Abstract
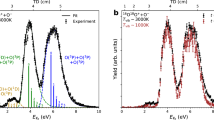
IT is not often that evidence in the form of steric hindrance is available for confirming the configurations of geometrical isomers among ethylenic compounds. The following observations are thus of interest. cis--Dibromocinnamic acid1 (m.p. 100°) esterifies to the extent of only 6 per cent in one hour, and 33 per cent in 16 hours, by the Fischer-Speier method, whereas the trans-acid1 (m.p. 136°) is esterified completely in one hour. This is in conformity with my earlier observation2 in regard to the different modes of addition of bromine to phenylpropiolic acid and its methyl ester.
Similar content being viewed by others
Article PDF
References
Annalen, 247, 139.
Ind. Sci. Congr. Chem. Abstr., No.75, 1934.
J. Chem. Soc., 83, 668.
Ber., 24, 251.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
AYYAR, P. Steric Hindrance and Geometrical Isomerism. Nature 134, 535 (1934). https://doi.org/10.1038/134535b0
Issue date:
DOI: https://doi.org/10.1038/134535b0