Abstract
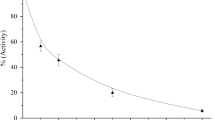
Anlotinib is a new oral tyrosine kinase inhibitor; this study was designed to characterize its pharmacokinetics and disposition. Anlotinib was evaluated in rats, tumor-bearing mice, and dogs and also assessed in vitro to characterize its pharmacokinetics and disposition and drug interaction potential. Samples were analyzed by liquid chromatography/mass spectrometry. Anlotinib, having good membrane permeability, was rapidly absorbed with oral bioavailability of 28%–58% in rats and 41%–77% in dogs. Terminal half-life of anlotinib in dogs (22.8±11.0 h) was longer than that in rats (5.1±1.6 h). This difference appeared to be mainly associated with an interspecies difference in total plasma clearance (rats, 5.35±1.31 L·h−1·kg−1; dogs, 0.40±0.06 L·h−1/kg−1). Cytochrome P450-mediated metabolism was probably the major elimination pathway. Human CYP3A had the greatest metabolic capability with other human P450s playing minor roles. Anlotinib exhibited large apparent volumes of distribution in rats (27.6±3.1 L/kg) and dogs (6.6±2.5 L/kg) and was highly bound in rat (97%), dog (96%), and human plasma (93%). In human plasma, anlotinib was predominantly bound to albumin and lipoproteins, rather than to α1-acid glycoprotein or γ-globulins. Concentrations of anlotinib in various tissue homogenates of rat and in those of tumor-bearing mouse were significantly higher than the associated plasma concentrations. Anlotinib exhibited limited in vitro potency to inhibit many human P450s, UDP-glucuronosyltransferases, and transporters, except for CYP3A4 and CYP2C9 (in vitro half maximum inhibitory concentrations, <1 μmol/L). Based on early reported human pharmacokinetics, drug interaction indices were 0.16 for CYP3A4 and 0.02 for CYP2C9, suggesting that anlotinib had a low propensity to precipitate drug interactions on these enzymes. Anlotinib exhibits many pharmacokinetic characteristics similar to other tyrosine kinase inhibitors, except for terminal half-life, interactions with drug metabolizing enzymes and transporters, and plasma protein binding.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Krause DS, Van Etten RA . Tyrosine kinases as targets for cancer therapy. N Engl J Med 2005; 353: 172–87.
Arora A, Scholar EM . Role of tyrosine kinase inhibitors in cancer therapy. J Pharmacol Exp Ther 2005; 315: 971–9.
Sun YK, Niu W, Du F, Du CX, Li ST, Wang JW, et al. Safety, pharmacokinetics, and antitumor properties of anlotinib, an oral multi-target tyrosine kinase inhibitor, in patients with advanced refractory solid tumors. J Hematol Oncol 2016; 9: 105.
Han BH . Efficiency and safety of anlotinib hydrochloride as 3rd line treatment or beyond in patients with advanced NSCLC in ALTER-003 study, a randomized, double-blind, placebo-controlled Phase III trial in China. The American Society of Clinical Oncology Annual Meeting, Chicago, IL, USA, June 2–6, 2017, Abstract 182187.
Guo B, Li C, Wang GJ, Chen LS . Rapid and direct measurement of free concentrations of highly protein-bound fluoxetine and its metabolite norfluoxetine in plasma. Rapid Commun Mass Spectrom 2006; 20: 39–47.
Li J, Brahnmer J, Messersmith W, Hidalgo M, Baker SD . Binding of gefitinib, an inhibitor of epidermal growth factor receptor-tyrosine kinase, to plasma proteins and blood cells: in vitro and in cancer patients. Invest New Drugs 2006; 24: 291–7.
Zsila F, Fitos I, Bencze G, Kéri G, Őrfi L . Determination of human serum α1-acid glycoprotein and albumin binding of various marketed and preclinical kinase inhibitors. Curr Med Chem 2009; 16: 1964–77.
Dai JY, Yang JL, Li C . Transport and metabolism of flavonoids from Chinese herbal remedy Xiaochaihu-tang across human intestinal Caco-2 cell monolayers. Acta Pharmacol Sin 2008; 29: 1086–93.
Li L, Zhao YS, Du FF, Yang JL, Xu F, Niu W, et al. Intestinal absorption and presystemic elimination of various chemical constituents present in GBE50 extract, a standardized extract of Ginkgo biloba leaves. Curr Drug Metab 2012; 13: 494–509.
Kerns EH, Di L . Drug-like properties: concepts, structure design and methods: from ADME to toxicity optimization. 1st ed. Burlington: Academic Press; 2008. p 283 (equilibrium shake flask thermodynamic solubility method), p 267–8 (shake flask method for lipophilicity).
Hu ZY, Yang JL, Cheng C, Huang YH, Du FF, Wang FQ, et al. Combinatorial metabolism notably affects human systemic exposure to ginsenosides from orally administered extract of Panx notoginseng roots (Sanqi). Drug Metab Dispos 2013; 41: 1457–69.
Jiang RR, Dong JJ, Li XX, Du FF, Jia WW, Xu F, et al. Molecular mechanisms governing different pharmacokinetics of ginsenosides and potential for ginsenoside-perpetrated herb-drug interactions on OATP1B3. Br J Pharmacol 2015; 172: 1059–73.
Jia WW, Du FF, Liu XW, Jiang RR, Xu F, Yang JL, et al. Renal tubular secretion of tanshinol: molecular mechanisms, impact on its systemic exposure, and propensity for dose-related nephrotoxicity and for renal herb-drug interactions. Drug Metab Dispos 2015; 43: 669–78.
Smith BP, Vandenhende FR, DeSante KA, Farid NA, Welch PA, Callaghan JT, et al. Confidence interval criteria for assessment of dose proportionality. Pharm Res 2000; 17: 1278–83.
Davies B, Morris T . Physiological parameters in laboratory animals and humans. Pharm Res 1993; 10: 1093–5.
Lipinski CA . Drug-like properties and the causes of poor solubility and poor permeability. J Pharmacol Toxicol Methods 2000; 44: 235–49.
van Leeuwen RWF, van Gelder T, Mathijssen RHJ, Ajansman FG . Drug-drug interactions with tyrosine-kinase inhibitors: a clinical perspective. Lancet Oncol 2014; 15: e315–26.
van Erp NP, Gelderblom H, Guchelaar HJ . Clinical pharmacokinetics of tyrosine kinase inhibitors. Cancer Treat Rev 2009; 35: 692–706.
Lee SY, Lim JW, Kim YM . Effect of alpha-1-acid glycoprotein expressed in cancer cells on malignant characteristics. Mol Cell 2001; 11: 341–5.
Widmer N, Decosterd LA, Csajka C, Leyvraz S, Duchosal MA, Rosselet A, et al. Population pharmacokinetics of imatinib and the role of α1-acid glycoprotein. Br J Clin Pharmacol 2006; 62: 97–112.
Apperley JF . Part I: mechanisms of resistance to imatinib in chronic myeloid leukaemia. Lancet Oncol 2007; 8: 1018–29.
Kaminsky LS, Zhang QY . The small intestine as a xenobiotic-metabolizing organ. Drug Metab Dispos 2003; 31: 1520–5.
Paine MF, Hart HL, Ludington SS, Haining RL, Rettie AE, Zeldin DC . The human intestinal cytochrome P450 “PIE”. Drug Metab Dispos 2006; 34: 880–6.
Thummel KE, Wilkinson GR . In vitro and in vivo drug interactions involving human CYP3A. Annu Rev Pharmacol Toxicol 1998; 38: 389–430.
Aoyama T, Yamano S, Waxman DJ, Lapenson DP, Meyer UA . Fischer Vpperley, et al . Cytochrome P-450 hPCN3, a novel cytochrome P-450 IIIA gene product that is differentially expressed in adult human liver. cDNA and deduced amino acid sequence and distinct specificities of cDNA-expressed hPCN1 and hPCN3 for the metabolism of steroid hormones and cyclosporine. J Biol Chem 1989; 264: 10388–95.
Shah RR, Morganroth J, Shah DR . Hepatotoxcity of tyrosine kinase inhibitors: clinical and regulatory perspectives. Drug Saf 2013; 36: 491–503.
Spraggs CF, Xu CF, Hunt CM . Genetic characterization to improve interpretation and clinical management of hepatotoxcity caused by tyrosine kinase inhibitors. Pharmacogenomics 2013; 14: 541–54.
Jakacki RI, Hamilton M, Gilbertson RJ, Blaney SM, Tesak J, Krailo MD, et al. Prediatric phase I and pharmacokinetic study of erlotinib followed by the combination of erlotinib and temozolomide: a children's oncology group phase I consortium study. J Clin Oncol 2008; 26: 4921–7.
Abou-Alfa GK, Amadori D, Santoro A, Figer A, De Greve J, Lathia C, et al. Safety and efficacy of sorafenib in patients with hepatocellular carcinoma (HCC) and Child-Pugh A versus B cirrhosis. Gastrointest Cancer Res 2011; 4: 40–4.
Bosma PJ, Goldhoorn B, Bakker C, Oude Elferink RP, Chowdhury JR, Chowdhury NR, et al. Bilirubin UDP-glucuonosyltransferase 1 is the only relevant bilirubin glucuronidating isoform in man. J Biol Chem 1994; 269: 17960–4.
Tukey RH, Strassburg CP . Human UDP-glucuonosyltransferases: metabolism, expression, and disease. Annu Rev Pharmacol Toxicol 2000; 40: 581–616.
Liu Y, Ramirez J, House L, Ratain M . Comparison of the drug-drug interactions potential of erlotinib and gefitinib via inhibition of UDP-glucuronosyltransferases. Drug Metab Dispos 2010; 38: 32–9.
Ai L, Zhu LL, Yang L, GE GB, Cao YF, Liu Y, et al. Selectivity for inhibition of nilotinib on the catalytic activity of human UDP-glucuronosyltransferases. Xenobiotica 2014; 44: 320–5.
Zhang N, Liu Y, Jeong H . Drug-drug interaction potentials of tyrosine kinase inhibitors via inhibition of UDP-glucuronosyltransferases. Sci Rep 2015; 5: 17778.
Miners JO, Chau N, Rowland A, Burns K, McKinnon RA, Mackenzie PI, et al. Inhibition of human UDP-glucuronosyltransferase enzymes by lapatinib, pazopanib, regorafenib and sorafenib: implications for hyperbilirubinemia. Biochem Pharmacol 2017; 129: 85–95.
Cheng XW, Lv X, Qu HY, Li DD, Hu MM, Guo WZ, et al. Comparison of the inhibition potentials of icotinib and erlotinib against human UDP-glucuronosyltransferase 1A1. Acta Pharm Sin B 2017; 7: 657–64.
Holohan C, Schaeybroeck SV, Longley DB, Johnston PG . Cancer drug resistance: an evolving paradigm. Nat Rev Cancer 2013; 13: 714–26.
Shukla S, Chen ZS, Ambudkar SV . Tyrosine kinase inhibitors as modulators of ABC transporter-mediated drug resistance. Drug Resist Update 2012; 15: 70–80.
Author information
Authors and Affiliations
Corresponding authors
Supplementary information
Supplementary Information
Supplementary Figure and Table (DOC 84 kb)
Rights and permissions
About this article
Cite this article
Zhong, Cc., Chen, F., Yang, Jl. et al. Pharmacokinetics and disposition of anlotinib, an oral tyrosine kinase inhibitor, in experimental animal species. Acta Pharmacol Sin 39, 1048–1063 (2018). https://doi.org/10.1038/aps.2017.199
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/aps.2017.199
Keywords
This article is cited by
-
Circulating metabolites of Borneolum syntheticum (Bingpian) ameliorate atherosclerosis in ApoE−/− mice via inhibiting macrophage foam-cell formation
Acta Pharmacologica Sinica (2025)
-
Anlotinib in cancer therapy: mechanisms of action, clinical applications, and future perspectives
Cancer Chemotherapy and Pharmacology (2025)
-
Off-label use of anlotinib in malignancies’ treatment: efficacy and management of adverse reactions
Pharmacological Reports (2025)
-
Cross-species comparison in nonclinical pharmacokinetics of lenvatinib by a simple HPLC with ultraviolet detection
Scientific Reports (2023)
-
Reactive oxygen species mediate anlotinib-induced apoptosis via activation of endoplasmic reticulum stress in pancreatic cancer
Cell Death & Disease (2020)