Abstract
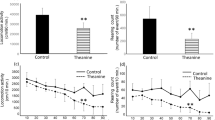
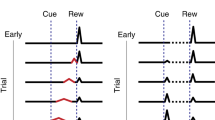
Central dopamine function is reduced by decreasing the availability of the catecholamine precursor, tyrosine, using a tyrosine-free amino acid mixture containing multiple large neutral as well as branched chain amino-acids, which compete with tyrosine for uptake into the brain. Current mixtures are cumbersome to make and administer, and unpalatable to patients and volunteers. Here, we investigate whether individual or limited amino-acid combinations could reduce brain tyrosine levels and hence dopamine function. Measurements of regional brain tyrosine levels, catecholamine and indoleamine synthesis (L-DOPA and 5-HTP accumulation, respectively) were used to identify an effective paradigm to test in neurochemical, behavioral and fos immunocytochemical models. Administration of leucine or isoleucine, or a mixture of leucine, isoleucine, and valine reduced tyrosine and 5-HTP, but not L-DOPA accumulation. A mixture of leucine, valine, and isoleucine supplemented with tryptophan reduced brain tyrosine and L-DOPA, but not 5-HTP. In microdialysis experiments this amino-acid mixture reduced basal and amphetamine-evoked striatal dopamine release, as well as amphetamine-induced hyperactivity. This mixture also reduced amphetamine-induced fos expression in striatal areas. In conclusion, the present study identified a small combination of amino acids that reduces brain tyrosine and dopamine function in a manner similar to mixtures of multiple amino acids. This minimal mixture may have use as a dopamine reducing paradigm in patient and volunteer studies.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Biggio G, Porceddu ML, Gessa GL (1976). Decrease of homovanillic, dihydroxyphenylacetic acid and cyclic-adenosine-3′,5′-monophosphate content in the rat caudate nucleus induced by the acute administration of an aminoacid mixture lacking tyrosine and phenylalanine. J Neurochem 26: 1253–1255.
Binek-Singer P, Johnson TC (1982). The effects of chronic hyperphenylalaninaemia on mouse brain protein synthesis can be prevented by other amino acids. Biochem J 206: 407–414.
Bing G, Stone EA, Zhang Y, Filer D (1992). Immunohistochemical studies of noradrenergic-induced expression of c-fos in the rat CNS. Brain Res 592: 57–62.
Carlsson A, Lindqvist M (1978). Dependence of 5-HT and catecholamine synthesis on concentrations of precursor amino-acids in rat brain. Naunyn-Schmiedeberg's Arch Pharmacol 303: 157–164.
De Deurwaerdere P, Navailles S, Berg KA, Clarke WP, Spampinato U (2004). Constitutive activity of the serotonin2C receptor inhibits in vivo dopamine release in the rat striatum and nucleus accumbens. J Neurosci 24: 3235–3241.
Ellenbogen M, Young S, Dean P, Palmour R, Benkelfat C (1996). Mood response to acute tryptophan depletion in healthy volunteers: sex differences and temporal stability. Neuropsychopharmacology 15: 465–474.
Fernstrom J (1983). Role of precursor availability in control of monoamine biosynthesis in brain. Physiol Rev 63: 484–546.
Fernstrom J (2000). Can nutrient supplements modify brain function? Am J Clin Nutr 71: 1669S–1675S.
Fernstrom M, Fernstrom J (1995). Acute tyrosine depletion reduces tyrosine hydroxylation rate in rat central nervous system. Life Sci 57: PL97–PL102.
Gessa GL, Biggio G, Fadda F, Corsini GU, Tagliamonte A (1974). Effect of the oral administration of tryptophan-free amino acid mixtures on serum tryptophan, brain tryptophan and serotonin metabolism. J Neurochem 22: 869–870.
Gijsman HJ, Scarna A, Harmer CJ, McTavish SB, Odontiadis J, Cowen PJ et al (2002). A dose-finding study on the effects of branch chain amino acids on surrogate markers of brain dopamine function. Psychopharmacology (Berlin) 160: 192–197.
Graybiel AM, Moratalla R, Robertson HA (1990). Amphetamine and cocaine induce drug-specific activation of the c-fos gene in striosome-matrix compartments and limbic subdivisions of the striatum. Proc Natl Acad Sci 87: 6912–6916.
Jaskiw GE, Bongiovanni R (2004). Brain tyrosine depletion attenuates haloperidol-induced striatal dopamine release in vivo and augments haloperidol-induced cataleosy in the rat. Psychopharmacology 172: 100–107.
Knudsen GM, Hasselbalch S, Toft PB, Christensen E, Paulson OB, Lou H (1995). Blood–brain barrier transport of amino acids in healthy controls and in patients with phenylketonuria. J Inherit Metab Dis 18: 653–664.
Le Masurier M, Cowen P, Sharp T (2004a). Fos immunocytochemical studies on the neuroanatomical sites of action of acute tyrosine depletion in the rat brain. Psychopharmacology (Berlin) 171: 435–440.
Le Masurier M, Houston G, Cowen P, Grasby P, Sharp T, Hume S (2004b). Tyrosine-free amino acid mixture attenuates amphetamine-induced displacement of [11C]raclopride in striatum in vivo: a rat PET study. Synapse 51: 151–157.
McTavish S, Callado L, Cowen PJ, Sharp T (1999a). Comparison of the effects of alpha-methyl-p-tyrosine and a tyrosine-free amino acid load on extracellular noradrenaline in the rat hippocampus in vivo. J Psychopharmacol 13: 379–384.
McTavish S, Cowen P, Sharp T (1999b). Effect of a tyrosine-free amino acid mixture on regional brain catecholamine synthesis and release. Psychopharmacology 141: 182–188.
McTavish SF, McPherson MH, Harmer CJ, Clark L, Sharp T, Goodwin GM et al (2001a). Antidopaminergic effects of dietary tyrosine depletion in healthy subjects and patients with manic illness. Br J Psychiatry 179: 356–360.
McTavish SF, Raumann B, Cowen PJ, Sharp T (2001b). Tyrosine depletion attenuates the behavioural stimulant effects of amphetamine and cocaine in rats. Eur J Pharmacol 424: 115–119.
Paxinos G, Watson C (1986). The Rat Brain in Stereotaxic Coordinates, 2nd edn. Academic Press: New York.
Richardson MA, Bevans ML, Weber JB, Gonzalez JJ, Flynn CJ, Amira L et al (1999). Branched chain amino acids decrease tardive dyskinesia symptoms. Psychopharmacology (Berlin) 143: 358–364.
Scarna A, Gijsman HJ, Harmer CJ, Goodwin GM, Cowen PJ (2002). Effect of branch chain amino acids supplemented with tryptophan on tyrosine availability and plasma prolactin. Psychopharmacology (Berlin) 159: 222–223.
Scarna A, Gijsman HJ, McTavish SF, Harmer CJ, Cowen PJ, Goodwin GM (2003). Effects of a branched-chain amino acid drink in mania. Br J Psychiatry 182: 210–213.
Sharp T, Zetterström T (1992). In vivo measurement of monoamine neurotransmitter release and metabolism using brain microdialysis. In: Stamford JA (ed). Monitoring Neuronal Activity: A Practical Approach. Oxford University Press: Oxford. pp 147–179.
Tam SY, Elsworth JD, Bradberry CW, Roth RH (1990). Mesocortical dopamine neurons: high basal firing frequency predicts tyrosine dependence of dopamine synthesis. J Neural Transm 81: 97–110.
Voog L, Eriksson T (1992). Diumal rhythms in rat brain large neutral amino acids (LNAAs), monoamines and monoamine metabolites. J Neural Transm 87: 215–224.
Wurtman RJ, Larin F, Mostafapour S, Fernstrom JD (1974). Brain catechol synthesis: control by train tyrosine concentration. Science 185: 183–184.
Zetterström T, Sharp T, Ungerstedt U (1986). Further evaluation of the mechanism by which amphetamine reduces striatal dopamine metabolism: a brain dialysis study. Eur J Pharmacol 132: 1–9.
Zielke HR, Zielke CL, Baab PJ, Collins RM (2002). Large neutral amino acids auto exchange when infused by microdialysis into the rat brain: implication for maple syrup urine disease and phenylketonuria. Neurochem Int 40: 347–354.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Le Masurier, M., Oldenzeil, W., Lehman, C. et al. Effect of Acute Tyrosine Depletion in Using a Branched Chain Amino-Acid Mixture on Dopamine Neurotransmission in the Rat Brain. Neuropsychopharmacol 31, 310–317 (2006). https://doi.org/10.1038/sj.npp.1300835
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/sj.npp.1300835
Keywords
This article is cited by
-
Acute phenylalanine/tyrosine depletion of phasic dopamine in the rat brain
Psychopharmacology (2016)
-
Acute oral administration of a tyrosine and phenylalanine-free amino acid mixture reduces exercise capacity in the heat
European Journal of Applied Physiology (2013)