Abstract
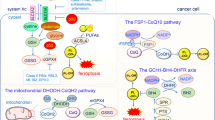
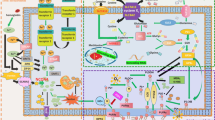
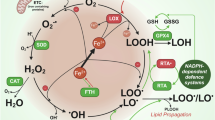
Ferroptosis is a recently recognized form of regulated cell death. It is characterized morphologically by the presence of smaller than normal mitochondria with condensed mitochondrial membrane densities, reduction or vanishing of mitochondria crista, and outer mitochondrial membrane rupture. It can be induced by experimental compounds (e.g., erastin, Ras-selective lethal small molecule 3, and buthionine sulfoximine) or clinical drugs (e.g., sulfasalazine, sorafenib, and artesunate) in cancer cells and certain normal cells (e.g., kidney tubule cells, neurons, fibroblasts, and T cells). Activation of mitochondrial voltage-dependent anion channels and mitogen-activated protein kinases, upregulation of endoplasmic reticulum stress, and inhibition of cystine/glutamate antiporter is involved in the induction of ferroptosis. This process is characterized by the accumulation of lipid peroxidation products and lethal reactive oxygen species (ROS) derived from iron metabolism and can be pharmacologically inhibited by iron chelators (e.g., deferoxamine and desferrioxamine mesylate) and lipid peroxidation inhibitors (e.g., ferrostatin, liproxstatin, and zileuton). Glutathione peroxidase 4, heat shock protein beta-1, and nuclear factor erythroid 2-related factor 2 function as negative regulators of ferroptosis by limiting ROS production and reducing cellular iron uptake, respectively. In contrast, NADPH oxidase and p53 (especially acetylation-defective mutant p53) act as positive regulators of ferroptosis by promotion of ROS production and inhibition of expression of SLC7A11 (a specific light-chain subunit of the cystine/glutamate antiporter), respectively. Misregulated ferroptosis has been implicated in multiple physiological and pathological processes, including cancer cell death, neurotoxicity, neurodegenerative diseases, acute renal failure, drug-induced hepatotoxicity, hepatic and heart ischemia/reperfusion injury, and T-cell immunity. In this review, we summarize the regulation mechanisms and signaling pathways of ferroptosis and discuss the role of ferroptosis in disease.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Abbreviations
- AA:
-
arachidonic acid
- ACC1:
-
acetyl-CoA carboxylase alpha
- ACSF2:
-
andacyl-CoA synthetase family member 2
- ACSL4:
-
acyl-CoA synthetase long-chain family member 4
- AOA:
-
aminooxyacetic acid
- AKF:
-
acute kidney failure
- ATP5G3:
-
ATP synthase F0 complex subunit C3
- BSO:
-
buthionine sulfoximine
- BHT:
-
butylated hydroxytoluene
- CARS:
-
cysteinyl-tRNA synthetase
- CHAC1:
-
cation transport regulator-like protein 1
- COX-2:
-
cyclooxygenase-2
- CS:
-
citrate synthase
- CSSG:
-
cysteine-glutathione disulfide
- DAMP:
-
damage-associated molecular pattern molecule
- DMT1:
-
divalent metal transporter 1
- ER:
-
endoplasmic reticulum
- Fe3+:
-
ferric iron
- Fe2+:
-
ferrous iron
- FIN:
-
ferroptosis-inducing agents
- FLT-3:
-
fms-like tyrosine kinase 3
- FTH1:
-
ferritin heavy chain 1
- FTL:
-
ferritin light chain
- GPX4:
-
glutathione peroxidase 4
- GSH:
-
glutathione
- HMGB1:
-
high mobility group box 1
- HD:
-
Huntington’s disease
- HETE:
-
hydroxyeicosatetraenoic acid
- HO-1:
-
heme oxygenase-1
- HPETE:
-
hydroperoxyeicosatetraenoic acid
- HSF-1:
-
heat shock factor-1
- HSPB1:
-
heat shock protein beta-1
- HCC:
-
hepatocellular carcinoma
- IREB2:
-
iron-responsive element-binding protein 2
- JNK:
-
c-Jun NH2-terminal kinase
- Keap1:
-
Kelch-like ECH-associated protein 1
- LOX:
-
lipoxygenases
- LPCAT3:
-
lysophosphatidylcholine acyltransferase 3
- MAPK:
-
mitogen-activated protein kinase
- MDM2:
-
murine double minute-2
- MEFs:
-
mouse embryonic fibroblasts
- NADPH:
-
nicotinamide adenine dinucleotide phosphate
- NAPQI:
-
N-acetyl-p-benzoquinone imine
- NOX:
-
NADPH oxidase
- NRF2:
-
nuclear factor erythroid 2-related factor 2
- OLs:
-
oligodendrocytes
- PDAC:
-
pancreatic ductal adenocarcinoma
- PGSK:
-
phen green SK
- PKC:
-
protein kinase C
- PPP:
-
pentose phosphate pathway
- PTGS:
-
prostaglandin-endoperoxide synthase
- PUFAs:
-
polyunsaturated fatty acids
- PVL:
-
periventricular leukomalacia
- RCD:
-
regulated cell death
- ROS:
-
reactive oxygen species
- RPL8:
-
ribosomal protein L8
- RSLs:
-
Ras-selective lethal small molecules
- SCP2:
-
sterol carrier protein 2
- TFR1:
-
transferrin receptor 1
- TTC35:
-
tetratricopeptide repeat domain 35
- VDAC:
-
mitochondrial voltage-dependent anion channel.
References
Dixon SJ, Lemberg KM, Lamprecht MR, Skouta R, Zaitsev EM, Gleason CE et al. Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell 2012; 149: 1060–1072.
Dolma S, Lessnick SL, Hahn WC, Stockwell BR . Identification of genotype-selective antitumor agents using synthetic lethal chemical screening in engineered human tumor cells. Cancer Cell 2003; 3: 285–296.
Yang WS, Stockwell BR . Synthetic lethal screening identifies compounds activating iron-dependent, nonapoptotic cell death in oncogenic-RAS-harboring cancer cells. Chem Biol 2008; 15: 234–245.
Yagoda N, von Rechenberg M, Zaganjor E, Bauer AJ, Yang WS, Fridman DJ et al. RAS-RAF-MEK-dependent oxidative cell death involving voltage-dependent anion channels. Nature 2007; 447: 864–868.
Friedmann Angeli JP, Schneider M, Proneth B, Tyurina YY, Tyurin VA, Hammond VJ et al. Inactivation of the ferroptosis regulator Gpx4 triggers acute renal failure in mice. Nat Cell Biol 2014; 16: 1180–1191.
Yang WS, SriRamaratnam R, Welsch ME, Shimada K, Skouta R, Viswanathan VS et al. Regulation of ferroptotic cancer cell death by GPX4. Cell 2014; 156: 317–331.
Dixon SJ, Patel DN, Welsch M, Skouta R, Lee ED, Hayano M et al. Pharmacological inhibition of cystine-glutamate exchange induces endoplasmic reticulum stress and ferroptosis. eLife 2014; 3: e02523.
Lorincz T, Jemnitz K, Kardon T, Mandl J, Szarka A . Ferroptosis is Involved in Acetaminophen Induced Cell Death. Pathol Oncol Res 2015; 21: 1115–1121.
Yang WS, Shimada K, Delva D, Patel M, Ode E, Skouta R et al. Identification of simple compounds with microtubule-binding activity that inhibit cancer cell growth with high potency. ACS Med Chem Lett 2012; 3: 35–38.
Weiwer M, Bittker JA, Lewis TA, Shimada K, Yang WS, MacPherson L et al. Development of small-molecule probes that selectively kill cells induced to express mutant RAS. Bioorg Med Chem Lett 2012; 22: 1822–1826.
Sakitama K, Ozawa Y, Aoto N, Tomita H, Ishikawa M . Effects of a new centrally acting muscle relaxant, NK433 (lanperisone hydrochloride) on spinal reflexes. Eur J Pharmacol 1997; 337: 175–187.
Shaw AT, Winslow MM, Magendantz M, Ouyang C, Dowdle J, Subramanian A et al. Selective killing of K-ras mutant cancer cells by small molecule inducers of oxidative stress. Proc Natl Acad Sci USA 2011; 108: 8773–8778.
Gout PW, Buckley AR, Simms CR, Bruchovsky N . Sulfasalazine, a potent suppressor of lymphoma growth by inhibition of the x(c)- cystine transporter: a new action for an old drug. Leukemia 2001; 15: 1633–1640.
Louandre C, Marcq I, Bouhlal H, Lachaier E, Godin C, Saidak Z et al. The retinoblastoma (Rb) protein regulates ferroptosis induced by sorafenib in human hepatocellular carcinoma cells. Cancer Lett 2015; 356: 971–977.
Louandre C, Ezzoukhry Z, Godin C, Barbare JC, Maziere JC, Chauffert B et al. Iron-dependent cell death of hepatocellular carcinoma cells exposed to sorafenib. Int J Cancer 2013; 133: 1732–1742.
Lachaier E, Louandre C, Godin C, Saidak Z, Baert M, Diouf M et al. Sorafenib induces ferroptosis in human cancer cell lines originating from different solid tumors. Anticancer Res 2014; 34: 6417–6422.
Sun X, Ou Z, Chen R, Niu X, Chen, Kang R et al. Activation of the p62-Keap1-NRF2 pathway protects against ferroptosis in hepatocellular carcinoma cells. Hepatology 2015 Sep 24. doi: 10.1002/hep.28251. [Epub ahead of print].
Nils Eling, Lukas Reuter, Hazin John, Hamacher-Brady Anne, Brady NR . Identification of artesunate as a specific activator of ferroptosis in pancreatic cancer cells. Oncoscience 2015; 2: 517–532.
Ooko E, Saeed ME, Kadioglu O, Sarvi S, Colak M, Elmasaoudi K et al. Artemisinin derivatives induce iron-dependent cell death (ferroptosis) in tumor cells. Phytomedicine 2015; 22: 1045–1054.
Dixon SJ, Stockwell BR . The role of iron and reactive oxygen species in cell death. Nat Chem Biol 2014; 10: 9–17.
Dixon SJ, Winter GE, Musavi LS, Lee ED, Snijder B, Rebsamen M et al. Human haploid cell genetics reveals roles for lipid metabolism genes in nonapoptotic cell death. ACS Chem Biol 2015; 10: 1604–1609.
Skouta R, Dixon SJ, Wang J, Dunn DE, Orman M, Shimada K et al. Ferrostatins inhibit oxidative lipid damage and cell death in diverse disease models. J Am Chem Soc 2014; 136: 4551–4556.
Yu Y, Xie Y, Cao L, Yang L, Yang M, Lotze MT et al. The ferroptosis inducer erastin enhances sensitivity of acute myeloid leukemia cells to chemotherapeutic agents. Mol Cell Oncol 2015 May 26. doi:10.1080/23723556.2015.1054549. [Epub ahead of print].
Bauer AJ, Gieschler S, Lemberg KM, McDermott AE, Stockwell BR . Functional model of metabolite gating by human voltage-dependent anion channel 2. Biochemistry 2011; 50: 3408–3410.
Maldonado EN, Sheldon KL, DeHart DN, Patnaik J, Manevich Y, Townsend DM et al. Voltage-dependent anion channels modulate mitochondrial metabolism in cancer cells: regulation by free tubulin and erastin. J Biol Chem 2013; 288: 11920–11929.
Matsushita M, Freigang S, Schneider C, Conrad M, Bornkamm GW, Kopf M . T cell lipid peroxidation induces ferroptosis and prevents immunity to infection. J Exp Med 2015; 212: 555–568.
Jiang L, Kon N, Li T, Wang SJ, Su T, Hibshoosh H et al. Ferroptosis as a p53-mediated activity during tumour suppression. Nature 2015; 520: 57–62.
Linkermann A, Skouta R, Himmerkus N, Mulay SR, Dewitz C, De Zen F et al. Synchronized renal tubular cell death involves ferroptosis. Proc Natl Acad Sci USA 2014; 111: 16836–16841.
Schott C, Graab U, Cuvelier N, Hahn H, Fulda S . Oncogenic RAS mutants confer resistance of RMS13 rhabdomyosarcoma cells to oxidative stress-induced ferroptotic cell death. Front Oncol 2015; 5: 131.
Gao M, Monian P, Quadri N, Ramasamy R, Jiang X . Glutaminolysis and transferrin regulate ferroptosis. Mol Cell 2015; 59: 298–308.
Li T, Kon N, Jiang L, Tan M, Ludwig T, Zhao Y et al. Tumor suppression in the absence of p53-mediated cell-cycle arrest, apoptosis, and senescence. Cell 2012; 149: 1269–1283.
Thomasova D, Bruns HA, Kretschmer V, Ebrahim M, Romoli S, Liapis H et al. Murinedouble minute-2 prevents p53-overactivation-related cell death (podoptosis) of podocytes. J Am Soc Nephrol 2014; 26: 1513–1523.
Hayano M, Yang WS, Corn CK, Pagano NC, Stockwell BR . Loss of cysteinyl-tRNA synthetase (CARS) induces the transsulfuration pathway and inhibits ferroptosis induced by cystine deprivation. Cell Death Differ 2015 Jul 17. doi: 10.1038/cdd.2015.93. [Epub ahead of print].
Chen L, Hambright WS, Na R, Ran Q . Ablation of ferroptosis inhibitor glutathione peroxidase 4 in neurons results in rapid motor neuron degeneration and paralysis. J Biol Chem 2015; 290: 28097–28106.
Canli Ö, Alankuş YB, Grootjans S, Vegi N, Hültner L, Hoppe PS et al. Glutathione peroxidase 4 prevents necroptosis in mouse erythroid precursors. Blood 2015 Oct 13. pii: blood-2015-06-654194. [Epub ahead of print].
Sun X, Ou Z, Xie M, Kang R, Fan Y, Niu X et al. HSPB1 as a novel regulator of ferroptotic cancer cell death. Oncogene 2015; 34: 5617–5625.
Kwon MY, Park E, Lee SJ, Chung SW . Heme oxygenase-1 accelerates erastin-induced ferroptotic cell death. Oncotarget 2015; 6: 24393–24403.
Wang XM, Terasaki PI, Rankin GW Jr, Chia D, Zhong HP, Hardy S . A new microcellular cytotoxicity test based on calcein AM release. Hum Immunol 1993; 37: 264–270.
Page B, Page M . Sensitive colorimetric cytotoxicity measurement using alarmar blue. Oncol Rep 1995; 2: 59–61.
Avelar-Freitas BA, Almeida VG, Pinto MC, Mourao FA, Massensini AR, Martins-Filho OA et al. Trypan blue exclusion assay by flow cytometry. Braz J Med Biol Res 2014; 47: 307–315.
Liu Y, Wang W, Li Y, Xiao Y, Cheng J, Jia J . The 5-lipoxygenase inhibitor zileuton confers neuroprotection against glutamate oxidative damage by inhibiting ferroptosis. Biol Pharm Bull 2015; 38: 1234–1239.
Chen L, Li X, Liu L, Yu B, Xue Y, Liu Y . Erastin sensitizes glioblastoma cells to temozolomide by restraining xCT and cystathionine-gamma-lyase function. Oncol Rep 2015; 33: 1465–1474.
Yamaguchi H, Hsu JL, Chen CT, Wang YN, Hsu MC, Chang SS et al. Caspase-independent cell death is involved in the negative effect of EGF receptor inhibitors on cisplatin in non-small cell lung cancer cells. Clin Cancer Res 2013; 19: 845–854.
Kang Y, Tiziani S, Park G, Kaul M, Paternostro G . Cellular protection using Flt3 and PI3Kalpha inhibitors demonstrates multiple mechanisms of oxidative glutamate toxicity. Nat Commun 2014; 5: 3672.
Kroemer G, Galluzzi L, Vandenabeele P, Abrams J, Alnemri ES, Baehrecke EH et al. Classification of cell death: recommendations of the Nomenclature Committee on Cell Death 2009. Cell Death Differ 2009 16: 3–11.
Galluzzi L, Vitale I, Abrams JM, Alnemri ES, Baehrecke EH, Blagosklonny MV et al. Molecular definitions of cell death subroutines: recommendations of the Nomenclature Committee on Cell Death 2012. Cell Death Differ 2012 19: 107–120.
Kroemer G, El-Deiry WS, Golstein P, Peter ME, Vaux D, Vandenabeele P et al. Classification of cell death: recommendations of the Nomenclature Committee on Cell Death. Cell Death Differ 2005; 12: 1463–1467.
Acknowledgements
We apologize to the researchers who were not referenced owing to space limitations. We thank Christine Heiner (Department of Surgery, University of Pittsburgh) for her critical reading of the manuscript. This work was supported by the National Institutes of Health of the USA (R01CA160417 and R01GM115366 to DT), The National Natural Science Foundation of China (31171229 and U1132005 to XS), and a Science and Information Technology of Guangzhou Key Project (201508020258 and 201400000003/4 to XS).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Edited by M Piacentini
Rights and permissions
About this article
Cite this article
Xie, Y., Hou, W., Song, X. et al. Ferroptosis: process and function. Cell Death Differ 23, 369–379 (2016). https://doi.org/10.1038/cdd.2015.158
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/cdd.2015.158
This article is cited by
-
TfR1 mediated iron metabolism dysfunction as a potential therapeutic target for osteoarthritis
Arthritis Research & Therapy (2024)
-
TrkA promotes MDM2-mediated AGPS ubiquitination and degradation to trigger prostate cancer progression
Journal of Experimental & Clinical Cancer Research (2024)
-
MiR-134-3p targets HMOX1 to inhibit ferroptosis in granulosa cells of sheep follicles
Journal of Ovarian Research (2024)
-
The microRNA-211-5p/P2RX7/ERK/GPX4 axis regulates epilepsy-associated neuronal ferroptosis and oxidative stress
Journal of Neuroinflammation (2024)
-
Exosomes derived from BMSCs enhance diabetic wound healing through circ-Snhg11 delivery
Diabetology & Metabolic Syndrome (2024)