Herranz M, Pellicer A, Fernández-Piqueras J . (1996). Oncogene 12: 669–676.
Schmitz I, Clayton LK, Reinherz EL . (2003). Int Immunol 15: 1237–1248.
Sibley K, Rollinson S, Allan JM, Smith AG, Law GR, Roddam PL et al. (2003). Cancer Res 63: 4327–4330.
Siegel RM, Chan KM, Chun HJ, Lebardo MJ . (2000). Nat Immunol 1: 469–474.
Zheng Y, Ouaaz F, Bruzzo P, Singh V, Gerondakis S, Beg AA . (2001). J Immunol 166: 4949–4957.
Acknowledgements
We thank L González, M Matabuena, M Guerrero, MJ Bueno and R Peregil for aid with animals, P Fernández for helping with statistical analysis and E Pérez-Gómez for technical assistance. We are also grateful to Professor A Morales for critical reading of the manuscript. This work was supported by grants from the European Commission (EURATOM Work Programme 2003, FI6R-CT-2003-508842 to JS), ‘Ministerio de Educación y Ciencia’ (SAF2003-05048) and ‘Ministerio de Salud y Consumo’ (Lymphoma Network, G03/179) to JF-P. MV-M is the recipient of a predoctoral fellowship from ‘Ministerio de Educación y Ciencia’ (FPU AP2002-0294).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Villa-Morales, M., Santos, J. & Fernández-Piqueras, J. Functional Fas (Cd95/Apo-1) promoter polymorphisms in inbred mouse strains exhibiting different susceptibility to γ-radiation-induced thymic lymphoma. Oncogene 25, 2022–2029 (2006). https://doi.org/10.1038/sj.onc.1209234
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/sj.onc.1209234
Keywords
This article is cited by
-
Deep genome sequencing and variation analysis of 13 inbred mouse strains defines candidate phenotypic alleles, private variation and homozygous truncating mutations
Genome Biology (2016)
-
FAS system deregulation in T-cell lymphoblastic lymphoma
Cell Death & Disease (2014)
-
Depletion of the receptor for advanced glycation end products (RAGE) sensitizes towards apoptosis via p53 and p73 posttranslational regulation
Oncogene (2013)
-
The stromal gene encoding the CD274 antigen as a genetic modifier controlling survival of mice with γ-radiation-induced T-cell lymphoblastic lymphomas
Oncogene (2010)
-
Identification of a novel cerebral malaria susceptibility locus (Berr5) on mouse chromosome 19
Genes & Immunity (2010)