ABSTRACT
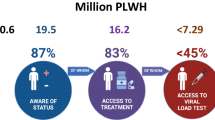
In China, the estimated number of HIV infected cases is approaching one million. Although public education has been initiated for awareness and behavioral modification for this devastating infection, better diagnostic methods are needed to identify infected persons and manage infection. Simple and more accurate diagnostic tools have become available, particularly for early detection and to monitor treatment in those who receive anti-retroviral treatment. In this short review, we summarize some of the common and new methodologies that can be used in clinical laboratories, in the field, or in private laboratories. These range from simple antibody tests to more sophistical methods that are used to monitor disease progression and identify drug resistance. These tools can assist physicians, medical practitioners, and laboratory personnel to select suitable diagnostic tools for the diagnosis, blood screening, monitoring of disease progression, and for detection of drug resistance to anti-retroviral therapies.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Constantine NT, ed. Retroviral Testing and Quality Assurance: Essentials for Laboratory Diagnosis. Ann Arbor, MI: Malloy Publishers, 2005.
Branson BM . FDA approves OraQuick for use in saliva. On March 25, the FDA approved the first rapid test for HIV in oral fluids. AIDS Clin Care 2004; 16:39.
Update CHATPN. Uncommon HIV-1 Virus Detected in China. Kyodo News Services. 2000.
Bulterys M, Jamieson DJ, O'Sullivan MJ, et al. Rapid HIV-1 testing during labor: a multicenter study. JAMA 2004; 292:219–23.
Prevention CfDCa. CDC: better needlestick reporting required. AIDS Alert 1997; 30–1.
Constantine NT, Ketema F . Rapid confirmation of HIV infection. Int J Infect Dis 2002; 6:170–7.
Constantine NT, Sill AM, Jack N, et al. Improved classification of recent HIV-1 infection by employing a two-stage sensitive/less-sensitive test strategy. J AIDS 2003; 32:94–103.
Albrecht H, Hoffmann C, Degen O, et al. Highly active antiretroviral therapy significantly improves the prognosis of patients with HIV-associated progressive multifocal leukoencephalopathy. AIDS 1998; 12:1149–54.
de Mendoza C, Soriano V, Perez-Olmeda M, et al. Different outcomes in patients achieving complete or partial viral load suppression on antiretroviral therapy. J Hum Virol 1999; 2:344–9.
Guatelli JC, Whitfield KM, Kwoh DY, Barringer KJ, Richman DD, Gingeras TR . Isothermal, in vitro amplification of nucleic acids by a multienzyme reaction modeled after retroviral replication. Proc Natl Acad Sci U S A. 1990; 87:1874–8.
Urdea MS, Horn T, Fultz TJ, et al. Branched DNA amplification multimers for the sensitive, direct detection of human hepatitis viruses. Nucleic Acids Symp Ser 1991; 197–200.
Shingadia D, Zhao Y . Mesurement of plasma viral RNA load of human immunodeficiency virus type 1 (HIV-1). Am Med Lab Int 1997; 2:4–5.
Yeghiazarian T, Zhao Y, Read SE, et al. Quantification of human immunodeficiency virus type 1 RNA levels in plasma by using small-volume-format branched-DNA assays. J Clin Microbiol 1998; 36:2096–8.
Gibellini D, Vitone F, Gori E, La Placa M, Re MC . Quantitative detection of human immunodeficiency virus type 1 (HIV-1) viral load by SYBR green real-time RT-PCR technique in HIV-1 seropositive patients. J Virol Meth 2004; 115:182–9.
Schupbach J, Boni J, Flepp M, et al. Antiretroviral treatment monitoring with an improved HIV-1 p24 antigen test: an inexpensive alternative to tests for viral RNA. J Med Virol 2001; 65:225–32.
Barletta JM, Edelman DC, Constantine NT . Lowering the detection limits of HIV-1 viral load using real-time immuno-PCR for HIV-1 p24 antigen. Am J Clin Pathol 2004; 122:20–7.
Chun TW, Stuyver L, Mizell SB, et al. Presence of an inducible HIV-1 latent reservoir during highly active antiretroviral therapy. Proc Natl Acad Sci U S A 1997; 94:13193–7.
Finzi D, Blankson J, Siliciano JD, et al. Latent infection of CD4+ T cells provides a mechanism for lifelong persistence of HIV-1, even in patients on effective combination therapy. Nat Med 1999; 5:512–7.
Finzi D, Hermankova M, Pierson T, et al. Identification of a reservoir for HIV-1 in patients on highly active antiretroviral therapy. Science 1997; 278:1295–300.
Bennett JM, Kaye S, Berry N, Tedder RS . A quantitative PCR method for the assay of HIV-1 provirus load in peripheral blood mononuclear cells. J Virol Methods 1999; 83:11–20.
Christopherson C, Kidane Y, Conway B, Krowka J, Sheppard H, Kwok S . PCR-Based assay to quantify human immunodeficiency virus type 1 DNA in peripheral blood mononuclear cells. J Clin Microbiol 2000; 38:630–4.
Guenthner PC, Hart CE . Quantitative, competitive PCR assay for HIV-1 using a microplate-based detection system. Biotechniques 1998; 24:810–6.
Izopet J, Tamalet C, Pasquier C, et al. Quantification of HIV-1 proviral DNA by a standardized colorimetric PCR-based assay. J Med Virol 1998; 54:54–9.
Zhao Y, Yu M, Miller JW, et al. Quantification of human immunodeficiency virus type 1 proviral DNA by using TaqMan technology. J Clin Microbiol 2002; 40:675–8.
Holland P, Abramso R, Watson R, Gelfand D . Detection of specific polymerase chain reaction product by utilizating the 5′-3′ exonuclease activity of Thermus aquaticus DNA polymerase. Proc Natl Acad Sci U S A 1991; 88:7276–80.
Larder BA, Darby G, Richman DD . HIV with reduced sensitivity to zidovudine (AZT) isolated during prolonged therapy. Science 1989; 243:1731–4.
Larder BA, Kemp SD . Multiple mutations in HIV-1 reverse transcriptase confer high-level resistance to zidovudine (AZT). Science 1989; 246:1155–8.
DeMeter LM, D'Aquila R, Weislow O, et al. Interlaboratory concordance of DNA sequence analysis to detect reverse transcriptase mutations in HIV-1 proviral DNA. ACTG Sequencing Working Group. AIDS Clinical Trials Group. J Virol Methods 1998; 75:93–104.
Halvas E, Advovand G, Balfe I, et al. Blinded, multicenter comparison of the sensitivity of different technologies to detect and quantify a minor drug-resistant HIV-1 variant. The. Paper presented at: 10th Conference on Retrovirus and Opportunistic Infections, 2003; Boston.
Hirsch MS, Brun-Vezinet F, Clotet B, Richman DD . Antiretroviral Drug Resistance Testing in Adults Infected with Human Immunodeficiency Virus Type 1: 2003 Recommendations of an International AIDS Society-USA Panel. Clin Inf Dise 2003: 113–28.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
CONSTANTINE, N., KABAT, W. & ZHAO, R. Update on the laboratory diagnosis and monitoring of HIV infection. Cell Res 15, 870–876 (2005). https://doi.org/10.1038/sj.cr.7290361
Issue date:
DOI: https://doi.org/10.1038/sj.cr.7290361
Keywords
This article is cited by
-
CMOS image sensor based HIV diagnosis: a smart system for point-of-care approach
BioChip Journal (2013)