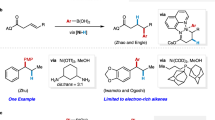
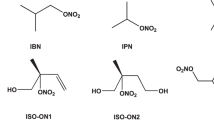
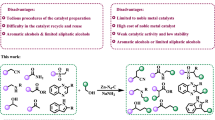
Abstract
EARLY work1 gives a very confused picture of the reactions which occur in either alkaline or acid hydrolysis of organic esters of nitric acid. The only clearly established point is the occurrence of reactions other than simple fission to alcohol and nitric acid, and the general view was that such side-reactions, including the formation of nitrite, arise from subsequent oxidation of the alcohol by the nitric acid. A more recent study, by Lucas and Hammett2, of the solvolysis of tert.-butyl nitrate revealed the concomitant formation of isobutene, while with benzyl nitrate, benzaldehyde and nitrite are also formed.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Cf., inter alia, Farmer, J. Chem. Soc., 117, 806 (1920); and references cited there.
J. Amer. Chem. Soc., 64, 1928 (1942).
For summarizing papers, cf. Hughes, Trans. Farad. Soc., 37, 603 (1941); Hughes and Ingold, ibid., 657.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
BAKER, J., EASTY, D. Hydrolysis of Organic Nitrates. Nature 166, 156 (1950). https://doi.org/10.1038/166156a0
Issue date:
DOI: https://doi.org/10.1038/166156a0