Abstract
Study design:
Controlled, repeat-measures study.
Objectives:
To determine if functional electrical stimulation (FES) can affect bone atrophy in early spinal cord injury (SCI), and the safety, tolerance and feasibility of this modality in bone loss remediation.
Setting:
Spinal Injuries Units, Royal Adelaide Hospital and Hampstead Rehabilitation Centre, South Australia.
Methods:
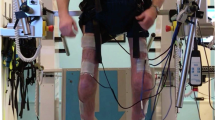
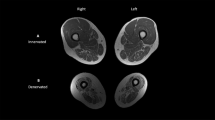
Patients with acute SCI (ASIA A–D) were allocated to FES (n=23, 28±9 years, C4–T10, 13 Tetra) and control groups (CON, n=10, 31±11 years, C5–T12, four Tetra). The intervention group received discontinuous FES to lower limb muscles (15 min sessions to each leg twice daily, over a 5-day week, for 5 months). Dual energy X-ray absorptiometry (DEXA) measured total body bone mineral density (tbBMD), hip, spine BMD and fat mass (FM) within 3 weeks, and 3 and 6 months postinjury.
Results:
FES and CON groups' tbBMD differed significantly at 3 months postinjury (P<0.01), but not thereafter. Other DEXA measures (hip, spine BMD, FM) did not differ between groups at any time. No adverse events were identified.
Conclusion:
Electrically stimulated muscle activation was elicited, and tetanic effects were reproducible; however, there were no convincing trends to suggest that FES can play a clinically relevant role in osteoporosis prevention (or subsequent fracture risk) in the recently injured patient. The lack of an osteogenic response in paralysed extremities to electrically evoked exercise during subacute and rehabilitation/recovery phases cannot be fully explained, and may warrant further evaluation.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Basmajian JV . Muscles Alive. Their Functions Revealed by Electromyography, 5th edn. Williams and Wilkins 1985.
Wallin BG, Fagius J . The sympathetic nervous system in man-aspects derived from micro-electrode recordings. Trends Neurosci 1986; 9: 63–67.
Woolsey CN . Cortical localisation as defined by evoked potential and electrical stimulation studies. In: Schaltenbrand G, Woolsey CN (eds). Cerebral Localisation and Organisation. University of Wisconsin Press: Wisconsin, MI 1964, pp 17–32.
Baldi JC, Jackson RD, Morallie R, Mysiw WJ . Muscle atrophy is prevented in patients with acute spinal cord injury using functional electrical stimulation. Spinal Cord 1998; 36: 463–469.
Nash MS et al. Reversal of adaptive left ventricular atrophy following electrically-stimulated exercise training in human tetrataplegics. Paraplegia 1991; 29: 590–599.
Mohr T, Anderson JL, Beiring-Sorensen F, Galbo H, Wagner A, Kjaer M . Long term adaptation to electrically induced cycle training in severe spinal cord injured individuals. Spinal Cord 1997; 35: 1–16.
Hangartner TN, Rodgers MM, Glaser RM, Barre PS . Tibial bone density loss in spinal cord injured patients: effects of FES exercise. J Rehab Res Dev 1994; 31: 50–61.
Talmadge RJ, Castro MJ, Apple Jr DF, Dudley GA . Phenotypic adaptations in human muscle fibres 6 and 24 weeks after spinal cord injury. J Appl Physiol 2002; 92: 142–154.
Hjeltnes N et al. Exercise-induced overexpression of key regulatory proteins involved in glucose uptake and metabolism in tetraplegic persons: molecular mechanisms for improved glucose homeostasis. FASEB J 1998; 12: 1701–1712.
Kler M et al. Regulation of glucose turnover and hormonal responses during electrical cycling in tetraplegic patients. Am J Physiol 1996; 271: R191–R199.
De Bell KK, Scremin AM, Perell KL, Kunkel CF . Effects of functional electrical stimulation-induced lower extremity cycling on bone density of spinal cord-injured patients. Am J Phys Med Rehab 1996; 75: 29–34.
Mohr T, Podenphant J, Biering-Sorensen F, Galbo H, Thamsborg G, Kjaer M . Increased bone mineral density after prolonged electrically induced cycle training of paralysed limbs in spinal cord injured man. Calcif Tiss Int 1997; 61: 22–25.
Eser P, de Bruin ED, Telley I, Lechner HE, Knecht H, Stussi E . Effect of electrical stimulation-induced cycling on bone mineral density in spinal cord injured patients. Eur J Clin Invest 2003; 33: 412–419.
Maimoun L et al. Changes in osteoprotegerin /RANKL system, bone mineral density, and bone biochemical markers in patients with recent spinal cord injury. Calcif Tissue Int 2005; 76: 404–411.
Roberts D et al. Longitudinal study of bone turnover after acute spinal cord injury. J Clin Endocrinol 1998; 83: 415–422.
Mechanick JI, Pomerantz F, Flanagan S, Atein A, Gordon WA, Ragnarsson KT . Parathyroid hormone suppression in spinal cord injury patients is associated with degree of neurologic impairment and not the level of impairment. Arch Phys Med Rehab 1997; 78: 692–696.
Garland DE et al. Osteoporosis after spinal cord injury. J Orthop Res 1992; 10: 371–378.
Eser P et al. Relationship between the duration of paralysis and bone structure: a pQCT study of spinal cord injured individuals. Bone 2004; 34: 869–880.
Garland DE, Foulkes GD, Adkins RH, Stewart CA, Yakura JS . Regional osteoporosis following incomplete spinal cord injury. Contemp Orthop 1994; 28: 134–139.
Dauty M, Verbe BP, Maugars Y, Dubois C, Mathe JF . Supralesional and sublesional bone mineral density in spinal cord-injured patients. Bone 2000; 27: 305–309.
Zhender Y et al. Prevention of bone loss in paraplegics over 2 years with alendronate. J Bone Miner Res 2004; 19: 1067–1074.
Pearson EG, Nance PW, Leslie WD, Ludwig S . Cyclical etidronate: its effect on bone density in patients with acute spinal cord injury. Arch Phys Med Rehab 1997; 79: 269–273.
Chen B, Mechanick JI, Nierman DM, Stein A . Combined calcitriol–pamidronate therapy for bone hyper-resorption in spinal cord injury. J Spinal Cord Med 2001; 24: 235–240.
Moran de Brito CM, Battistella LR, Saito ET, Sakamoto H . Effect of alendronate on bone mineral density in spinal cord injury patients: a pilot study. Spinal Cord 2005; 43: 341–348.
Bracken MB et al. A randomized controlled trial of methylprednisolone or naloxone in the treatment of acute spinal cord injury. Results of the Second National Acute Spinal Cord Injury Study. N Engl J Med 1990; 322: 1405–1411.
The Uniform Data System for Medical Rehabilitation. The Functional Independence Measure (FIM sm) Participant Manual, Version 5. 0 (Australia). State University of New York: Buffalo, NY 1999.
Stover SL, Apple DF, Donovan WH, Maynard FM (eds). International Standards for Neurological and Functional Classification of Spinal Cord Injury. American Spinal Injury Association: Chicago, IL 1996.
Jones LM, Goulding A, Gerrard DF . DEXA: A practical and accurate tool to demonstrate total and regional bone loss, lean tissue loss and fat mass gain in paraplegia. Spinal Cord 1998; 36: 637–640.
Jaovisidha S, Sarotis DJ, Martin EME, Foldes K, Szollar SM, Deftos LJ . Influence of heterotopic ossification of the hip on bone densitometry: a study in spinal cord injured patients. Spinal Cord 1998; 36: 647–653.
De Bruin ED, Frey-Rindova P, Herzog RE, Deitz V, Dambacher MA, Stuhl E . Changes to tibia bone properties after spinal cord injury: effects of early intervention. Arch Phys Med Rehab 1999; 80: 214–220.
Warden SJ, Bennell KL, Matthews B, Brown DJ, McMeeken JM, Wark L . Efficacy of low-intensity pulsed ultrasound in the prevention of osteoporosis following spinal cord injury. Bone 2001; 29: 431–436.
Alsop WK, Caulton J, Rubin C, Admans J, Mughal Z . Low magnitude mechanical loading is osteogenic in children with disabling conditions. J Bone Miner Res 2004; 19: 360–369.
Rubin C et al. Quality and quantity of trabecular bone in the femur are enhanced by a strongly anabolic, noninvasive mechanical intervention. J Bone Miner Res 2002; 17: 349–357.
Bizzarini E, Saccavini M, Lipanje F, Magrin P, Malisan C, Zampa A . Exercise prescription in subjects with spinal cord injuries. Arch Phys Med Rehab 2005; 86: 1170–1175.
Huang TS, Wang YH, Chiang HS, Lien YN . Pituitary-testicular and pituitary–thyroid axes in spinal cord-injured males. Metabolism 1993; 42: 516–521.
Wang YH, Huang TS, Lien IN . Hormone changes in men with spinal cord injuries A. J Phys Med Rehab 1992; 71: 328–332.
Tsitouros PD, Zhang YG, Spungen AM, Baumann WA . Serum testosterone and growth hormone/insulin-like growth factor-1 in adults with spinal cord injury. Horm Metab Res 1995; 27: 287–292.
Yang L, Blumbergs PC, Jones NR, Manavis J, Sarvestini GT, Ghabriel MN . Early expression and cellular localization of proinflammatory cytokines interleukin-1-beta, interleukin-6 and tumor necrosis factor-alpha in human traumatic spinal cord injury. Spine 2004; 29: 966–971.
Gerrits HL, Hopman MT, Sargent AJ, De Haan A . Reproducibility of contractile properties of the human paralysed and non-paralysed quadriceps muscle. Clin Physiol 2001; 21: 105–113.
Bolotin HH, Sievanen H . Inaccuracies inherent in dual-energy X-ray absorptiometry in vivo bone mineral density can seriously mislead diagnostic/prognostic interpretations of patient-specific bone fragility. J Bone Miner Res 2001; 16: 799–805.
La Porte RE, Montoyne HJ, Casperson CJ . Assessment of physical activity in epidemiological research: problems and prospects. Public Health Report 1985; 100: 131–146.
Desharnais R, Jobin J, Cote C, Levesque L, Godin G . Aerobic exercise and the placebo effect: a controlled study. Psychosomatic Med 1993; 55: 149–155.
Freehafer AA, Mast WA . Lower extremity fractures in patients with spinal cord injury. J Bone Joint Surg 1965; 47-A: 683–694.
Frisbie J . Fractures after myelopathy: the risk quantified. J Spinal Cord Med 1997; 20: 66–69.
Vestergaarde P, Krogh K, Rejnmark L, Modekilde L . Fracture rates and risk factors for fractures in patients with spinal cord injury. Spinal Cord 1998; 36: 790–796.
Ragnarsson KT, Sell GH . Lower extremity fractures of patients with spinal cord injuries. Arch Phys Med Rehab 1981; 62: 418–423.
Minaire P, Edouard C, Arlot M, Meunier PJ . Marrow changes in paraplegic patients. Calcif Tiss Int 1984; 36: 338–340.
Gu Y, Genever PG, Skerry TM, Publicover SJ . The NMDA type glutamate receptors expressed by primary rat osteoblasts have the same electrophysiological characteristics as neuronal receptors. Calcif Tiss Int 2002; 70: 194–203.
Kooijman M, Rongen GA, Smits P, Hopman MTE . Preserved alpha-adrenergic tone in the leg vascular bed of spinal cord injured individuals. Circulation 2003; 108: 2361–2363.
Quin T, Guo X, Levi AD, Vanni S, Shebert RT, Sipski ML . High-dose methylprednisolone may cause myopathy in acute spinal cord injury patients. Spinal Cord 2005; 43: 199–203.
Huang TS, Wang YH, Lee SH, Lai JS . Impaired hypothalamic–pituitary–adrenal axis in men with spinal cord injuries. Am J Phys Med Rehab 1998; 77: 108–112.
DeGroot PCE, Van Kuppevelt DHJM, Snoek G, Van Der Woude LHV, Hopman MTE . Time course of arterial vascular adaptations to inactivity and paralyses in humans. Med Sci Sports Exerc 2003; 35: 1977–1985.
Hopman MT, van Ansten WN, Oeseburg B . Changes in blood flow in the common femoral artery related to inactivity and muscle atrophy in individuals with long- standing paraplegia. Adv Exp Med Biol 1996; 388: 379–383.
Boot CR, Groothuis JT, Van Langen H, Hopman MT . Shear stress levels in paralyzed legs of spinal cord-injured individuals with and without nerve degeneration. J App Physiol 2002; 92: 2335–2340.
Acknowledgements
We acknowledge the excellent technical support of Ms Trish Wesley, of the Department of Nuclear Medicine, Royal Adelaide Hospital and Ms Sheelagh Donohoe, Ms Julie Wheeler, Ms Marie Hogg and Ms Annette van den Broek of the FES Clinic, Hampstead Rehabilitation Centre. The study was funded by a grant from the Motor Accident Commission of South Australia and the State Government of South Australia. The protocol was approved by the Royal Adelaide Hospital Human Research Ethics Committee, and conducted in accordance with the guidelines of the Declaration of Helsinki.
Author information
Authors and Affiliations
Additional information
We certify that all applicable institutional and governmental regulations concerning the ethical use of human volunteers were followed during the course of this research
Rights and permissions
About this article
Cite this article
Clark, J., Jelbart, M., Rischbieth, H. et al. Physiological effects of lower extremity functional electrical stimulation in early spinal cord injury: lack of efficacy to prevent bone loss. Spinal Cord 45, 78–85 (2007). https://doi.org/10.1038/sj.sc.3101929
Published:
Issue date:
DOI: https://doi.org/10.1038/sj.sc.3101929
Keywords
This article is cited by
-
Motorless cadence control of standard and low duty cycle-patterned neural stimulation intensity extends muscle-driven cycling output after paralysis
Journal of NeuroEngineering and Rehabilitation (2022)
-
Selective neural stimulation methods improve cycling exercise performance after spinal cord injury: a case series
Journal of NeuroEngineering and Rehabilitation (2021)
-
Evidence-based prevention and treatment of osteoporosis after spinal cord injury: a systematic review
European Spine Journal (2018)
-
Fabrication and in vitro biological properties of piezoelectric bioceramics for bone regeneration
Scientific Reports (2017)
-
Musculoskeletal Health in the Context of Spinal Cord Injury
Current Osteoporosis Reports (2017)