Abstract
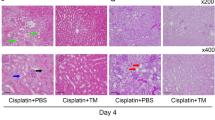
Sensory neuropathies are a frequent and dose-limiting complication resulting from treatment with cisplatin. Neurotrophin-3 (NT-3) promotes the survival of the large fiber sensory neurones that are impaired in cisplatin-induced neuropathy, and may therefore serve as a preventive agent. However, the short half-life of recombinant NT-3 after systemic administration limits its clinical applications. We compared two muscle-based gene transfer strategies for the continuous delivery of NT-3 to the bloodstream in an experimental model of cisplatin-induced neuropathy. Electrophysiological studies showed that the intramuscular injection of an adenovirus encoding NT-3 partially prevented the cisplatin-induced increase in sensory distal latencies. Similar effects were observed in cisplatin-treated mice that received intramuscular injections of a plasmid encoding NT-3 associated with in vivo electroporation. The two techniques were well tolerated and induced only slight muscle toxicity. Measurement of renal function, weight and survival showed that neither technique increased the toxicity of cisplatin. Our study shows that gene therapy, using either a viral or a non-viral vector, is a promising strategy for the prevention of cisplatin-induced neuropathy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 print issues and online access
$259.00 per year
only $43.17 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Thompson SW, Davis LE, Kornfeld M, Hilgers RD, Standefer JC . Cisplatin neuropathy. Clinical, electrophysiologic, morphologic, and toxicologic studies Cancer 1984 54: 1269–1275
DiStefano PS et al. The neurotrophins BDNF, NT-3 and NGF display distinct patterns of retrograde axonal transport in peripheral and central neurons Neuron 1992 8: 983–993
Gregg RW et al. Cisplatin neurotoxicity: the relationship between dosage, time, and platinum concentration in neurologic tissues, and morphologic evidence of toxicity J Clin Oncol 1992 10: 795–803
Olsson Y . Vascular permeability in the peripheral nervous system Dyck PJ (eds); Peripheral Neuropathy WB Saunders 1984 pp 579–597
Gao WQ et al. Neurotrophin-3 reverses experimental cisplatin-induced peripheral sensory neuropathy Ann Neurol 1995 38: 30–37
Helgren ME et al. Neurotrophin-3 administration attenuates deficits of pyridoxine-induced large-fiber sensory neuropathy J Neurosci 1997 17: 372–382
Poduslo JF, Curran GL . Permeability at the blood-brain and blood-nerve barriers of the neurotrophic factors: NGF, CNTF, NT3, BDNF Mol Brain Res 1996 36: 280–286
Pradat PF et al. Partial prevention of cisplatin-induced neuropathy by electroporation-mediated nonviral gene transfer Hum Gene Ther 2001 12: 367–375
Haase G et al. Gene therapy of murine motor neuron disease using adenoviral vectors for neurotrophic factors Nat Med 1997 3: 429–436
Pradat PF et al. Continuous delivery of NT-3 by gene therapy has a neuroprotective effect in experimental models of diabetic and acrylamide neuropathies Hum Gene Ther 2001 12: 2237–2249
Chattopadhyay M et al. In vivo gene therapy for pyridoxine-induced neuropathy by herpes simplex virus-mediated gene transfer of neurotrophin-3 Ann Neurol 2002 51: 19–27
Windebank AJ . Use of growth factors in the treatment of motor neuron diseases Adv Neurol 1995 68: 229–234
Rodriguez-Pena A, Botana M, Gonzalez M, Requejo F . Expression of neurotrophins and their receptors in sciatic nerve of experimentally diabetic rats Neurosci Lett 1995 200: 37–40
Ihara C et al. Decreased neurotrophin-3 expression in skeletal muscles of streptozotocin-induced diabetic rats Neuropeptides 1996 30: 309–312
Rodriguez-Pena A, Botana M, Gonzalez M, Requejo F . Expression of neurotrophins and their receptors in sciatic nerve of experimentally diabetic rats Neurosci Lett 1995 200: 37–40
McMahon SB, Priestley JV . Peripheral neuropathies and neurotrophic factors: animal models and clinical perspectives Curr Opin Neurobiol 1995 5: 616–624
Cavaletti G et al. Neuroprotectant drugs in cisplatin neurotoxicity Anticancer Res 1996 16: 3149–3160
Oh SJ . 2nd edn Williams& Wilkins 1993
Schmalbruch H, Krarup C . Animal models of neuropathy Baillieres Clin Neurol 1996 5: 77–105
Verdu E et al. Physiological and immunohistochemical characterization of cisplatin-induced neuropathy in mice Muscle Nerve 1999 22: 329–340
Dai Y et al. Cellular and humoral immune responses to adenoviral vectors containing factor IX gene: tolerization of factor IX and vector antigens allows for long-term expression Proc Natl Acad Sci USA 1995 92: 1401–1405
Aihara H, Miyazaki J . Gene transfer into muscle by electroporation in vivo Nat Biotechnol 1998 16: 867–870
Maruyama H et al. Continuous erythropoietin delivery by muscle-targeted gene transfer using in vivo electroporation Hum Gene Ther 2000 11: 429–437
Herzog RW et al. Long-term correction of canine hemophilia B by gene transfer of blood coagulation factor IX mediated by adeno-associated viral vector Nat Med 1999 5: 56–63
Muramatsu T et al. In vivo gene electroporation in skeletal muscle with special reference to the duration of gene expression Int J Mol Med 2001 7: 37–42
Apfel SC et al. Nerve growth factor administration protects against experimental diabetic sensory neuropathy Brain Res 1994 634: 7–12
Yuen EC, Mobley WC . Therapeutic applications of neurotrophic factors in disorders of motor neurons and peripheral nerves Mol Med Today 1995 1: 278–286
Apfel SC . Efficacy and safety of recombinant human nerve growth factor in patients with diabetic polyneuropathy: a randomized controlled trial JAMA 2000 284: 2215–2221
Wolfe D, Goins WF, Kaplan TJ . Herpesvirus-mediated systemic delivery of nerve growth factor Mol Ther 2001 3: 61–69
Simovic D et al. Improvement in chronic ischemic neuropathy after intramuscular phVEGF165 gene transfer in patients with critical limb ischemia Arch Neurol 2001 58: 761–768
Crouzet J et al. Recombinational construction in Escherichia coli of infectious adenoviral genomes Proc Natl Acad Sci USA 1997 94: 1414–1419
Acknowledgements
We thank Caterine Dubertret for developing the in vivo electrotransfer technique; Isabelle Loquet, Estelle Arnould and Delphine Casanova for excellent technical support; Leïla Houhou and Sylvie Berrard for critically reading the manuscript. This work was supported by the Centre National de la Recherche Scientifique and the Institut pour la Recherche sur la Moelle Epinière (IRME).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pradat, PF., Kennel, P., Naimi-Sadaoui, S. et al. Viral and non-viral gene therapy partially prevents experimental cisplatin-induced neuropathy. Gene Ther 9, 1333–1337 (2002). https://doi.org/10.1038/sj.gt.3301801
Received:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/sj.gt.3301801
Keywords
This article is cited by
-
Astaxanthin reduces oxidative stress and alleviates diabetic neuropathy in STZ-induced diabetic mice
International Journal of Diabetes in Developing Countries (2023)
-
Gene therapy for the treatment of diabetic neuropathy
Current Diabetes Reports (2008)
-
Prolonged preservation of nerve function in diabetic neuropathy in mice by herpes simplex virus-mediated gene transfer
Diabetologia (2007)