Abstract
Aim:
The substrate cocktail is frequently used to evaluate cytochrome P450 (CYP) enzyme-mediated drug interactions and potential interactions among the probe substrates. Here, we re-optimized the substrate cocktail method to increase the reliability and accuracy of screening for candidate compounds and expanded the method from a direct CYP inhibition assay to a time-dependent inhibition (TDI) assay.
Methods:
In the reaction mixtures containing human liver microsome (0.1 mg/mL), both the concentrations of a substrate cocktail (phenacetin for 1A2, coumarin for 2A6, bupropion for 2B6, diclofenac for 2C9, dextromethorphan for 2D6, and testosterone for 3A4) and the incubation time were optimized. Metabolites of the substrate probes were simultaneously analyzed by multiple-reaction monitoring (MRM) using a routine LC/MS/MS. Direct CYP inhibition was validated using 7 inhibitors (α-naphthoflavone, tranylcypromine, ticlopidine, fluconazole, quinidine, ketoconazole and 1-ABT). The time-dependent inhibition was partially validated with 5 inhibitors (ketoconazole, verapamil, quinidine, paroxetine and 1-ABT).
Results:
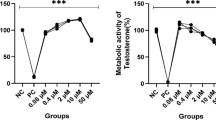
The inhibition curve profiles and IC50 values of 7 CYP inhibitors were approximate when a single substrate and the substrate cocktail were tested, and were consistent with the previously reported values. Similar results were obtained in the IC50 shifts of 5 inhibitors when a single substrate and the substrate cocktail were tested in the TDI assay.
Conclusion:
The 6-in-1 substrate cocktail (for 1A2, 2A6, 2B6, 2C9, 2D6 and 3A) is reliable for assessing CYP inhibition and time-dependent inhibition of drug candidates.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Ideura T, Muramatsu T, Higuchi M, Tachibana N, Hora K, Kiyosawa K . Tacrolimus/itraconazole interactions: a case report of ABO-incompatible living-related renal transplantation. Nephrol Dial Transplant 2000; 15: 1721–3
Chonlahan J, Halloran MA, Hammonds A . Leflunomide and warfarin interaction: case report and review of the literature. Pharmacotherapy 2006; 26: 868–71
Honig PK, Wortham DC, Zamani K, Conner DP, Mullin JC, Cantilena LR . Terfenadine-ketoconazole interaction. Pharmacokinetic and electrocardiographic consequences. JAMA 1993; 269: 1513–8
Krayenbuhl JC, Vozeh S, Kondo-Oestreicher M, Dayer P . Drug-drug interactions of new active substances: mibefradil example. Eur J Clin Pharmacol 1999; 55: 559–65
Wysowski DK, Bacsanyi J . Cisapride and fatal arrhythmia. N Engl J Med 1996; 335: 290–1
Williams JA, Hyland R, Jones BC, Smith DA, Hurst S, Goosen TC, et al. Drug-drug interactions for UDP-glucuronosyltransferase substrates: a pharmacokinetic explanation for typically observed low exposure (AUCi/AUC) ratios. Drug Metab Dispos 2004; 32: 1201–8
Food U, Administration D . Guidance for industry drug interaction studies—study design, data analysis, implications for dosing, and labeling recommendations. Food Drug Administration: Rockville, MD 2012.
Yamazaki H, Inui Y, Yun CH, Guengerich FP, Shimada T . Cytochrome P450 2E1 and 2A6 enzymes as major catalysts for metabolic activation of N-nitrosodialkylamines and tobacco-related nitrosamines in human liver microsomes. Carcinogenesis 1992; 13: 1789–94.
Shimada T, Yamazaki H, Guengerich FP . Ethnic-related differences in coumarin 7-hydroxylation activities catalyzed by cytochrome P4502A6 in liver microsomes of Japanese and Caucasian populations. Xenobiotica 1996; 26: 395–403.
Ueng YF, Chen CC, Chung YT, Liu TY, Chang YP, Lo WS, et al. Mechanism-based inhibition of cytochrome P450 (CYP)2A6 by chalepensin in recombinant systems, in human liver microsomes and in mice in vivo. Br J Pharmacol 2011; 163: 1250–62.
Kozakai K, Yamada Y, Oshikata M, Kawase T, Suzuki E, Haramaki Y, et al. Reliable high-throughput method for inhibition assay of 8 cytochrome P450 isoforms using cocktail of probe substrates and stable isotope-labeled internal standards. Drug Metab Pharmacokinet 2012; 27: 520–9.
Youdim KA, Lyons R, Payne L, Jones BC, Saunders K . An automated, high-throughput, 384 well cytochrome P450 cocktail IC50 assay using a rapid resolution LC-MS/MS end-point. J Pharm Biomed Anal 2008; 48: 92–9.
Tolonen A, Petsalo A, Turpeinen M, Uusitalo J, Pelkonen O . In vitro interaction cocktail assay for nine major cytochrome P450 enzymes with 13 probe reactions and a single LC/MSMS run: analytical validation and testing with monoclonal anti-CYP antibodies. J Mass Spectrom 2007; 42: 960–6.
Otten JN, Hingorani GP, Hartley DP, Kragerud SD, Franklin RB . An in vitro, high throughput, seven CYP cocktail inhibition assay for the evaluation of new chemical entities using LC-MS/MS. Drug Metab Lett 2011; 5: 17–24.
FDA. Guidance for Industry-Drug Interaction Studies-Study Design. Data Analysis, and Implications for Dosing and Labelling 2006.
Ortiz de Montellano PR, Mathews JM . Autocatalytic alkylation of the cytochrome P-450 prosthetic haem group by 1-aminobenzotriazole. Isolation of an NN-bridged benzyne-protoporphyrin IX adduct. Biochem J 1981; 195: 761–4.
Linder CD, Renaud NA, Hutzler JM . Is 1-aminobenzotriazole an appropriate in vitro tool as a nonspecific cytochrome P450 inactivator? Drug Metab Dispos 2009; 37: 10–3.
Kozakai K, Yamada Y, Oshikata M, Kawase T, Suzuki E, Haramaki Y, et al. Cocktail-substrate approach-based high-throughput assay for evaluation of direct and time-dependent inhibition of multiple cytochrome P450 isoforms. Drug Metab Pharmacokinet 2014; 29: 198–207.
Emoto C, Murase S, Sawada Y, Iwasaki K . In vitro inhibitory effect of 1-aminobenzotriazole on drug oxidations in human liver microsomes: a comparison with SKF-525A. Drug Metab Pharmacokinet 2005; 20: 351–7.
Dierks EA, Stams KR, Lim HK, Cornelius G, Zhang H, Ball SE . A method for the simultaneous evaluation of the activities of seven major human drug-metabolizing cytochrome P450s using an in vitro cocktail of probe substrates and fast gradient liquid chromatography tandem mass spectrometry. Drug Metab Dispos 2001; 29: 23–9.
Bu HZ, Knuth K, Magis L, Teitelbaum P . High-throughput cytochrome P450 (CYP) inhibition screening via a cassette probe-dosing strategy. V. Validation of a direct injection/on-line guard cartridge extraction—tandem mass spectrometry method for CYP1A2 inhibition assessment. Eur J Pharm Sci 2001; 12: 447–52.
Niwa T, Shiraga T, Takagi A . Effect of antifungal drugs on cytochrome P450 (CYP) 2C9, CYP2C19, and CYP3A4 activities in human liver microsomes. Biol Pharm Bull 2005; 28: 1805–8.
Dinger J, Meyer MR, Maurer HH . Development of an in vitro cytochrome P450 cocktail inhibition assay for assessing the inhibition risk of drugs of abuse. Toxicol Lett 2014; 230: 28–35.
Grimm SW, Einolf HJ, Hall SD, He K, Lim HK, Ling KH, et al. The conduct of in vitro studies to address time-dependent inhibition of drug-metabolizing enzymes: a perspective of the pharmaceutical research and manufacturers of America. Drug Metab Dispos 2009; 37: 1355–70.
Galetin A, Clarke SE, Houston JB . Multisite kinetic analysis of interactions between prototypical CYP3A4 subgroup substrates: midazolam, testosterone, and nifedipine. Drug Metab Dispos 2003; 31: 1108–16.
Acknowledgements
The Industry-Academia-Research Foundation of Chancheng, Foshan (No 2013B1009), and the Foundation of Foshan Innovation Team (No 2014IT100031) provided the financial support for this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, Zh., Zhang, Sx., Long, N. et al. An improved substrate cocktail for assessing direct inhibition and time-dependent inhibition of multiple cytochrome P450s. Acta Pharmacol Sin 37, 708–718 (2016). https://doi.org/10.1038/aps.2016.10
Received:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/aps.2016.10
Keywords
This article is cited by
-
Development of MEMS directed evolution strategy for multiplied throughput and convergent evolution of cytochrome P450 enzymes
Science China Life Sciences (2022)
-
Evaluation of In Vitro Cytochrome P450 Inhibition and In Vitro Fate of Structurally Diverse N-Oxide Metabolites: Case Studies with Clozapine, Levofloxacin, Roflumilast, Voriconazole and Zopiclone
European Journal of Drug Metabolism and Pharmacokinetics (2017)