Abstract
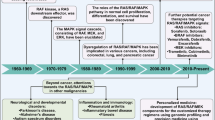
The extracellular signal-regulated kinase 1/2 (ERK1/2) cascade is the prototype mammalian mitogen-activated protein kinase (MAPK) signaling cascade that regulates a number of processes, including proliferation, differentiation, survival, migration, stress responses and apoptosis. How this seemingly linear cascade is modulated to achieve a specific cellular function has been a main focus of the field. In this review, we describe new as well as old findings in the regulation of the ERK1/2 pathway in normal and disease states via MAP3Ks.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Boulton TG, Yancopoulos GD, Gregory JS, et al. An insulin-stimulated protein kinase similar to yeast kinases involved in cell cycle control. Science 1990; 249:64–67.
Robinson LC, Gibbs JB, Marshall MS, Sigal IS, Tatchell K . CDC25: a component of the RAS-adenylate cyclase pathway in Saccharomyces cerevisiae. Science 1987; 235:1218–1221.
Broek D, Toda T, Michaeli T, et al. The S. cerevisiae CDC25 gene product regulates the RAS/adenylate cyclase pathway. Cell 1987; 48:789–799.
Simon MA, Bowtell DD, Dodson GS, Laverty TR, Rubin GM . Ras1 and a putative guanine nucleotide exchange factor perform crucial steps in signaling by the sevenless protein tyrosine kinase. Cell 1991; 67:701–716.
Olivier JP, Raabe T, Henkemeyer M, et al. A Drosophila SH2-SH3 adaptor protein implicated in coupling the sevenless tyrosine kinase to an activator of Ras guanine nucleotide exchange. Sos. Cell 1993; 73:179–191.
Lowenstein EJ, Daly RJ, Batzer AG, et al. The SH2 and SH3 domain-containing protein GRB2 links receptor tyrosine kinases to ras signaling. Cell 1992; 70:431–442.
Rogge RD, Karlovich CA, Banerjee U . Genetic dissection of a neurodevelopmental pathway: Son of sevenless functions downstream of the sevenless and EGF receptor tyrosine kinases. Cell 1991; 64:39–48.
Van Aelst L, Barr M, Marcus S, Polverino A, Wigler M . Complex formation between RAS and RAF and other protein kinases. Proc Natl Acad Sci USA 1993; 90:6213–6217.
Luo Z, Tzivion G, Belshaw PJ, Vavvas D, Marshall M, Avruch J . Oligomerization activates c-Raf-1 through a Ras-dependent mechanism. Nature 1996; 383:181–185.
Dent P, Haser W, Haystead TA, Vincent LA, Roberts TM, Sturgill TW . Activation of mitogen-activated protein kinase kinase by v-Raf in NIH 3T3 cells and in vitro. Science 1992; 257:1404–1407.
Kyriakis JM, App H, Zhang XF, et al. Raf-1 activates MAP kinase-kinase. Nature 1992; 358:417–421.
Seger R, Ahn NG, Posada J, et al. Purification and characterization of mitogen-activated protein kinase activator(s) from epidermal growth factor-stimulated A431 cells. J Biol Chem 1992; 267:14373–14381.
Robbins DJ, Cobb MH . Extracellular signal-regulated kinases 2 autophosphorylates on a subset of peptides phosphorylated in intact cells in response to insulin and nerve growth factor: analysis by peptide mapping. Mol Biol Cell 1992; 3:299–308.
Marshall CJ . Specificity of receptor tyrosine kinase signaling: transient versus sustained extracellular signal-regulated kinase activation. Cell 1995; 80:179–185.
Michaloglou C, Vredeveld LC, Mooi WJ, Peeper DS . BRAF(E600) in benign and malignant human tumours. Oncogene 2008; 27:877–895.
Greenman C, Stephens P, Smith R, et al. Patterns of somatic mutation in human cancer genomes. Nature 2007; 446:153–158.
Wan PT, Garnett MJ, Roe SM, et al. Mechanism of activation of the RAF-ERK signaling pathway by oncogenic mutations of B-RAF. Cell 2004; 116:855–867.
Davies H, Bignell GR, Cox C, et al. Mutations of the BRAF gene in human cancer. Nature 2002; 417:949–954.
Mason CS, Springer CJ, Cooper RG, Superti-Furga G, Marshall CJ, Marais R . Serine and tyrosine phosphorylations cooperate in Raf-1, but not B-Raf activation. EMBO J 1999; 18:2137–2148.
Weber CK, Slupsky JR, Kalmes HA, Rapp UR . Active Ras induces heterodimerization of cRaf and BRaf. Cancer Res 2001; 61:3595–3598.
Emuss V, Garnett M, Mason C, Marais R . Mutations of C-RAF are rare in human cancer because C-RAF has a low basal kinase activity compared with B-RAF. Cancer Res 2005; 65:9719–9726.
Hatzivassiliou G, Song K, Yen I, et al. RAF inhibitors prime wild-type RAF to activate the MAPK pathway and enhance growth. Nature 2010; 464:431–435.
Poulikakos PI, Zhang C, Bollag G, Shokat KM, Rosen N . RAF inhibitors transactivate RAF dimers and ERK signalling in cells with wild-type BRAF. Nature 2010; 464:427–430.
Tsai J, Lee JT, Wang W, et al. Discovery of a selective inhibitor of oncogenic B-Raf kinase with potent antimelanoma activity. Proc Natl Acad Sci USA 2008; 105:3041–3046.
Heidorn SJ, Milagre C, Whittaker S, et al. Kinase-dead BRAF and oncogenic RAS cooperate to drive tumor progression through CRAF. Cell 2010; 140:209–221.
McKay MM, Ritt DA, Morrison DK . RAF inhibitor-induced KSR1/B-RAF binding and its effects on ERK cascade signaling. Curr Biol 2011; 21:563–568.
Therrien M, Chang HC, Solomon NM, Karim FD, Wassarman DA, Rubin GM . KSR, a novel protein kinase required for RAS signal transduction. Cell 1995; 83:879–888.
Therrien M, Michaud NR, Rubin GM, Morrison DK . KSR modulates signal propagation within the MAPK cascade. Genes Dev 1996; 10:2684–2695.
Goettel JA, Liang D, Hilliard VC, et al. KSR1 is a functional protein kinase capable of serine autophosphorylation and direct phosphorylation of MEK1. Exp Cell Res 2011; 317:452–463.
Roy F, Laberge G, Douziech M, Ferland-McCollough D, Therrien M . KSR is a scaffold required for activation of the ERK/MAPK module. Genes Dev 2002; 16:427–438.
Sundaram M, Han M . The C. elegans ksr-1 gene encodes a novel Raf-related kinase involved in Ras-mediated signal transduction. Cell 1995; 83:889–901.
Dougherty MK, Ritt DA, Zhou M, et al. KSR2 is a calcineurin substrate that promotes ERK cascade activation in response to calcium signals. Mol Cell 2009; 34:652–662.
Xu B, English JM, Wilsbacher JL, Stippec S, Goldsmith EJ, Cobb MH . WNK1, a novel mammalian serine/threonine protein kinase lacking the catalytic lysine in subdomain II. J Biol Chem 2000; 275:16795–16801.
Matheny SA, Chen C, Kortum RL, Razidlo GL, Lewis RE, White MA . Ras regulates assembly of mitogenic signalling complexes through the effector protein IMP. Nature 2004; 427:256–260.
Chen C, Lewis RE, White MA . IMP modulates KSR1-dependent multivalent complex formation to specify ERK1/2 pathway activation and response thresholds. J Biol Chem 2008; 283:12789–12796.
Nguyen A, Burack WR, Stock JL, et al. Kinase suppressor of Ras (KSR) is a scaffold which facilitates mitogen-activated protein kinase activation in vivo. Mol Cell Biol 2002; 22:3035–3045.
Shalin SC, Hernandez CM, Dougherty MK, Morrison DK, Sweatt JD . Kinase suppressor of Ras1 compartmentalizes hippocampal signal transduction and subserves synaptic plasticity and memory formation. Neuron 2006; 50:765–779.
Costanzo-Garvey DL, Pfluger PT, Dougherty MK, et al. KSR2 is an essential regulator of AMP kinase, energy expenditure, and insulin sensitivity. Cell Metab 2009; 10:366–378.
Arnette D, Gibson TB, Lawrence MC, et al. Regulation of ERK1 and ERK2 by glucose and peptide hormones in pancreatic beta cells. J Biol Chem 2003; 278:32517–32525.
Lawrence MC, McGlynn K, Park BH, Cobb MH . ERK1/2-dependent activation of transcription factors required for acute and chronic effects of glucose on the insulin gene promoter. J Biol Chem 2005; 280:26751–26759.
Marshak S, Totary H, Cerasi E, Melloul D . Purification of the beta-cell glucose-sensitive factor that transactivates the insulin gene differentially in normal and transformed islet cells. Proc Natl Acad Sci USA 1996; 93:15057–15062.
Naya FJ, Stellrecht CM, Tsai MJ . Tissue-specific regulation of the insulin gene by a novel basic helix-loop-helix transcription factor. Genes Dev 1995; 9:1009–1019.
Lawrence MC, Bhatt HS, Easom RA . NFAT regulates insulin gene promoter activity in response to synergistic pathways induced by glucose and glucagon-like peptide-1. Diabetes 2002; 51:691–698.
Lawrence MC, Bhatt HS, Watterson JM, Easom RA . Regulation of insulin gene transcription by a Ca(2+)-responsive pathway involving calcineurin and nuclear factor of activated T cells. Mol Endocrinol 2001; 15:1758–1767.
Sander M, Griffen SC, Huang J, German MS . A novel glucose-responsive element in the human insulin gene functions uniquely in primary cultured islets. Proc Natl Acad Sci USA 1998; 95:11572–11577.
German MS, Wang J . The insulin gene contains multiple transcriptional elements that respond to glucose. Mol Cell Biol 1994; 14:4067–4075.
Khoo S, Griffen SC, Xia Y, Baer RJ, German MS, Cobb MH . Regulation of insulin gene transcription by ERK1 and ERK2 in pancreatic beta cells. J Biol Chem 2003; 278:32969–32977.
Luo C, Burgeon E, Carew JA, et al. Recombinant NFAT1 (NFATp) is regulated by calcineurin in T cells and mediates transcription of several cytokine genes. Mol Cell Biol 1996; 16:3955–3966.
Robertson RP . Cyclosporin-induced inhibition of insulin secretion in isolated rat islets and HIT cells. Diabetes 1986; 35:1016–1019.
Holz GG, Leech CA, Heller RS, Castonguay M, Habener JF . cAMP-dependent mobilization of intracellular Ca2+ stores by activation of ryanodine receptors in pancreatic beta-cells. A Ca2+ signaling system stimulated by the insulinotropic hormone glucagon-like peptide-1-(7-37). J Biol Chem 1999; 274:14147–14156.
Heit JJ, Apelqvist AA, Gu X, et al. Calcineurin/NFAT signalling regulates pancreatic beta-cell growth and function. Nature 2006; 443:345–349.
Duan L, Cobb MH . Calcineurin increases glucose activation of ERK1/2 by reversing negative feedback. Proc Natl Acad Sci USA 2010; 107:22314–22319.
Bokemeyer D, Sorokin A, Yan M, Ahn NG, Templeton DJ, Dunn MJ . Induction of mitogen-activated protein kinase phosphatase 1 by the stress-activated protein kinase signaling pathway but not by extracellular signal-regulated kinase in fibroblasts. J Biol Chem 1996; 271:639–642.
Beltman J, McCormick F, Cook SJ . The selective protein kinase C inhibitor, Ro-31-8220, inhibits mitogen-activated protein kinase phosphatase-1 (MKP-1) expression, induces c-Jun expression, and activates Jun N-terminal kinase. J Biol Chem 1996; 271:27018–27024.
Ritt DA, Monson DM, Specht SI, Morrison DK . Impact of feedback phosphorylation and Raf heterodimerization on normal and mutant B-Raf signaling. Mol Cell Biol 2010; 30:806–819.
Seger R, Ahn NG, Posada J, et al. Purification and characterization of mitogen-activated protein kinase activator(s) from epidermal growth factor-stimulated A431 cells. J Biol Chem 1992; 267:14373–14381.
Anderson NG, Maller JL, Tonks NK, Sturgill TW . Requirement for integration of signals from two distinct phosphorylation pathways for activation of MAP kinase. Nature 1990; 343:651–653.
Alessi DR, Smythe C, Keyse SM . The human CL100 gene encodes a Tyr/Thr-protein phosphatase which potently and specifically inactivates MAP kinase and suppresses its activation by oncogenic ras in Xenopus oocyte extracts. Oncogene 1993; 8:2015–2020.
Camps M, Nichols A, Gillieron C, et al. Catalytic activation of the phosphatase MKP-3 by ERK2 mitogen-activated protein kinase. Science 1998; 280:1262–1265.
Brion L, Maloberti PM, Gomez NV, et al. MAPK phosphatase-1 (MKP-1) expression is up-regulated by hCG/cAMP and modulates steroidogenesis in MA-10 Leydig cells. Endocrinology 2011; 152:2665–2677.
Ally S, Clair T, Katsaros D, et al. Inhibition of growth and modulation of gene expression in human lung carcinoma in athymic mice by site-selective 8-Cl-cyclic adenosine monophosphate. Cancer Res 1989; 49:5650–5655.
Cook SJ, McCormick F . Inhibition by cAMP of Ras-dependent activation of Raf. Science 1993; 262:1069–1072.
Gagelin C, Toru-Delbauffe D, Gavaret JM, Pierre M . Effects of cyclic AMP on components of the cell cycle machinery regulating DNA synthesis in cultured astrocytes. J Neurochem 1999; 73:1799–1805.
Kikukawa M, Okamoto Y, Fukui H, Nakano H . Effect of dibutyryl cyclic AMP on the cyclin-dependent kinase inhibitor p27Kip1 in the human hepatoma cells PLC/PRF/5. Anticancer Res 1997; 17:3287–3291.
Miller MJ, Rioux L, Prendergast GV, Cannon S, White MA, Meinkoth JL . Differential effects of protein kinase A on Ras effector pathways. Mol Cell Biol 1998; 18:3718–3726.
Li H, Robinson PJ, Kawashima S, Funder JW, Liu JP . Differential regulation of MAP kinase activity by corticotropin-releasing hormone in normal and neoplastic corticotropes. Int J Biochem Cell Biol 1998; 30:1389–1401.
Dumaz N, Hayward R, Martin J, et al. In melanoma, RAS mutations are accompanied by switching signaling from BRAF to CRAF and disrupted cyclic AMP signaling. Cancer Res 2006; 66:9483–9491.
Frodin M, Peraldi P, Van Obberghen E . Cyclic AMP activates the mitogen-activated protein kinase cascade in PC12 cells. J Biol Chem 1994; 269:6207–6214.
Bos JL . Epac: a new cAMP target and new avenues in cAMP research. Nature reviews. Mol Cell Biol 2003; 4:733–738.
Busca R, Abbe P, Mantoux F, et al. Ras mediates the cAMP-dependent activation of extracellular signal-regulated kinases (ERKs) in melanocytes. EMBO J 2000; 19:2900–2910.
Kiermayer S, Biondi RM, Imig J, et al. Epac activation converts cAMP from a proliferative into a differentiation signal in PC12 cells. Mol Biol Cell 2005; 16:5639–5648.
Jivan A, Earnest S, Juang YC, Cobb MH . Radial spoke protein 3 is a mammalian protein kinase A-anchoring protein that binds ERK1/2. J Biol Chem 2009; 284:29437–29445.
Diviani D, Soderling J, Scott JD . AKAP-Lbc anchors protein kinase A and nucleates Galpha 12-selective Rho-mediated stress fiber formation. J Biol Chem 2001; 276:44247–44257.
Smith FD, Langeberg LK, Cellurale C, et al. AKAP-Lbc enhances cyclic AMP control of the ERK1/2 cascade. Nat Cell Biol 2010; 12:1242–1249.
Dumaz N, Marais R . Protein kinase A blocks Raf-1 activity by stimulating 14-3-3 binding and blocking Raf-1 interaction with Ras. J Biol Chem 2003; 278:29819–29823.
Wu J, Dent P, Jelinek T, Wolfman A, Weber MJ, Sturgill TW . Inhibition of the EGF-activated MAP kinase signaling pathway by adenosine 3′,5′-monophosphate. Science 1993; 262:1065–1069.
Garnett MJ, Rana S, Paterson H, Barford D, Marais R . Wild-type and mutant B-RAF activate C-RAF through distinct mechanisms involving heterodimerization. Mol Cell 2005; 20:963–969.
Tzivion G, Luo Z, Avruch J . A dimeric 14-3-3 protein is an essential cofactor for Raf kinase activity. Nature 1998; 394:88–92.
Marquette A, André J, Bagot M, Bensussan A, Dumaz N . ERK and PDE4 cooperate to induce RAF isoform switching in melanoma. Nat Struct Mol Biol 2011; 18:584–591.
Balmanno K, Millar T, McMahon M, Cook SJ . DeltaRaf-1:ER* bypasses the cyclic AMP block of extracellular signal-regulated kinase 1 and 2 activation but not CDK2 activation or cell cycle reentry. Mol Cell Biol 2003; 23:9303–9317.
Pearson GW, Earnest S, Cobb MH . Cyclic AMP selectively uncouples mitogen-activated protein kinase cascades from activating signals. Mol Cell Biol 2006; 26:3039–3047.
Kraus AC, Ferber I, Bachmann SO, et al. In vitro chemo- and radio-resistance in small cell lung cancer correlates with cell adhesion and constitutive activation of AKT and MAP kinase pathways. Oncogene 2002; 21:8683–8695.
Sun S, Schiller JH, Spinola M, Minna JD . New molecularly targeted therapies for lung cancer. J Clin Invest 2007; 117:2740–2750.
Ravi RK, Weber E, McMahon M, et al. Activated Raf-1 causes growth arrest in human small cell lung cancer cells. J Clin Invest 1998; 101:153–159.
Ma PC, Tretiakova MS, Nallasura V, Jagadeeswaran R, Husain AN, Salgia R . Downstream signalling and specific inhibition of c-MET/HGF pathway in small cell lung cancer: implications for tumour invasion. Br J Cancer 2007; 97:368–377.
Arcaro A, Khanzada UK, Vanhaesebroeck B, Tetley TD, Waterfield MD, Seckl MJ . Two distinct phosphoinositide 3-kinases mediate polypeptide growth factor-stimulated PKB activation. EMBO J 2002; 21:5097–5108.
Bondzi C, Litz J, Dent P, Krystal GW . Src family kinase activity is required for Kit-mediated mitogen-activated protein (MAP) kinase activation, however loss of functional retinoblastoma protein makes MAP kinase activation unnecessary for growth of small cell lung cancer cells. Cell Growth Differ 2000; 11:305–314.
Meuwissen R, Linn SC, Linnoila RI, Zevenhoven J, Mooi WJ, Berns A . Induction of small cell lung cancer by somatic inactivation of both Trp53 and Rb1 in a conditional mouse model. Cancer Cell 2003; 4:181–189.
Sutherland KD, Proost N, Brouns I, Adriaensen D, Song TY, Berns A Berns A . Cell of origin of small cell lung cancer: inactivation of Trp53 and Rb1 in distinct cell types of adult mouse lung. Cancer Cell 2011; 19: 754–764.
Blackhall FH, Pintilie M, Michael M, et al. Expression and prognostic significance of kit, protein kinase B, and mitogen-activated protein kinase in patients with small cell lung cancer. Clin Cancer Res 2003; 9:2241–2247.
Nayeem N, Kerr F, Naumann H, Linehan J, Lovestone S, Brandner S . Hyperphosphorylation of tau and neurofilaments and activation of CDK5 and ERK1/2 in PTEN-deficient cerebella. Mol Cell Neurosci 2007; 34:400–408.
Bost F, Aouadi M, Caron L, et al. The extracellular signal-regulated kinase isoform ERK1 is specifically required for in vitro and in vivo adipogenesis. Diabetes 2005; 54:402–411.
Rodriguez A, Durán A, Selloum M, et al. Mature-onset obesity and insulin resistance in mice deficient in the signaling adapter p62. Cell Metab 2006; 3:211–222.
Bamford S, Dawson E, Forbes S, et al. The COSMIC (Catalogue of Somatic Mutations in Cancer) database and website. Br J Cancer 2004; 91:355–358.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is cited by
-
Genetic variants in Ras/Raf/MEK/ERK pathway are associated with gastric cancer risk in Chinese Han population
Archives of Toxicology (2020)
-
The lack of Raf-1 kinase feedback regulation enhances antiapoptosis in cancer cells
Oncogene (2017)
-
The nuclear translocation of ERK1/2 as an anticancer target
Nature Communications (2015)
-
C-Raf deficiency leads to hearing loss and increased noise susceptibility
Cellular and Molecular Life Sciences (2015)
-
Protein interaction switches coordinate Raf-1 and MST2/Hippo signalling
Nature Cell Biology (2014)