Abstract
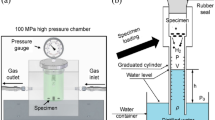
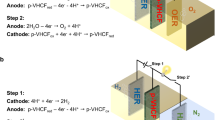
THE hydrogen electrode is the primary standard to which all electrode potentials are referred and is used in cells without liquid junction for measurements of high accuracy. The potential of the hydrogen electrode is corrected to the standard pressure of one atmosphere on the basis that the electrode is in equilibrium with the measured partial pressure of hydrogen above the solution. It is, however, not the gas phase which directly determines the potential, but the hydrogen in solution ; and a tacit assumption that the concentration of dissolved hydrogen is unequivocally defined by the gas pressure is false. This error can arise in two ways.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
HILLS, G., IVES, D. The Hydrogen Electrode. Nature 163, 997 (1949). https://doi.org/10.1038/163997a0
Issue date:
DOI: https://doi.org/10.1038/163997a0
This article is cited by
-
Transference numbers of aqueous HCl up to 15 mol-kg?1 at 25�C: A systematization and reassessment
Journal of Solution Chemistry (1995)