Abstract
Aim:
Several nicotinic acetylcholine receptor (nAChR) subunits have been engineered as fluorescent protein (FP) fusions and exploited to illuminate features of nAChRs. The aim of this work was to create a FP fusion in the nAChR α7 subunit without compromising formation of functional receptors.
Methods:
A gene construct was generated to introduce yellow fluorescent protein (YFP), in frame, into the otherwise unaltered, large, second cytoplamsic loop between the third and fourth transmembrane domains of the mouse nAChR α7 subunit (α7Y). SH-EP1 cells were transfected with mouse nAChR wild type α7 subunits (α7) or with α7Y subunits, alone or with the chaperone protein, hRIC-3. Receptor function was assessed using whole-cell current recording. Receptor expression was measured with 125I-labeled α-bungarotoxin (I-Bgt) binding, laser scanning confocal microscopy, and total internal reflectance fluorescence (TIRF) microscopy.
Results:
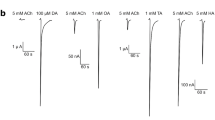
Whole-cell currents revealed that α7Y nAChRs and α7 nAChRs were functional with comparable EC50 values for the α7 nAChR-selective agonist, choline, and IC50 values for the α7 nAChR-selective antagonist, methyllycaconitine. I-Bgt binding was detected only after co-expression with hRIC-3. Confocal microscopy revealed that α7Y had primarily intracellular rather than surface expression. TIRF microscopy confirmed that little α7Y localized to the plasma membrane, typical of α7 nAChRs.
Conclusion:
nAChRs composed as homooligomers of α7Y subunits containing cytoplasmic loop YFP have functional, ligand binding, and trafficking characteristics similar to those of α7 nAChRs. α7Y nAChRs may be used to elucidate properties of α7 nAChRs and to identify and develop novel probes for these receptors, perhaps in high-throughput fashion.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Chalfie M, Tu Y, Euskirchen G, Ward WW, Prasher DC . Green fluorescent protein as a marker for gene expression. Science 1994; 263: 802–5.
Tsien RY . The green fluorescent protein. Ann Rev Bioch 1998; 67: 509–44.
Khakh BS, Fisher JA, Nashmi R, Bowser DN, Lester HA . An angstrom scale interaction between plasma membrane ATP-gated P2X2 and α4β2 nicotinic channels measured with fluorescence resonance energy transfer and total internal reflection fluorescence microscopy. J Neurosci 2005; 25: 6911–20.
Fucile S, Palma E, Martínez-Torres A, Miledi R, Eusebi F . The single-channel properties of human acetylcholine α7 receptors are altered by fusing α7 to the green fluorescent protein. Proc Natl Acad Sci USA 2002; 99: 3956–61.
Palma E, Mileo AM, Martínez-Torres A, Eusebi F, Miledi R . Some properties of human neuronal α7 nicotinic acetylcholine receptors fused to the green fluorescent protein. Proc Natl Acad Sci USA 2002; 99: 3950–5.
Prasher DC, Eckenrode VK, Ward WW, Prendergast FG, Cormier MJ . Primary structure of the Aequorea victoria green-fluorescent protein. Gene 1992; 111: 229–33.
Valor LM, Mulet J, Sala F, Sala S, Ballesta JJ, Criado M . Role of the large cytoplasmic loop of the alpha 7 neuronal nicotinic acetylcholine receptor subunit in receptor expression and function. Biochem 2002; 417931–8.
Toulmé E, Soto F, Garret M, Boué-Grabot E . Functional properties of internalization-deficient P2X4 receptors reveal a novel mechanism of ligand-gated channel facilitation by ivermectin. Mol Pharmacol 2005; 69: 576–87.
Nashmi R, Dickinson ME, McKinney S, Jareb M, Labarca C, Fraser SE, et al. Assembly of α4β2 nicotinic acetylcholine receptors assessed with functional fluorescently labeled subunits: effects of localization, trafficking, and nicotine-induced upregulation in clonal mammalian cells and in cultured midbrain neurons. J Neurosci 2003; 23: 11554–67.
Drenan M, Nashmi R, Imoukhuede P, Just H, McKinney S, Lester HA . Subcellular trafficking, pentameric assembly, and subunit stoichiometry of neuronal nicotinic acetylcholine receptors containing fluorescently labeled α6 and α3 subunits. Mol Pharmacol 2008; 73: 27–41.
Xu J, Zhu Y, Heinemann SF . Identification of sequence motifs that target neuronal nicotinic receptors to dendrites and axons. J Neurosci 2006; 26: 978–93.
Graihle R, de Carvalho LP, Pass Y, Le Poupon C, Soudant M, Bregestovski P, et al. Distinct subcellular targeting of fluorescent nicotinic α4β3 and serotoninergic 5-HT3A receptors in hippocampal neurons. Eur J Neurosci 2004; 19: 855–62.
Slimko EM, Lester HA . Codon optimization of Caenorhabditis elegans GluCl ion channel genes for mammalian cells dramatically improves expression levels. J Neurosci Methods 2003; 124: 75–81.
Ilegems E, Pick HM, Deluz C, Kellenberger S, Vogel H . Noninvasive imaging of 5-HT3 receptor trafficking in live cells: from biosynthesis to endocytosis. J Biol Chem 2004; 279: 53346–52.
Pantoja R, Rodriguez EA, Dibas MI, Dougherty DA, Lester HA . Single-molecule imaging of a fluorescent unnatural amino acid incorporated into nicotinic receptors. Biophysical J 2009; 96: 226–37.
Harkness PC, Millar NS . Changes in conformation and subcellular distribution of α4β2 nicotinic acetylcholine receptors revealed by chronic nicotine treatment and expression of subunit chimeras. J Neurosci 2002; 22: 10172–81.
Zhang J, Berg DK . Reversible inhibition of GABAA receptors by α7-containing nicotinic receptors on the vertebrate postsynaptic neurons. J Physiol 2007; 579: 753–63.
Dajas-Bailador FA, Soliakov L, Wonnacott S . Nicotine activates the extracellular signal-regulated kinase 1/2 via the alpha7 nicotinic acetylcholine receptor and protein kinase A, in SH-SY5Y cells and hippocampal neurones. J Neurochem 2002; 80: 520–30.
Song C, Murray TA, Kimura R, Wakui M, Ellsworth K, Javedan SP, et al. Role of alpha7-nicotinic acetylcholine receptors in tetanic stimulation-induced gamma oscillations in rat hippocampal slices. Neuropharm 2005; 48: 869–80.
Picciotto MR, Caldarone BJ, King SL, Zachariou V . Nicotinic receptors in the brain: links between molecular biology and behavior. Neuropsychopharmacology 2000; 22: 451–65.
Lukas RJ, Bencherif M . Recent developments in nicotinic acetylcholine receptor biology. In: Biological and biophysical aspects of ligand-gated ion channel receptor superfamilies. Arias H, editor. Kerala, India: Research Signpost; 2006. p27–59.
Ross RA, Spengler BA, Biedler JL . Coordinate morphological and biochemical interconversion of human neuroblastoma cells. J Natl Cancer Inst 1983; 71: 741–7.
Walton JD, Kattan DR, Thomas SK, Spengler BA, Guo H, Biedler JL, et al. Characteristics of stem cells from human neuroblastoma cell lines and in tumors. Neoplasia 2004; 6: 838–45.
Peng JH, Lucero L, Fryer J, Herl J, Leonard SS, Lukas RJ . Inducible, heterologous expression of human alpha7-nicotinic acetylcholine receptors in a native nicotinic receptor-null human clonal line. Brain Res 1999; 825:172–9.
Halevi S, Yassin L, Eshel M, Sala F, Sala S, Criado M, et al. Conservation within the RIC-3 gene family: effectors of mammalian nicotinic acetylcholine receptor expression. J Biol Chem 2003; 278: 34411–7.
Trichas G, Begbie J, Srinivas S . Use of the viral 2A peptide for bicistronic expression in transgenic mice. BMC Biology 2008; 6: 40.
Wu J, Kuo YP, George AA, Xu L, Hu J, Lukas RJ . beta-Amyloid directly inhibits human alpha4beta2-nicotinic acetylcholine receptors heterologously expressed in human SH-EP1 cells. J Biol Chem 2004; 279: 37842–51.
Fryer JD, Lukas RJ . Noncompetitive functional inhibition at diverse, human nicotinic acetylcholine receptor subtypes by bupropion, phencyclidine, and ibogaine. J Pharmacol Exp Ther 1999; 288: 88–92.
Castillo M, Mulet J, Gutierrez LM, Ortiz JA, Castelan F, Gerber S, et al. Dual role of the RIC-3 protein in trafficking serotonin and nicotinic receptors. J Biol Chem 2005; 280: 27062–8.
Blom N, Gammeltoft S, Brunak S . Sequence- and structure-based prediction of eukaryotic protein phosphorylation sites. J Mol Biol 1999; 294:1351–62.
Quick MW & Lester RA . Desensitization of neuronal nicotinic receptors. J Neurobiol 2002; 53: 457–78.
Alberts B, Johnson A, Lewis J, Raff M, Roberts K, Walter P . Molecular Biology of the Cell. 4th Ed. New York: Garland Science. 2002.
Miwa JM, Stevens TR, King SL, Caldarone BJ, Ibanez-Tallon I, Xiao C, et al. The prototoxin lynx1 acts on nicotinic acetylcholine receptors to balance neuronal activity and survival in vivo. Neuron 2006; 5: 587–600.
Tuerk C, Gold L . Systematic evolution of ligands by exponential enrichment: RNA ligands to bacteriophage T4 DNA polymerase. Science 1990; 3: 505–10.
McGuire MJ, Samli KN, Chang YC, Brown KC . Novel ligands for cancer diagnosis: Selection of peptide ligands for identification and isolation of B-cell lymphomas. Exper Hematol 2006; 34: 443–52.
Wilson DS, Keefe AD, Szostak JW . The use of mRNA display to select high-affinity protein-binding peptides. Proc Natl Acad Sci USA 2001; 98: 3750–5.
Ni F . Bivalent Bridged Polypeptides for Tumour-targeting, Imaging and Controllable Therapeutic Intervention [homepage on the Internet]. Biotechnology Research Institute, Canada. [Updated 01-25-2007; cited 2-1-2009] Available at: http://www.irb-bri.cnrc-nrc.gc.ca/rd/health/nmr/2007activities_e.html#networks.
Moser N, Mechawar N, Jones I, Gochberg-Sarver A, Orr-Urtreger A, Plomann M, et al. Evaluating the suitability of nicotinic acetylcholine receptor antibodies for standard immunodetection procedures. J Neurochem 2007; 102: 479–92.
Eisenstein M . 'Chain gang' delivers for hard labor. Nat Meth 2006; 3: 753–62.
George S, O'Dowd BF, Lee SP . G-protein-coupled receptor oligomerization and its potential for drug discovery. Nat Rev Drug Discov 2002; 1:808–20.
Gensler S, Sander A, Korngreen A, Traina G, Giese G, Witzemann V . Assembly and clustering of acetylcholine receptors containing GFP-tagged ɛ or γ subunits: Selective targeting to the neuromuscular junction in vivo. Eur J Biochem 2001; 268: 2209–17.
Williams ME, Burton B, Urrutia A, Shcherbatko A, Chavez-Noriega LE, Cohen CJ, et al. Ric-3 promotes functional expression of the nicotinic acetylcholine receptor subunit α7 in mammalian cells. J Biol Chem 2005; 280: 1257–63.
Whiteaker P, Sharples CG, Wonnacott S . Agonist-induced up-regulation of alpha4beta2 nicotinic acetylcholine receptors in M10 cells: pharmacological and spatial definition. Mol Pharmacol 1998; 53: 950–62.
North AJ . Seeing is believing? A beginners' guide to practical pitfalls in image acquisition. J Cell Biol 2006; 172: 9–18.
George N . TIRF microscopy: the evanescent wave of the future. Amer Lab 2004; 8: 26–68.
Herber DL, Severance EG, Cuevas J, Morgan D, Gordon MN . Biochemical and histochemical evidence of nonspecific binding of alpha7 nAChR antibodies to mouse brain tissue. J Histochem Cytochem 2004; 52:1367–76.
Dahan M, Lévi S, Luccardini C, Rostaing P, Riveau B, Triller A . Diffusion dynamics of glycine receptors revealed by single-quantum dot tracking. Science 2003; 302: 442–5.
Pons S, Fattore L, Cossu G, Tolu S, Porcu E, McIntosh JM, et al. Crucial role of alpha4 and alpha6 nicotinic acetylcholine receptor subunits from ventral tegmental area in systemic nicotine self-administration. J Neurosci 2008; 28:12318–27.
Azam L, Winzer-Serhan U, Leslie FM . Co-expression of α7 and β2 nicotinic acetylcholine receptor subunit mRNAS within rat brain cholinergic neurons. Neurosci 2003; 119: 965–77.
Khiroug SS, Harkness PC, Lamb PW, Sudweeks SN, Khiroug L, Millar NS, et al. Rat nicotinic Ach receptor α7 and β2 subunits co-assemble to form functional heteromeric nicotinic receptor channels. J Physiol 2002; 540.2: 425–34.
Peng JH, Fryer JD, Hurst RS, Schroeder KM, George AA, Morrissy S, et al. High-affinity epibatidine binding of functional, human α7-nicotinic acetylcholine receptors stably and heterologously expressed de novo in human SH-EP1 cells. J Pharm Exp Ther 2005; 313: 24–35.
Laummonerie C, Mutterer J . Colocalization Finder Version 1.2 plug-in for Image J image analysis software. Available online http://rsb.info.nih.gov/ij/plugins/colocalization-finder.html. 2006.
Acknowledgements
We wish to thank Dr Henry LESTER, California Institute of Technology, for the generous use of his facilities for performing confocal and TIRF studies. We also thank Dr Rigo PANTOJA and Dr Rahul SRINIVASAN for instruction and assistance using the TIRF and confocal microscope systems in Dr Lester's lab. We also gratefully acknowledge Dr Sean MEGASON, California Institute of Technology for the kind gift of the pCS2-mCherry construct; Dr Henry LESTER and Dr Raad NASHMI (formerly of the Lester lab and now at the University of Vancouver) for their gift of the α4-HA-YFP nAChR construct; and Dr Millet TREININ of Hebrew University for the gift of the hRIC-3 construct through Dr William GREEN of the University of Chicago.
This work was supported by a National Institutes of Health Grant DA015389, the Barrow Neurological Foundation, and the Biodesign Institute at Arizona State University. TA MURRAY was supported by a National Science Foundation Graduate Research Fellowship and a Catholic Healthcare West SEED grant.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Murray, T., Liu, Q., Whiteaker, P. et al. Nicotinic acetylcholine receptor α7 subunits with a C2 cytoplasmic loop yellow fluorescent protein insertion form functional receptors. Acta Pharmacol Sin 30, 828–841 (2009). https://doi.org/10.1038/aps.2009.78
Received:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/aps.2009.78