Abstract
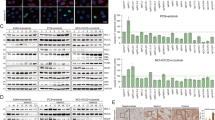
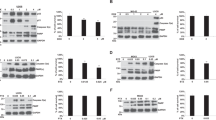
A significant variation in susceptibility to paclitaxel-mediated killing was observed among a panel of short-term cultured non-small-cell lung cancer (NSCLC) cell lines. Susceptibility to killing by paclitaxel correlated with expression of the BH3-only protein, Bim, but not with other members of Bcl-2 family. NSCLC cell lines with the highest level of Bim expression are most susceptible to apoptosis induction after paclitaxel treatment. Forced expression of Bim increased paclitaxel-mediated killing of cells expressing an undetectable level of Bim. Conversely, knock down of Bim, but not Bcl-2 expression, decreased the susceptibility of tumor cells to paclitaxel-mediated killing. Similar observations were made using a panel of breast and prostate cancer cell lines. Paclitaxel impairs microtubule function, causes G2/M cell cycle blockade, mitochondria damage, and p53-independent apoptosis. These results established Bim as a critical molecular link between the microtubule poison, paclitaxel, and apoptosis.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Abbreviations
- BH:
-
Bcl-2 homologous (domain)
- siRNA:
-
small interfering RNA
References
Green MR (2001) The current status of docetaxel for advanced non-small cell lung cancer. Anticancer Drugs 12 (Suppl 1): S11–S16
Basu A and Haldar S (2003) Identification of a novel Bcl-xL phosphorylation site regulating the sensitivity of taxol- or 2-methoxyestradiol-induced apoptosis. FEBS Lett. 538: 41–47
Basu A, Mohanty S and Sun B (2001) Differential sensitivity of breast cancer cells to tumor necrosis factor-alpha: involvement of protein kinase C. Biochem. Biophys. Res. Commun. 280: 883–891
Burns TF, Fei P, Scata KA, Dicker DT and El-Deiry WS (2003) Silencing of the novel p53 target gene Snk/Plk2 leads to mitotic catastrophe in paclitaxel (taxol)-exposed cells. Mol. Cell. Biol. 23: 5556–5571
Fan S, Cherney B, Reinhold W, Rucker K and O'Connor PM (1998) Disruption of p53 function in immortalized human cells does not affect survival or apoptosis after taxol or vincristine treatment. Clin. Cancer Res. 4: 1047–1054
Wahl AF, Donaldson KL, Fairchild C, Lee FY, Foster SA, Demers GW and Galloway DA (1996) Loss of normal p53 function confers sensitization to taxol by increasing G2/M arrest and apoptosis. Nat. Med. 2: 72–79
Strasser A, Puthalakath H, Bouillet P, Huang DC, O'Connor L, O'Reilly LA, Cullen L, Cory S and Adams JM (2000) The role of bim, a proapoptotic BH3-only member of the Bcl-2 family in cell-death control. Ann. NY Acad. Sci. 917: 541–558
Green DR and Reed JC (1998) Mitochondria and apoptosis. Science 281: 1309–1312
Bouillet P, Purton JF, Godfrey DI, Zhang LC, Coultas L, Puthalakath H, Pellegrini M, Cory S, Adams JM and Strasser A (2002) BH3-only Bcl-2 family member Bim is required for apoptosis of autoreactive thymocytes. Nature 415: 922–996
Cory S and Adams JM (2002) The Bcl2 family: regulators of the cellular life-or-death switch. Nat. Rev. Cancer 2: 647–656
Puthalakath H and Strasser A (2002) Keeping killers on a tight leash: transcriptional and post-translational control of the pro-apoptotic activity of BH3-only proteins. Cell Death Differ. 9: 505–512
Coultas L, Huang DC, Adams JM and Strasser A (2002) Pro-apoptotic BH3-only Bcl-2 family members in vertebrate model organisms suitable for genetic experimentation. Cell Death Differ. 9: 1163–1166
Wei MC, Zong WX, Cheng EH, Lindsten T, Panoutsakopoulou V, Ross AJ, Roth KA, MacGregor GR, Thompson CB and Korsmeyer SJ (2001) Proapoptotic BAX and BAK: a requisite gateway to mitochondrial dysfunction and death. Science 292: 727–730
Tsujimoto Y (2003) Cell death regulation by the Bcl-2 protein family in the mitochondria. J. Cell. Physiol. 195: 158–167
Srivastava RK, Mi QS, Hardwick JM and Longo DL (1999) Deletion of the loop region of Bcl-2 completely blocks paclitaxel-induced apoptosis. Proc. Natl. Acad. Sci. USA 96: 3775–3780
Srivastava RK, Sasaki CY, Hardwick JM and Longo DL (1999) Bcl-2-mediated drug resistance: inhibition of apoptosis by blocking nuclear factor of activated T lymphocytes (NFAT)-induced Fas ligand transcription. J. Exp. Med. 190: 253–265
Yamamoto K, Ichijo H and Korsmeyer SJ (1999) BCL-2 is phosphorylated and inactivated by an ASK1/Jun N-terminal protein kinase pathway normally activated at G(2)/M. Mol. Cell. Biol. 19: 8469–8478
Huisman C, Ferreira CG, Broker LE, Rodriguez JA, Smit EF, Postmus PE, Kruyt FA and Giaccone G (2002) Paclitaxel triggers cell death primarily via caspase-independent routes in the non-small cell lung cancer cell line NCI-H460. Clin. Cancer Res. 8: 596–606
Puthalakath H, Huang DC, O'Reilly LA, King SM and Strasser A (1999) The proapoptotic activity of the Bcl-2 family member Bim is regulated by interaction with the dynein motor complex. Mol. Cell 3: 287–296
Bouillet P, Metcalf D, Huang DC, Tarlinton DM, Kay TW, Kontgen F, Adams JM and Strasser A (1999) Proapoptotic Bcl-2 relative Bim required for certain apoptotic responses, leukocyte homeostasis, and to preclude autoimmunity. Science 286: 1735–1738
Broker LE, Huisman C, Ferreira CG, Rodriguez JA, Kruyt FA and Giaccone G (2002) Late activation of apoptotic pathways plays a negligible role in mediating the cytotoxic effects of discodermolide and epothilone B in non-small cell lung cancer cells. Cancer Res. 62: 4081–4088
Song Z, Yao X and Wu M (2003) Direct interaction between survivin and Smac/DIABLO is essential for the anti-apoptotic activity of survivin during taxol-induced apoptosis. J. Biol. Chem. 278: 23130–23140
Yuan SY, Hsu SL, Tsai KJ and Yang CR (2002) Involvement of mitochondrial pathway in Taxol-induced apoptosis of human T24 bladder cancer cells. Urol. Res. 30: 282–288
Blajeski AL, Kottke TJ and Kaufmann SH (2001) A multistep model for paclitaxel-induced apoptosis in human breast cancer cell lines. Exp. Cell Res. 270: 277–288
Cheng EH, Wei MC, Weiler S, Flavell RA, Mak TW, Lindsten T and Korsmeyer SJ (2001) BCL-2, BCL-X(L) sequester BH3 domain-only molecules preventing BAX- and BAK-mediated mitochondrial apoptosis. Mol. Cell 8: 705–711
Wang TH, Wang HS and Soong YK (2000) Paclitaxel-induced cell death: where the cell cycle and apoptosis come together. Cancer 88: 2619–2628
Wassmann K and Benezra R (2001) Mitotic checkpoints: from yeast to cancer. Curr. Opin. Genet. Dev. 11: 83–90
Taylor SS and McKeon F (1997) Kinetochore localization of murine Bub1 is required for normal mitotic timing and checkpoint response to spindle damage. Cell 89: 727–735
Anand S, Penrhyn-Lowe S and Venkitaraman AR (2003) AURORA-A amplification overrides the mitotic spindle assembly checkpoint, inducing resistance to taxol. Cancer Cell 3: 51–62
Masuda A, Maeno K, Nakagawa T, Saito H and Takahashi T (2003) Association between mitotic spindle checkpoint impairment and susceptibility to the induction of apoptosis by anti-microtubule agents in human lung cancers. Am. J. Pathol. 163: 1109–1116
Ofir R, Seidman R, Rabinski T, Krup M, Yavelsky V, Weinstein Y and Wolfson M (2002) Taxol-induced apoptosis in human SKOV3 ovarian and MCF7 breast carcinoma cells is caspase-3 and caspase-9 independent. Cell Death Differ. 9: 636–642
Hildeman DA, Zhu Y, Mitchell TC, Bouillet P, Strasser A, Kappler J and Marrack P (2002) Activated T cell death invivo mediated by proapoptotic bcl-2 family member bim. Immunity 16: 759–767
Putcha GV, Moulder KL, Golden JP, Bouillet P, Adams JA, Strasser A and Johnson EM (2001) Induction of BIM, a proapoptotic BH3-only BCL-2 family member, is critical for neuronal apoptosis. Neuron 29: 615–628
von Haefen C, Wieder T, Essmann F, Schulze-Osthoff K, Dorken B and Daniel PT (2003) Paclitaxel-induced apoptosis in BJAB cells proceeds via a death receptor-independent, caspases-3/-8-driven mitochondrial amplification loop. Oncogene 22: 2236–2247
Wolf BB, Schuler M, Li W, Eggers-Sedlet B, Lee W, Tailor P, Fitzgerald P, Mills GB and Green DR (2001) Defective cytochrome c-dependent caspase activation in ovarian cancer cell lines due to diminished or absent apoptotic protease activating factor-1 activity. J. Biol. Chem. 276: 34244–34251
Liu JR, Opipari AW, Tan L, Jiang Y, Zhang Y, Tang H and Nunez G (2002) Dysfunctional apoptosome activation in ovarian cancer: implications for chemoresistance. Cancer Res. 62: 924–931
Joseph B, Ekedahl J, Lewensohn R, Marchetti P, Formstecher P and Zhivotovsky B (2001) Defective caspase-3 relocalization in non-small cell lung carcinoma. Oncogene 20: 2877–2888
Joseph B, Marchetti P, Formstecher P, Kroemer G, Lewensohn R and Zhivotovsky B (2002) Mitochondrial dysfunction is an essential step for killing of non-small cell lung carcinomas resistant to conventional treatment. Oncogene 21: 65–77
Egger L, Schneider J, Rheme C, Tapernoux M, Hacki J and Borner C (2003) Serine proteases mediate apoptosis-like cell death and phagocytosis under caspase-inhibiting conditions. Cell Death Differ. 10: 1188–1203
Vande VC, Cizeau J, Dubik D, Alimonti J, Brown T, Israels S, Hakem R and Greenberg AH (2000) BNIP3 and genetic control of necrosis-like cell death through the mitochondrial permeability transition pore. Mol. Cell. Biol. 20: 5454–5468
Guo K, Searfoss G, Krolikowski D, Pagnoni M, Franks C, Clark K, Yu KT, Jaye M and Ivashchenko Y (2001) Hypoxia induces the expression of the pro-apoptotic gene BNIP3. Cell Death Differ. 8: 367–376
Arnt CR, Chiorean MV, Heldebrant MP, Gores GJ and Kaufmann SH (2002) Synthetic Smac/DIABLO peptides enhance the effects of chemotherapeutic agents by binding XIAP and cIAP1 insitu. J. Biol. Chem. 277: 44236–44243
McNeish IA, Bell S, McKay T, Tenev T, Marani M and Lemoine NR (2003) Expression of Smac/DIABLO in ovarian carcinoma cells induces apoptosis via a caspase-9-mediated pathway. Exp. Cell Res. 286: 186–198
Yang L, Mashima T, Sato S, Mochizuki M, Sakamoto H, Yamori T, Oh-Hara T and Tsuruo T (2003) Predominant suppression of apoptosome by inhibitor of apoptosis protein in non-small cell lung cancer H460 cells: therapeutic effect of a novel polyarginine-conjugated Smac peptide. Cancer Res. 63: 831–837
O'Connor DS, Wall NR, Porter AC and Altieri DC (2002) A p34(cdc2) survival checkpoint in cancer. Cancer Cell 2: 43–54
O'Reilly LA, Cullen L, Visvader J, Lindeman GJ, Print C, Bath ML, Huang DC and Strasser A (2000) The proapoptotic BH3-only protein bim is expressed in hematopoietic, epithelial, neuronal, and germ cells. Am. J. Pathol. 157: 449–461
O'Connor L, Strasser A, O'Reilly LA, Hausmann G, Adams JM, Cory S and Huang DC (1998) Bim: a novel member of the Bcl-2 family that promotes apoptosis. EMBO J. 17: 384–395
Dijkers PF, Medema RH, Lammers JW, Koenderman L and Coffer PJ (2000) Expression of the pro-apoptotic Bcl-2 family member Bim is regulated by the forkhead transcription factor FKHR-L1. Curr. Biol. 10: 1201–1204
Gilley J, Coffer PJ and Ham J (2003) FOXO transcription factors directly activate bim gene expression and promote apoptosis in sympathetic neurons. J. Cell Biol. 162: 613–622
Sunters A, Fernandez De Mattos S, Stahl M, Brosens JJ, Zoumpoulidou G, Saunders CA, Coffer PJ, Medema RH, Coombes RC and Lam EW (2003) FoxO3a transcriptional regulation of Bim controls apoptosis in paclitaxel treated breast cancer cell lines. J. Biol. Chem.
Putcha GV, Le S, Frank S, Besirli CG, Clark K, Chu B, Alix S, Youle RJ, LaMarche A, Maroney AC and Johnson Jr EM. (2003) JNK-mediated BIM phosphorylation potentiates BAX-dependent apoptosis. Neuron 38: 899–914
Lei K and Davis RJ (2003) JNK phosphorylation of Bim-related members of the Bcl2 family induces Bax-dependent apoptosis. Proc. Natl. Acad. Sci. USA 100: 2432–2437
Weston CR, Balmanno K, Chalmers C, Hadfield K, Molton SA, Ley R, Wagner EF and Cook SJ (2003) Activation of ERK1/2 by deltaRaf-1:ER* represses Bim expression independently of the JNK or PI3K pathways. Oncogene 22: 1281–1293
Ley R, Balmanno K, Hadfield K, Weston C and Cook SJ (2003) Activation of the ERK1/2 signaling pathway promotes phosphorylation and proteasome-dependent degradation of the BH3-only protein, Bim. J. Biol. Chem. 278: 18811–18816
Luciano F, Jacquel A, Colosetti P, Herrant M, Cagnol S, Pages G and Auberger P (2003) Phosphorylation of Bim-EL by Erk1/2 on serine 69 promotes its degradation via the proteasome pathway and regulates its proapoptotic function. Oncogene 22: 6785–6793
Reginato MJ, Mills KR, Paulus JK, Lynch DK, Sgroi DC, Debnath J, Muthuswamy SK and Brugge JS (2003) Integrins and EGFR coordinately regulate the pro-apoptotic protein Bim to prevent anoikis. Nat. Cell Biol. 5: 733–740
Bacus SS, Gudkov AV, Lowe M, Lyass L, Yung Y, Komarov AP, Keyomarsi K, Yarden Y and Seger R (2001) Taxol-induced apoptosis depends on MAP kinase pathways (ERK and p38) and is independent of p53. Oncogene 20: 147–155
Lee LF, Li G, Templeton DJ and Ting JP (1998) Paclitaxel (Taxol)-induced gene expression and cell death are both mediated by the activation of c-Jun NH2-terminal kinase (JNK/SAPK). J. Biol. Chem. 273: 28253–28260
Okano J and Rustgi AK (2001) Paclitaxel induces prolonged activation of the Ras/MEK/ERK pathway independently of activating the programmed cell death machinery. J. Biol. Chem. 276: 19555–19564
Deacon K, Mistry P, Chernoff J, Blank JL and Patel R (2003) p38 mitogen-activated protein kinase mediates cell death and p21-activated kinase mediates cell survival during chemotherapeutic drug-induced mitotic arrest. Mol. Cell. Biol. 14: 2071–2087
Johnson GL and Lapadat R (2002) Mitogen-activated protein kinase pathways mediated by ERK, JNK, and p38 protein kinases. Science 298: 1911–1912
Li R, Ruttinger D, Urba WJ, Fox BA and Hu H-M (2004) Targeting and amplification of immune killing of tumor cells by pro-Smac. Int. J. Cancer 109: 85–94
Yang D, Buchholz F, Huang Z, Goga A, Chen CY, Brodsky FM and Bishop JM (2002) Short RNA duplexes produced by hydrolysis with Escherichia coli RNase III mediate effective RNA interference in mammalian cells. Proc. Natl. Acad. Sci. USA 99: 9942–9947
Acknowledgements
We thank Dr. Stanley J Korsmeyer for providing the retroviral vectors, Dr. Walter J Urba for the critical reading of this manuscript, Dr. Bernard A Fox for his support, and Mr. Dan Haley for the cell sorting. This study was supported by grants from the Chiles Foundation, the Providence Portland Medical Foundation, the MJ Murdock Charitable Trust, and Cell Genesis Corporation.
Author information
Authors and Affiliations
Corresponding author
Additional information
Edited by G Melino
Rights and permissions
About this article
Cite this article
Li, R., Moudgil, T., Ross, H. et al. Apoptosis of non-small-cell lung cancer cell lines after paclitaxel treatment involves the BH3-only proapoptotic protein Bim. Cell Death Differ 12, 292–303 (2005). https://doi.org/10.1038/sj.cdd.4401554
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/sj.cdd.4401554
Keywords
This article is cited by
-
Acetylation of FOXO1 activates Bim expression involved in CVB3 induced cardiomyocyte apoptosis
Apoptosis (2023)
-
Utidelone inhibits growth of colorectal cancer cells through ROS/JNK signaling pathway
Cell Death & Disease (2021)
-
PRKCQ inhibition enhances chemosensitivity of triple-negative breast cancer by regulating Bim
Breast Cancer Research (2020)
-
Penfluridol overcomes paclitaxel resistance in metastatic breast cancer
Scientific Reports (2019)
-
Resistance to anti-microtubule drug-induced cell death is determined by regulation of BimEL expression
Oncogene (2019)