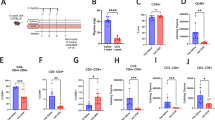
Abstract
The peripheral repertoire of CD4+ T lymphocytes contains autoreactive cells that remain tolerant through several mechanisms. However, nonspecific CD4+ T cells can be activated in physiological conditions as in the course of an ongoing immune response, and their outcome is not yet fully understood. Here, we investigate the fate of human naive CD4+ lymphocytes activated by dendritic cells (DCs) presenting endogenous self-peptides in comparison with lymphocytes involved in alloresponses. We generated memory cells (Tmem) from primary effectors activated with mature autologous DCs plus interleukin (IL)-2 (Tmauto), simulating the circumstances of an active immune response, or allogeneic DCs (Tmallo). Tmem were generated from effector cells that were rested in the absence of antigenic stimuli, with or without IL-7. Tmem were less activated than effectors (demonstrated by CD25 downregulation) particularly with IL-7, suggesting that this cytokine may favour the transition to quiescence. Tmauto and Tmallo showed an effector memory phenotype, and responded similarly to polyclonal and antigen-specific stimuli. Biochemically, IL-7-treated Tmallo were closely related to conventional memory lymphocytes based on Erk-1/2 activation, whereas Tmauto were more similar to effectors. Autologous effectors exhibited lower responses to IL-7 than allogeneic cells, which were reflected in their reduced proliferation and higher cell death. This was not related to IL-7 receptor expression but rather to signalling deficiencies, according to STAT5 activation These results suggest that ineffective responses to IL-7 could impair the transition to memory cells of naive CD4+ T lymphocytes recognizing self-peptides in the setting of strong costimulation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Kuchroo VK, Anderson AC, Waldner H, Munder M, Bettelli E, Nicholson LB . T cell response in experimental autoimmune encephalomyelitis (EAE): role of self and cross-reactive antigens in shaping, tuning, and regulating the autopathogenic T cell repertoire. Annu Rev Immunol 2002; 20: 101–123.
Tang AL, Bingaman AW, Kadavil EA, Leeser DB, Farber DL . Generation and functional capacity of polyclonal alloantigen-specific memory CD4 T cells. Am J Transplant 2006; 6: 1275–1284.
Boyman O, Krieg C, Homann D, Sprent J . Homeostatic maintenance of T cells and natural killer cells. Cell Mol Life Sci 2012; 69: 1597–1608.
Cai G, Hafler DA . Multispecific responses by T cells expanded by endogenous self-peptide/MHC complexes. Eur J Immunol 2007; 37: 602–612.
Weksler ME, Kozak R . Lymphocyte transformation induced by autologous cells. V. Generation of immunologic memory and specificity during the autologous mixed lymphocyte reaction. J Exp Med 1977; 146: 1833–1838.
Verhasselt V, Vosters O, Beuneu C, Nicaise C, Stordeur P, Goldman M . Induction of FOXP3-expressing regulatory CD4pos T cells by human mature autologous dendritic cells. Eur J Immunol 2004; 34: 762–772.
Zwickey HL, Unternaehrer JJ, Mellman I . Presentation of self-antigens on MHC class II molecules during dendritic cell maturation. Int Immunol 2006; 18: 199–209.
Choi JY, Craft J . Activation of naive CD4+ T cells in vivo by a self-peptide mimic: mechanism of tolerance maintenance and preservation of immunity. J Immunol 2004; 172: 7399–7407.
Marshall HD, Prince AL, Berg LJ, Welsh RM . IFN-alphabeta and self-MHC divert CD8 T cells into a distinct differentiation pathway characterized by rapid acquisition of effector functions. J Immunol 2010; 185: 1419–1428.
Scheinecker C, Machold KP, Majdic O, Höcker P, Knapp W, Smolen JS . Initiation of the autologous mixed lymphocyte reaction requires the expression of costimulatory molecules B7-1 and B7-2 on human peripheral blood dendritic cells. J Immunol 1998; 161: 3966–3973.
Chakir H, Lam DK, Lemay AM, Webb JR . ‘Bystander polarization’ of CD4+ T cells: activation with high-dose IL-2 renders naive T cells responsive to IL-12 and/or IL-18 in the absence of TCR ligation. Eur J Immunol 2003; 33: 1788–1798.
McKinstriy KK, Golech S, Lee WH, Huston G, Weng NP, Swain SL . Rapid default transition of CD4 T cell effectors to functional memory cells. J Exp Med 2007; 204: 2199–2211.
Harrington LE, Janowsk KM, Oliver JR, Zajac AJ, Weaver CT . Memory CD4 T cells emerge from effector T-cell progenitors. Nature 2008; 452: 356–360.
Kondrack RM, Harbertson J, Tan JT, McBreen ME, Surh CD, Bradley LM . Interleukin 7 regulates the survival and generation of memory CD4 cells. J Exp Med 2003; 198: 1797–1806.
Purton JF, Martin CE, Surh CD . Enhancing T cell memory: IL-7 as an adjuvant to boost memory T-cell generation. Immunol Cell Biol 2008; 86: 385–386.
Li J, Huston G, Swain SL . IL-7 promotes the transition of CD4 effectors to persistent memory cells. J Exp Med 2003; 198: 1807–1815.
Dooms H, Wolslegel K, Lin P, Abbas AK . Interleukin-2 enhances CD4+ T cell memory by promoting the generation of IL-7R alpha-expressing cells. J Exp Med 2007; 204: 547–557.
Harbertson J, Biederman E, Bennett KE, Kondrack RM, Bradley LM . Withdrawal of stimulation may initiate the transition of effector to memory CD4 cells. J Immunol 2002; 168: 1095–1102.
Hu H, Huston G, Duso D, Lepak N, Roman E, Swain SL . CD4+ T cell effectors can become memory cells with high efficiency and without further division. Nat Immunol 2001; 2: 705–710.
Brinkman CC, Sheasley-O'Neill SL, Ferguson AR, Engelhard VH . Activated CD8 T cells redistribute to antigen-free lymph nodes and exhibit effector and memory characteristics. J Immunol 2008; 181: 1814–1824.
Jelley-Gibbs DM, Brown DM, Dibble JP, Haynes L, Eaton SM, Swain SL . Unexpected prolonged presentation of influenza antigens promotes CD4 T cell memory generation. J Exp Med 2005; 202: 697–706.
Blair DA, Lefrançois L . Increased competition for antigen during priming negatively impacts the generation of memory CD4 T cells. Proc Natl Acad Sci USA 2007; 104: 15045–15050.
Williams MA, Ravkov EV, Bevan MJ . Rapid culling of the CD4+ T cell repertoire in the transition from effector to memory. Immunity 2008; 28: 533–545.
Sallusto F, Geginat J, Lanzavecchia A . Central memory and effector memory T cell subsets: function, generation, and maintenance. Annu Rev Immunol 2004; 22: 745–763.
Torres-Aguilar H, Aguilar-Ruiz SR, González-Pérez G, Munguía R, Bajaña S, Meraz-Ríos MA et al. Tolerogenic dendritic cells generated with different immunosuppressive cytokines induce antigen-specific anergy and regulatory properties in memory CD4+ T cells. J. Immunol 2010; 184: 1765–1775.
Geginat J, Sallusto F, Lanzavecchia A . Cytokine-driven proliferation and differentiation of human naive, central memory, and effector memory CD4+ T cells. J Exp Med 2001; 194: 1711–1719.
Ahmadzadeh M, Hussain SF, Farber DL . Effector CD4 T cells are biochemically distinct from the memory subset: evidence for long-term persistence of effectors in vivo. J Immunol 1999; 163: 3053–3063.
Chandok MR, Okoye FI, Ndejembi MP, Farber DL . A biochemical signature for rapid recall of memory CD4 T cells. J Immunol 2007; 179: 3689–3698.
Farber DL . Differential TCR signaling and the generation of memory T cells. J Immunol 1998; 160: 535–539.
Farber DL, Acuto O, Bottomly K . Differential T cell receptor-mediated signaling in naive and memory CD4 T cells. Eur J Immunol 1997; 27: 2094–2101.
Hussain SF, Anderson CF, Farber DL . Differential SLP-76 expression and TCR-mediated signaling in effector and memory CD4 T cells. J Immunol 2002; 168: 1557–1565.
Watson ARO, Lee WT . Differences in signaling molecule organization between naive and memory CD4+ T lymphocytes. J Immunol 2004; 173: 33–41.
Krishnan S, Warke VG, Nambiar MP, Wong HK, Tsokos GC, Farber DL . Generation and biochemical analysis of human effector CD4 T cells: alterations in tyrosine phosphorylation and loos of CD3ζ expression. Blood 2001; 97: 3851–3859.
Krishnan S, Warke VG, Nambiar MP, Tsokos GC, Farber DL . The FcRγ subunit and Syk kinase replace the CD3ζ-chain and ZAP-70 kinase in the TCR signaling complex of human effector CD4 T cells. J Immunol 2003; 170: 4189–4195.
Lozza L, Rivino L, Guarda G, Jarrossay D, Rinaldi A, Bertoni F et al. The strength of T cell stimulation determines IL-7 responsiveness, secondary expansion, and lineage commitment of primed human CD4+IL-7Rhi T cells. Eur J Immunol 2008; 38: 30–39.
Al-Rawi MA, Mansel RE, Jiang WG . Interleukin-7 (IL-7) and IL-7 receptor (IL-7R) signalling complex in human solid tumours. Histol Histopathol 2003; 18: 911–923.
Suzuki R, Suzuki S, Takahashi T, Kumagai K . Production of a cytokine with interleukin 3-like properties and cytokine-dependent proliferation in human autologous mixed lymphocyte reaction. J Exp Med 1986; 164: 1682–1699.
Knospe S, Köhler E, Rjasanowski I, Titze K . Autologous mixed lymphocyte reaction in newly diagnosed type-1 diabetes. Exp Clin Endocrinol 1990; 95: 148–156.
Hirsch RL . Defective autologous mixed lymphocyte reactivity in multiple sclerosis. Clin Exp Immunol 1986; 64: 107–113.
Felix NJ, Donermeyer DL, Horvath S, Walters JJ, Gross ML, Suri A et al. Alloreactive T cells respond specifically to multiple distinct peptide–MHC complexes. Nat Immunol 2007; 8: 388–397.
Eberl G, Brawand P, MacDonald HR . Selective bystander proliferation of memory CD41 and CD81 T cells upon NK T or T cell activation. J Immunol 2000; 165: 4305–4311.
Di Genova G, Roddick J, McNicholl F, Stevenson FK . Vaccination of human subjects expands both specific and bystander memory T cells but antibody production remains vaccine specific. Blood 2006; 107: 2806–2813.
Bangs SC, Baban D, Cattan HJ, Li CK, McMichael AJ, Xu XN . Human CD4+ memory T cells are preferential targets for bystander activation and apoptosis. J Immunol 2009; 182: 1962–1971.
Germain RN, Stefanová I . The dynamics of T cell receptor signaling: complex orchestration and the key roles of tempo and cooperation. Annu Rev Immunol 1999; 17: 467–522.
Marrack P, Ignatowicz L, Kappler JW, Boymel J, Freed JH . Comparison of peptides bound to spleen and thymus class II. J Exp Med 1993; 178: 2173–2183.
Ge Q, Palliser D, Eisen HN, Chen J . Homeostatic T cell proliferation in a T cell-dendritic cell coculture system. Proc Natl Acad Sci USA 2002; 99: 2983–2988.
Rogers PR, Dubey C, Swain SL . Qualitative changes accompany memory T cell generation: faster, more effective responses at lower doses of antigen. J Immunol 2000; 164: 2338–2346.
Chetoui N, Boisvert M, Gendron S, Aoudjit F . Interleukin-7 promotes the survival of human CD4+ effector/memory T cells by up-regulating Bcl-2 proteins and activating the JAK/STAT signalling pathway. Immunology 2010; 130: 418–426.
Kovanen PE, Rosenwald A, Fu J, Hurt EM, Lam LT, Giltnane JM et al. Analysis of gamma c-family cytokine target genes. Identification of dual-specificity phosphatase 5 (DUSP5) as a regulator of mitogen-activated protein kinase activity in interleukin-2 signaling. J Biol Chem 2003; 278: 5205–5213.
Arnaud M, Crouin C, Deon C, Loyaux D, Bertoglio J . Phosphorylation of Grb2-associated binder 2 on serine 623 by ERK MAPK regulates its association with the phosphatase SHP-2 and decreases STAT5 activation. J Immunol 2004; 173: 3962–3971.
Gett AV, Sallusto F, Lanzavecchia A, Geginat J . T cell fitness determined by signal strength. Nat Immunol 2003; 4: 355–360.
Kieper WC, Burghardt JT, Surh CD . A role for TCR affinity in regulating naive T cell homeostasis. J Immunol 2004; 172: 40–44.
Acknowledgements
We gratefully acknowledge the generosity of the blood donors and the support of the Blood Bank staff at Centro Medico Nacional La Raza Hospital (Mexico City) in providing the blood samples. We also thank Victor H. Rosales for help with flow cytometry, Dr. Nicolás Villegas for his aid in caspase-3 detection, Julio C. Ramírez for technical assistance, and Ms. Ninfa Arreola for her aid in the preparation of the manuscript. This work was supported by grants to C.S.T. from CONACYT (no. 81112 and 140980).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Supplementary information
Rights and permissions
About this article
Cite this article
González-Pérez, G., Segovia, N., Rivas-Carvalho, A. et al. Human CD4+ effector T lymphocytes generated upon TCR engagement with self-peptides respond defectively to IL-7 in their transition to memory cells. Cell Mol Immunol 10, 261–274 (2013). https://doi.org/10.1038/cmi.2012.71
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/cmi.2012.71