Abstract
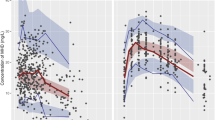
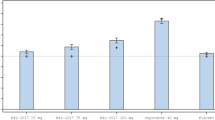
Epilepsy and bipolar disorder are commonly treated by combination drug therapy, such as lamotrigine and oxcarbazepine. To ensure the safety of this combination, information on pharmacokinetics and tolerability must be available. The objective of study was to evaluate the pharmacokinetics and tolerability of coadministered lamotrigine and oxcarbazepine in healthy subjects. This randomized, single-blind, parallel-group study comprised three cohorts: lamotrigine (200 mg daily) plus oxcarbazepine (600 mg twice daily), lamotrigine (200 mg daily) plus placebo, and oxcarbazepine (600 mg twice daily) plus placebo. Serial blood samples were collected at steady state to determine serum concentrations of lamotrigine and plasma concentrations of oxcarbazepine and its active metabolite 10-monohydroxy metabolite (MHD). Pharmacokinetic parameters were determined by noncompartmental methods. Tolerability was monitored through adverse event reports, clinical laboratory results, vital signs, and electrocardiograms. A total of 47 male volunteers received study drugs. At steady state, lamotrigine AUC(0–24) and Cmax were not significantly affected by oxcarbazepine cotherapy, nor were MHD AUC(0–12) and Cmax significantly affected by lamotrigine cotherapy. The most common adverse events, headache, dizziness, nausea, and somnolence, occurred more frequently during lamotrigine and oxcarbazepine combination therapy than during the monotherapy. No significant changes in clinical laboratory parameters, vital signs, or electrocardiograms were reported. In conclusion, the combination of lamotrigine and oxcarbazepine does not require dose adjustments based on pharmacokinetic data. However, it is important to recognize that the combination therapy was associated with more frequent adverse events.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Anderson GD, Gidal BE, Messenheimer JA, Gilliam FG (2002). Time course of lamotrigine de-induction: impact of step-wise withdrawal of carbamazepine or phenytoin. Epilepsy Res 49: 211–217.
Callaham ML, Wears RL, Weber EJ, Barton C, Young G (1998). Positive-outcome bias and other limitations in the outcome of research abstracts submitted to a scientific meeting. JAMA 280: 254–257 [Erratum in: JAMA (1998) 280: 1232.].
Cohen AF, Land GS, Breimer DD, Yuen WC, Winton C, Peck AW (1987). Lamotrigine, a new anticonvulsant: pharmacokinetics in normal humans. Clin Pharmacol Ther 42: 535–541.
Deckers CL, Hekster YA, Keyser A, Meinardi H, Renier WO (1997). Drug load in clinical trials: a neglected factor. Clin Pharmacol Ther 62: 592–595.
Deckers CLP (2002). The place of combination therapy in the early treatment of epilepsy. CNS Drugs 16: 155–163.
Fattore C, Cipolla G, Gatti G, Limido GL, Sturm Y, Bernasconi C et al (1999). Induction of ethinylestradiol and levonorgestrel metabolism by oxcarbazepine in healthy women. Epilepsia 40: 783–787.
Flesch G (2004). Overview of the clinical pharmacokinetics of oxcarbazepine. Clin Drug Invest 24: 185–203.
Food and Drug Administration (2003). Guidance for Industry: Bioavailability and Bioequivalence Studies for Orally Administered Drug Products—General Considerations. http://www.fda.gov/cder/guidance/index.htm.
Glauser TA (2001). Oxcarbazepine in the treatment of epilepsy. Pharmacotherapy 21: 904–919.
Goa KL, Ross SR, Chrisp P (1993). Lamotrigine. A review of its pharmacological properties and clinical efficacy in epilepsy. Drugs 46: 152–176.
Greenblatt DJ, von Moltke LL, Harmatz JS, Chen G, Weemhoff JL, Jen C . et al (2003). Time course of recovery of cytochrome p450 3A function after single doses of grapefruit juice. Clin Pharmacol Ther 74: 121–129.
Guenault N, Odou P, Robert H (2003). Increase in dihydroxycarbamazepine serum levels in patients co-medicated with oxcarbazepine and lamotrigine. Eur J Clin Pharmacol 59: 781–782.
Hirschfeld RM, Kasper S (2004). A review of the evidence for carbamazepine and oxcarbazepine in the treatment of bipolar disorder. Int J Neuropsychopharmacol 30: 1–16 [Epub ahead of print].
Hulsman JARJ, Rentmeester TW, Banfield CR, Reidenberg P, Colucci RD, Meehan JW et al (1995). Effects of felbamate on the pharmacokinetics of the monohydroxy and dihydroxy metabolites of oxcarbazepine. Clin Pharmacol Ther 58: 383–389.
Keranen T, Jolkkonen J, Jensen PK, Menge GP, Andersson P (1992a). Absence of interaction between oxcarbazepine and erythromycin. Acta Neurol Scand 86: 120–123.
Keranen T, Jolkkonen J, Klosterskov-Jensen P, Menge GP (1992b). Oxcarbazepine does not interact with cimetidine in healthy volunteers. Acta Neurol Scand 85: 239–242.
Kramer G, Dorn T, Etter H (2003). Oxcarbazepine: clinically relevant drug interaction with lamotrigine. Epilepsia 44(Suppl 9): 95–96; Abstract No. 1.267. Presented at the Annual Meeting of the American Epilepsy Society. Boston, MA, USA, 05 December 2003–10 December 2003.
Lamictal® (lamotrigine) [package insert] (2004) GlaxoSmithKline: Research Triangle Park, NC.
Lloyd P, Flesch G, Dieterle W (1994). Clinical pharmacology and pharmacokinetics of oxcarbazepine. Epilepsia 35(Suppl 3): S10–S13.
May TW, Korn-Merker E, Rambeck B (2003). Clinical pharmacokinetics of oxcarbazepine. Clin Pharmacokinet 42: 1023–1042.
May TW, Rambeck B, Jurgens U (1999). Influence of oxcarbazepine and methsuximide on lamotrigine concentrations in epileptic patients with and without valproic acid comedication: results of a retrospective study. Ther Drug Monit 21: 175–181.
Ohnhaus EE, Breckenridge AM, Park BK (1989). Urinary excretion of 6 beta-hydroxycortisol and the time course measurement of enzyme induction in man. Eur J Clin Pharmacol 36: 39–46.
Park BK, Breckenridge AM (1981). Clinical implications of enzyme induction and enzyme inhibition. Clin Pharmacokinet 6: 1–24.
Shorvon S (2000). Oxcarbazepine: a review. Seizure 9: 75–79.
Tecoma ES (1999). Oxcarbazepine. Epilepsia 40(Suppl 5): S37–S46.
Trileptal® (oxcarbazepine) [package insert] (2004) Novartis: East Hanover, NJ.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Theis, J., Sidhu, J., Palmer, J. et al. Lack of Pharmacokinetic Interaction between Oxcarbazepine and Lamotrigine. Neuropsychopharmacol 30, 2269–2274 (2005). https://doi.org/10.1038/sj.npp.1300831
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/sj.npp.1300831
Keywords
This article is cited by
-
Antiepileptic drug therapy in the elderly: a clinical pharmacological review
Acta Neurologica Belgica (2019)
-
Pharmacokinetics and Drug Interaction of Antiepileptic Drugs in Children and Adolescents
Pediatric Drugs (2018)
-
Unterschiedlicher Effekt von Monotherapien mit Carbamazepin, Oxcarbazepin und Lamotrigin auf klinisch relevante Laborparameter
Zeitschrift für Epileptologie (2007)