Abstract
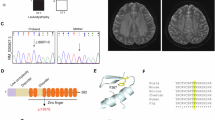
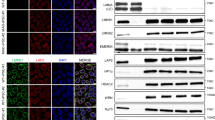
Mutation in ZMPSTE24 gene, encoding a major metalloprotease, leads to defective prelamin A processing and causes type B mandibuloacral dysplasia, as well as the lethal neonatal restrictive dermopathy syndrome. Phenotype severity is correlated with the residual enzyme activity of ZMPSTE24 and accumulation of prelamin A. We had previously demonstrated that a complete loss of function in ZMPSTE24 was lethal in the neonatal period, whereas compound heterozygous mutations including one PTC and one missense mutation were associated with type B mandibuloacral dysplasia. In this study, we report a 30-year longitudinal clinical survey of a patient harboring a novel severe and complex phenotype, combining an early-onset progeroid syndrome and a congenital myopathy with fiber-type disproportion. A unique homozygous missense ZMPSTE24 mutation (c.281T>C, p.Leu94Pro) was identified and predicted to produce two possible ZMPSTE24 conformations, leading to a partial loss of function. Western blot analysis revealed a major reduction of ZMPSTE24, together with the presence of unprocessed prelamin A and decreased levels of lamin A, in the patient's primary skin fibroblasts. These cells exhibited significant reductions in lifespan associated with major abnormalities of the nuclear shape and structure. This is the first report of MAD presenting with confirmed myopathic abnormalities associated with ZMPSTE24 defects, extending the clinical spectrum of ZMPSTE24 gene mutations. Moreover, our results suggest that defective prelamin A processing affects muscle regeneration and development, thus providing new insights into the disease mechanism of prelamin A-defective associated syndromes in general.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Kilic F, Johnson DA, Sinensky M : Subcellular localization and partial purification of prelamin A endoprotease: an enzyme which catalyzes the conversion of farnesylated prelamin A to mature lamin A. FEBS Lett 1999; 450: 61–65.
Leung GK, Schmidt WK, Bergo MO et al: Biochemical studies of Zmpste24-deficient mice. J Biol Chem 2001; 276: 29051–29058.
Lutz RJ, Trujillo MA, Denham KS, Wenger L, Sinensky M : Nucleoplasmic localization of prelamin A: implications for prenylation-dependent lamin A assembly into the nuclear lamina. Proc Natl Acad Sci USA 1992; 89: 3000–3004.
Varela I, Pereira S, Ugalde AP et al: Combined treatment with statins and aminobisphosphonates extends longevity in a mouse model of human premature aging. Nat Med 2008; 14: 767–772.
Sinensky M, Fantle K, Trujillo M, McLain T, Kupfer A, Dalton M : The processing pathway of prelamin A. J Cell Sci 1994; 107 (Part 1): 61–67.
Corrigan DP, Kuszczak D, Rusinol AE et al: Prelamin A endoproteolytic processing in vitro by recombinant Zmpste24. Biochem J 2005; 387: 129–138.
Broers J, Ramaekers F, Bonne G, Ben Yaou R, Hutchison C : The nuclear lamins: laminopathies and their role in premature ageing. Physiological Reviews 2006; 86: 967–1008.
Worman HJ, Bonne G : ‘Laminopathies’: a wide spectrum of human diseases. Exp Cell Res 2007; 313: 2121–2133.
Agarwal AK, Fryns JP, Auchus RJ, Garg A : Zinc metalloproteinase, ZMPSTE24, is mutated in mandibuloacral dysplasia. Hum Mol Genet 2003; 12: 1995–2001.
Navarro C, De Sandre-Giovannoli A, Bernard R et al: Lamin A and ZMPSTE24 (FACE-1) defects cause nuclear disorganisation and identify restrictive dermopathy as a lethal neonatal laminopathy. Hum Mol Genet 2004; 13: 2493–2503.
Navarro CL, Cadinanos J, Sandre-Giovannoli AD et al: Loss of ZMPSTE24 (FACE-1) causes autosomal recessive restrictive dermopathy and accumulation of Lamin A precursors. Hum Mol Genet 2005; 14: 1503–1513.
Witt DR, Hayden MR, Holbrook KA, Dale BA, Baldwin VJ, Taylor GP : Restrictive dermopathy: a newly recognized autosomal recessive skin dysplasia. Am J Med Genet 1986; 24: 631–648.
Novelli G, Muchir A, Sangiuolo F et al: Mandibuloacral dysplasia is caused by a mutation in LMNA encoding lamins A/C. Am J Hum Genet 2002; 71: 426–431.
Shackleton S, Smallwood DT, Clayton P et al: Compound heterozygous ZMPSTE24 mutations reduce prelamin A processing and result in a severe progeroid phenotype. J Med Genet 2005; 42: e36.
Denecke J, Brune T, Feldhaus T et al: A homozygous ZMPSTE24 null mutation in combination with a heterozygous mutation in the LMNA gene causes Hutchinson-Gilford progeria syndrome (HGPS): insights into the pathophysiology of HGPS. Hum Mutat 2006; 27: 524–531.
Sander CS, Salman N, van Geel M et al: A newly identified splice site mutation in ZMPSTE24 causes restrictive dermopathy in the Middle East. Br J Dermatol 2008; 159: 961–967.
Smigiel R, Jakubiak A, Esteves-Vieira V et al: Novel frameshifting mutations of the ZMPSTE24 gene in two siblings affected with restrictive dermopathy and review of the mutations described in the literature. Am J Med Genet A 2010; 152A: 447–452.
Pendas AM, Zhou Z, Cadinanos J et al: Defective prelamin A processing and muscular and adipocyte alterations in Zmpste24 metalloproteinase-deficient mice. Nat Genet 2002; 31: 94–99.
Bergo MO, Gavino B, Ross J et al: Zmpste24 deficiency in mice causes spontaneous bone fractures, muscle weakness, and a prelamin A processing defect. Proc Natl Acad Sci USA 2002; 99: 13049–13054.
Fong LG, Frost D, Meta M et al: A protein farnesyltransferase inhibitor ameliorates disease in a mouse model of progeria. Science 2006; 311: 1621–1623.
Ben Yaou R, Toutain A, Arimura T et al: Multitissular involvement in a family with LMNA and EMD mutations: role of digenic mechanism? Neurology 2007; 68: 1883–1894.
Muchir A, Medioni J, Laluc M et al: Nuclear envelope alterations in fibroblasts from patients with muscular dystrophy, cardiomyopathy, and partial lipodystrophy carrying lamin A/C gene mutations. Muscle Nerve 2004; 30: 444–450.
Decary S, Mouly V, Hamida CB, Sautet A, Barbet JP, Butler-Browne GS : Replicative potential and telomere length in human skeletal muscle: implications for satellite cell-mediated gene therapy. Hum Gene Ther 1997; 8: 1429–1438.
Thill M, Nguyen TD, Wehnert M et al: Restrictive dermopathy: a rare laminopathy. Arch Gynecol Obstet 2008; 278: 201–208.
Moulson CL, Go G, Gardner JM et al: Homozygous and compound heterozygous mutations in ZMPSTE24 cause the laminopathy restrictive dermopathy. J Invest Dermatol 2005; 125: 913–919.
Morais P, Magina S, Ribeiro Mdo C et al: Restrictive dermopathy—a lethal congenital laminopathy. case report and review of the literature. Eur J Pediatr 2009; 168: 1007–1012.
Kariminejad A, Goodarzi P, Thanh Huong le T, Wehnert MS : Restrictive dermopathy. Molecular diagnosis of restrictive dermopathy in a stillborn fetus from a consanguineous Iranian family. Saudi Med J 2009; 30: 150–153.
Chen M, Kuo HH, Huang YC et al: A case of restrictive dermopathy with complete chorioamniotic membrane separation caused by a novel homozygous nonsense mutation in the ZMPSTE24 gene. Am J Med Genet A 2009; 149A: 1550–1554.
Jagadeesh S, Bhat L, Suresh I, Muralidhar SL : Prenatal diagnosis of restrictive dermopathy. Indian Pediatr 2009; 46: 349–351.
Li C : Homozygosity for the common mutation c.1085dupT in the ZMPSTE24 gene in a Mennonite baby with restrictive dermopathy and placenta abruption. Am J Med Genet A 2010; 152A: 262–263.
Fong LG, Ng JK, Meta M et al: Heterozygosity for Lmna deficiency eliminates the progeria-like phenotypes in Zmpste24-deficient mice. Proc Natl Acad Sci USA 2004; 101: 18111–18116.
Agarwal AK, Zhou XJ, Hall RK et al: Focal segmental glomerulosclerosis in patients with mandibuloacral dysplasia owing to ZMPSTE24 deficiency. J Investig Med 2006; 54: 208–213.
Miyoshi Y, Akagi M, Agarwal AK et al: Severe mandibuloacral dysplasia caused by novel compound heterozygous ZMPSTE24 mutations in two Japanese siblings. Clin Genet 2008; 73: 535–544.
Cunningham VJ, D'Apice MR, Licata N, Novelli G, Cundy T : Skeletal phenotype of mandibuloacral dysplasia associated with mutations in ZMPSTE24. Bone 2010; 47: 591–597.
Goldman RD, Shumaker DK, Erdos MR et al: Accumulation of mutant lamin A causes progressive changes in nuclear architecture in Hutchinson-Gilford progeria syndrome. Proc Natl Acad Sci USA 2004; 101: 8963–8968.
Agarwal AK, Kazachkova I, Ten S, Garg A : Severe mandibuloacral dysplasia-associated lipodystrophy and progeria in a young girl with a novel homozygous Arg527Cys LMNA mutation. J Clin Endocrinol Metab 2008; 93: 4617–4623.
Van Esch H, Agarwal AK, Debeer P, Fryns JP, Garg A : A homozygous mutation in the lamin A/C gene associated with a novel syndrome of arthropathy, tendinous calcinosis, and progeroid features. J Clin Endocrinol Metab 2006; 91: 517–521.
Hennekam RC : Hutchinson-Gilford progeria syndrome: review of the phenotype. Am J Med Genet A 2006; 140: 2603–2624.
Kirschner J, Brune T, Wehnert M et al: p.S143F mutation in lamin A/C: a new phenotype combining myopathy and progeria. Ann Neurol 2005; 57: 148–151.
Madej-Pilarczyk A, Kmiec T, Fidzianska A et al: Progeria caused by a rare LMNA mutation p.S143F associated with mild myopathy and atrial fibrillation. Eur J Paediatr Neurol 2008; 12: 427–430.
Zirn B, Kress W, Grimm T et al: Association of homozygous LMNA mutation R471C with new phenotype: mandibuloacral dysplasia, progeria, and rigid spine muscular dystrophy. Am J Med Genet A 2008; 146A: 1049–1054.
Lombardi F, Gullotta F, Columbaro M et al: Compound heterozygosity for mutations in LMNA in a patient with a myopathic and lipodystrophic mandibuloacral dysplasia type A phenotype. J Clin Endocrinol Metab 2007; 92: 4467–4471.
Acknowledgements
We thank Jean-Paul Leroy for his help in the clinical investigation of the patient, Thomas Voit for critical reading and fruitful discussion and the Myocastor study group for its constant support. This work was financially supported by the Institut National de la Santé et de la Recherche Médicale; the Université Pierre et Marie Curie Paris 06; the Université de la Méditerranée, the Centre National de la Recherche Scientifique; the Association Française contre les Myopathies (no. 11057, no. 11034); the European Union Sixth (Euro-laminopathies no. 018690) and Seventh (Myoage no. 223576) Framework Programmes.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Yaou, R., Navarro, C., Quijano-Roy, S. et al. Type B mandibuloacral dysplasia with congenital myopathy due to homozygous ZMPSTE24 missense mutation. Eur J Hum Genet 19, 647–654 (2011). https://doi.org/10.1038/ejhg.2010.256
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/ejhg.2010.256
Keywords
This article is cited by
-
Loss of MTX2 causes mandibuloacral dysplasia and links mitochondrial dysfunction to altered nuclear morphology
Nature Communications (2020)
-
Mandibuloacral dysplasia type B (MADB): a cohort of eight patients from Suriname with a homozygous founder mutation in ZMPSTE24 (FACE1), clinical diagnostic criteria and management guidelines
Orphanet Journal of Rare Diseases (2019)
-
New ZMPSTE24 (FACE1) mutations in patients affected with restrictive dermopathy or related progeroid syndromes and mutation update
European Journal of Human Genetics (2014)
-
Inner nuclear membrane proteins: impact on human disease
Chromosoma (2012)