Abstract
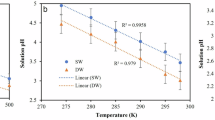
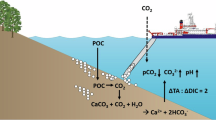
IN addition to neutral salts, sea-water contains a small quantity of ‘excess base’ in the form of bicarbonate and of carbonate, together with free carbonic acid. The ratio of HCO3′ to CO3″, free CO2, and consequently hydrogen ion concentration, varies with time and place, dependent upon the intensity of assimilation by plants, of respiration by animals and plants, exchange with the carbon dioxide of the atmosphere, production of calcium carbonate in the skeletons of coccolithophores and animals, and its solution from calcareous sediments. Further, the interrelation between carbon dioxide tension, hydrogen ion concentration, and excess base in saline physiological fluids has been the subject of much research, concerning which there is a considerable and growing literature.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
BUCH, K., WATTENBERG, H. & HARVEY, H. Apparent Dissociation Constants of Carbon Dioxide in Sea-water of Different Salt Contents. Nature 128, 411–412 (1931). https://doi.org/10.1038/128411a0
Issue date:
DOI: https://doi.org/10.1038/128411a0