Abstract
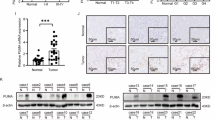
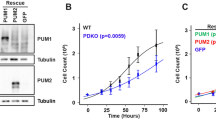
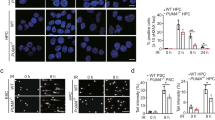
PUMA, a key mediator of p53-induced apoptosis, is a BH3-only domain proapoptotic protein that localizes to mitochondria and interacts with antiapoptotic Bcl-2 and Bcl-XL. Recent evidence implicates Bax to be an important mediator of PUMA-activated apoptotic signals. We have previously demonstrated that Bax deficiency significantly affects thapsigargin (TG)-mediated endoplasmic reticulum calcium pool depletion-induced apoptosis. We now present evidence that TG upregulates PUMA expression and that although Bax-deficient cells exhibit resistance to TG, Bax deficiency does not attenuate TG upregulation of PUMA expression. Furthermore, TG transcriptionally upregulates PUMA expression in a p53-independent manner and that PUMA-deficient cells are more resistant to undergo TG-induced apoptosis than the PUMA-proficient counterparts. Thus, our results demonstrate that TG engages PUMA and Bax for full transduction of apoptotic signals and both PUMA and Bax appear to exist in the same TG-activated apoptotic pathway in which PUMA may reside upstream of Bax.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Abbreviations
- Ca2+:
-
calcium
- CaM:
-
calmodulin
- CREB:
-
cAMP response element-binding protein
- bbc3:
-
Bcl-2 binding component 3
- DMEM:
-
Dulbecco's modified Eagle's medium
- DKO:
-
double knockout
- DR5:
-
death receptor 5
- ER:
-
endoplasmic reticulum
- ERSEs:
-
ER stress response elements
- ETOP:
-
etoposide
- InsP3R:
-
inositol (1,4,5) triphosphate receptor
- PBS:
-
phospate-buffered saline
- grp:
-
glucose-regulated protein
- PUMA:
-
p53 upregulated modulator of apoptosis
- TG:
-
thapsigargin
References
Nakano K and Vousden KH (2001) PUMA, a novel proapoptotic gene, is induced by p53. Mol. Cell 7: 683–694
Yu J, Zhang L, Hwang PM, Kinzler KW and Vogelstein B (2001) PUMA induces the rapid apoptosis of colorectal cancer cells. Mol. Cell 7: 673–682
Han J, Flemington C, Houghton AB, Gu Z, Zambetti GP, Lutz RJ, Zhu L and Chittenden T (2001) Expression of bbc3, a pro-apoptotic BH3-only gene, is regulated by diverse cell death and survival signals. Proc. Natl. Acad. Sci. USA 98: 11318–11323
Reimertz C, Kogel D, Rami A, Chittenden T and Prehn JH (2003) Gene expression during ER stress-induced apoptosis in neurons: induction of the BH3-only protein Bbc3/PUMA and activation of the mitochondrial apoptosis pathway. J. Cell Biol. 162: 587–597
Futami T, Miyagishi M and Taira K (2005) Identification of a network involved in thapsigargin-induced apoptosis using a library of small interfering RNA expression vectors. J. Biol. Chem. 280: 826–831
Sheikh MS and Fornace Jr AJ (2000) Role of p53 family members in apoptosis. J. Cell Physiol. 182: 171–181
Vousden KH and Lu X (2002) Live or let die: the cell's response to p53. Nat. Rev. Cancer 2: 594–604
Hofseth LJ, Hussain SP and Harris CC (2004) p53: 25 years after its discovery. Trends Pharm. Sci. 25: 177–181
Jeffers JR, Parganas E, Lee Y, Yang C, Wang J, Brennan J, MacLean KH, Han J, Chittenden T, Ihle JN, McKinnon PJ, Cleveland Jl and Zambetti GP (2003) Puma is an essential mediator of p53-dependent and -independent apoptotic pathways. Cancer Cell 4: 321–328
Villunger A, Michalak EM, Coultas L, Mullauer F, Bock G, Ausserlechner MJ, Adams JM and Strasser A (2003) p53- and drug-induced apoptotic responses mediated by BH3-only proteins puma and noxa. Science 302: 1036–1038
Yu J, Wang Z, Kinzler KW, Vogelstein B and Zhang L (2003) PUMA mediates the apoptotic response to p53 in colorectal cancer cells. Proc. Natl. Acad. Sci. USA 100: 1931–1936
Kim BC, Kim HT, Mamura M, Ambudkar IS, Choi KS and Kim SJ (2002) Tumor necrosis factor induces apoptosis in hepatoma cells by increasing Ca2+ release from the endoplasmic reticulum and suppressing Bcl-2 expression. J. Biol. Chem. 277: 31381–31389
Nutt LK, Chandra J, Pataer A, Fang B, Roth JA, Swisher SG, O'Neil RG and McConkey DJ (2002) Bax-mediated Ca2+ mobilization promotes cytochrome c release during apoptosis. J. Biol. Chem. 277: 20301–20308
Sheikh MS and Huang Y (2004) TRAIL death receptors, Bcl-2 protein family, and endoplasmic reticulum calcium pool. Vitam. Horm. 67: 169–188
Foyouzi-Youssefi R, Arnaudeau S, Broner C, Kelley WL, Tschopp J, Lew DP, Demaurex N and Krause KH (2000) Bcl-2 decreases the free Ca2+ concentration within the endoplasmic reticulum. Proc. Natl. Acad. Sci. USA 97: 5723–5728
Pinton P, Ferrari D, Magalhaes P, Schulze-Osthoff K, DiVirgilio F, Pozzan T and Rizzuto R (2000) Reduced loading of intracellular Ca2+ stores and downregulation of capacitative Ca2+ influx in Bcl-2-overexpressing cells. J. Cell Biol. 148: 857–862
Demaurex N and Distelhorst C (2003) Cell biology. Apoptosis – the calcium connection. Science 300: 65–67
Nutt LK, Pataer A, Pahler J, Fang B, Roth JA, McConkey DJ and Swisher SG (2002) Bax and Bak promote apoptosis by modulating endoplasmic reticular and mitochondrial Ca2+ stores. J. Biol. Chem. 277: 9219–9225
Scorrano L, Oakes SA, Opferman JT, Cheng EH, Sorcinelli MD, Pozzan T and Korsmeyer SJ (2003) BAX and BAK regulation of endoplasmic reticulum Ca2+: a control point for apoptosis. Science 300: 135–139
Boehning D, Patterson RL, Sedaghat L, Glebova NO, Kurosaki T and Snyder SH (2003) Cytochrome c binds to inositol (1,4,5) trisphosphate receptors, amplifying calcium-dependent apoptosis. Nat. Cell Biol. 5: 1051–1061
Sagara Y and Inesi G (1991) Inhibition of the sarcoplasmic reticulum Ca2+ transport ATPase by thapsigargin at subnanomolar concentrations. J. Biol. Chem. 266: 13503–13506
Pinton P, Ferrari D, Rapizzi E, DiVirgilio FD, Pozzan T and Rizzuto R (2001) The Ca2+ concentration of the endoplasmic reticulum is a key determinant of ceramide-induced apoptosis: significance for the molecular mechanism of Bcl-2 action. EMBO J. 20: 2690–2701
Rapizzi E, Pinton P, Szabadkai G, Wieckowski MR, Vandecasteele G, Baird G, Tuft RA, Fogarty KE and Rizzuto R (2002) Recombinant expression of the voltage-dependent anion channel enhances the transfer of Ca2+ microdomains to mitochondria. J. Cell Biol. 159: 613–624
He Q, Lee DI, Rong R, Yu M, Luo X, Klein M, El-Deiry WS, Huang Y, Hussain A and Sheikh MS (2002) Endoplasmic reticulum calcium pool depletion-induced apoptosis is coupled with activation of the death receptor 5 pathway. Oncogene 21: 2623–2633
He Q, Montalbano J, Corcoran C, Jin W, Huang Y and Sheikh MS (2003) Effect of Bax deficiency on death receptor 5 and mitochondrial pathways during endoplasmic reticulum calcium pool depletion-induced apoptosis. Oncogene 22: 2674–2679
Melino G, Bernassola F, Ranalli M, Yee K, Zong WX, Corazzari M, Knight RA, Green DR, Thompson C and Vousden KH (2004) p73 Induces apoptosis via PUMA transactivation and Bax mitochondrial translocation. J. Biol. Chem. 279: 8076–8083
Zhang L, Yu J, Park BH, Kinzler KW and Vogelstein B (2000) Role of BAX in the apoptotic response to anticancer agents. Science 290: 989–992
Wang X (2001) The expanding role of mitochondria in apoptosis. Gene Dev. 15: 2922–2933
Wolter KG, Hsu YT, Smith CL, Nechushtan A, Xi XG and Youle RJ (1997) Movement of Bax from the cytosol to mitochondria during apoptosis. J. Cell Biol. 139: 1281–1292
Hogan PG, Chen L, Nardone J and Rao A (2003) Transcriptional regulation by calcium, calcineurin, and NFAT. Genes Dev. 17: 2205–2232
Hook SS and Means AR (2001) Ca2+/CaM-dependent kinases: from activation to function. Annu. Rev. Pharmocol. Toxicol. 41: 471–505
Roy B and Lee AS (1999) The mammalian endoplasmic reticulum stress response element consists of an evolutionarily conserved tripartite structure and interacts with a novel stress-inducible complex. Nucleic Acids Res. 27: 1437–1443
Kokame K, Kato H and Miyata T (2001) Identification of ERSE-II, a new cis-acting element responsible for the ATF6-dependent mammalian unfolded protein response. J. Biol. Chem. 276: 9199–9205
Johnstone RW, Ruefli AA and Lowe SW (2002) Apoptosis: a link between cancer genetics and chemotherapy. Cell 108: 153–164
Nicholson DW (2000) From bench to clinic with apoptosis-based therapeutic agents. Nature 407: 810–816
Reed JC (2002) Apoptosis-based therapies. Nat. Rev. Drug Dev. 1: 111–121
Huang Y, He Q, Rong R, Hillman MJ and Sheikh MS (2001) Sulindac sulfide-induced apoptosis involves death receptor 5 and the caspase 8-dependent pathway in human colon and prostate cancer cells. Cancer Res. 61: 6918–6924
Luo X, Huang Y and Sheikh MS (2003) Cloning and characterization of a novel gene PDRG that is differentially regulated by p53 and ultraviolet radiation. Oncogene 22: 7247–7257
He Q, Luo X, Huang Y and Sheikh MS (2004) Apo2L/TRAIL differentially modulates the apoptotic effects of sulindac and a COX-2 selective non-steroidal anti-inflammatory agent in Bax-deficient cells. Oncogene 21: 6032–6040
Acknowledgements
We thank Dr. Bert Vogelstein (Johns Hopkins University, Baltimore, MD, USA) for kindly providing the Bax and PUMA-proficient and -deficient HCT116 cells, and Dr. Allen (SUNY Upstate Medical University, Syracuse, NY, USA) and Dr. Oscar Colamonici (The University of Illinios at Chicago, Chicago, IL, USA) for Saos-2 cells. We are also thankful to Dr. Chittenden (ImmunoGen, Cambridge, MA, USA) for providing the human PUMA/bbc promoter-luciferase construct, and Dr. Richard J Youle (National Institutes of Health, Bethesda, MD, USA) for providing the pEGFP-Bax construct. This work was partly supported by the NIH Grants CA86945, DK062136, DK067271 and funds from the Sinsheimer Scholar Award by the Alxandrine and Alexander Sinsheimer Foundation.
Author information
Authors and Affiliations
Corresponding author
Additional information
Edited by V De Laurenzi
Rights and permissions
About this article
Cite this article
Luo, X., He, Q., Huang, Y. et al. Transcriptional upregulation of PUMA modulates endoplasmic reticulum calcium pool depletion-induced apoptosis via Bax activation. Cell Death Differ 12, 1310–1318 (2005). https://doi.org/10.1038/sj.cdd.4401659
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/sj.cdd.4401659
Keywords
This article is cited by
-
A metabolic associated fatty liver disease risk variant in MBOAT7 regulates toll like receptor induced outcomes
Nature Communications (2022)
-
Puma, noxa, p53, and p63 differentially mediate stress pathway induced apoptosis
Cell Death & Disease (2021)
-
Non-apoptotic functions of BCL-2 family proteins
Cell Death & Differentiation (2017)
-
Overexpression of a dominant-negative mutant of SIRT1 in mouse heart causes cardiomyocyte apoptosis and early-onset heart failure
Science China Life Sciences (2014)
-
Bcl-2 family in inter-organelle modulation of calcium signaling; roles in bioenergetics and cell survival
Journal of Bioenergetics and Biomembranes (2014)