Abstract
Non-Mendelian parental effects are a form of phenotypic plasticity that influences offspring development and modifies their phenotype. They could have major evolutionary impact by changing the process of selection and the correspondence between offspring genotypes and phenotypes. To examine the influence of parental effects on life-history traits in the obliquebanded leafroller, Choristoneura rosaceana (Harris), three successive generations were raised in a constant laboratory environment, the first and subsequent generations being produced by field-raised and laboratory-raised parents, respectively. Between-generation variation in phenotypic means, variances and among- and within-family variance components were used to assess the magnitude of the influence of parental effects on allelic expression. The differences between parent-offspring regressions derived using parents from the first and second generations were used to estimate the directionality of the influence of parental effects on the family means of the traits.
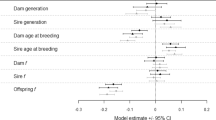
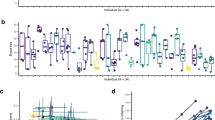
It appears that parental effects influenced the mean and variance of life-history traits at population and family levels. For development time and within-family diapausing propensity, the families of parents developing in a specific environment (laboratory or field) were apparently affected by a similar parental effect. For larval growth rate and pupal weight, it appears that different parental effects transmitted by field parents influenced the family means. Consequently, parental effects transmitted by field parents did not change the correspondence between the offspring phenotypic and breeding values in the first two traits but modified this relationship in the later traits. Parental effects influenced simultaneously the range of phenotypes produced by a genotype and its mean phenotype. Therefore, they may represent a major developmental mechanism by which organisms adapt to unpredictable and predictable environments.
Similar content being viewed by others
Article PDF
References
Becker, W A. 1984. Manual of Quantitative Genetics. Academic Enterprise, Pullman, WA.
Bradshaw, A D. 1965. Evolutionary significance of phenotypic plasticity in plants. Adv Genet, 13, 115–155.
Bull, J J. 1987. Evolution of phenotypic variance. Evolution, 41, 303–315.
Carrière, Y. 1991. Ecological Genetics of Host Exploitation in a Generalist Herbivore, the Obliquebanded Leafroller. Ph.D. Thesis, Simon Fraser University.
Carrière, Y. 1992. Host plant exploitation within a population of a generalist herbivore, Choristoneura rosaceana. Entomol Exp Appl, 65, 1–10.
Carrière, Y, and Roitberg, B D. 1994. Trade-offs in responses to host plants within a population of a generalist herbivore, Choristoneura rosaceana (Lepidoptera: Tortricidae). Entomol Exp Appl, (in press).
Chapman, P J, and Lienk, S E. 1971. Tortricid Fauna of Apple in New York. Special publication. New York State Agricultural Experimental Station Geneva, NY.
Falconer, D S. 1981. Introduction to Quantitative Genetics 2nd edn. Longmans, New York.
Gangavalli, R R, and Aliniazee, M T. 1985. Diapause induction in the obliquebanded leafroller Choristoneura rosaceana (Lepidoptera: Tortricidae): role of photoperiod and temperature. J Insect Physiol, 31, 831–835.
Giesel, J T. 1986. Genetic correlation structure of life history variables in outbred, wild Drosophila melanogaster. effects of photoperiod regimen. Am Nat, 128, 593–603.
Giesel, J T. 1988. Effects of parental photoperiod on development time and density sensitivity of progeny of Drosophila melanogaster. Evolution, 42, 1348–1350.
Gomulkiewicz, R, and Kirkpatrick, M. 1992. Quantitative genetics and the evolution of reaction norms. Evolution, 46, 390–411.
Groeters, F R, and Dingle, H. 1987. Genetic and maternal influences on life history plasticity in response to photoperiod by milkweed bugs (Oncopeltus fasciatus). Am Nat, 129, 332–346.
Gupta, A R, and Lewontin, R C. 1982. A study of reaction norms in natural populations of Drosophila pseudoobscura. Evolution, 36, 934–948.
Hardie, J, and Lee, A D. 1985. Endocrine control of polymorphism and polyphenism. In: G. A. Kerkuk and L. I. Gilbert (eds) Comprehensive Insect Physiology, Biochemistry and Pharmacology, 8, pp. 441–490. Pergamon Press, Toronto.
Holloway, G J, Povey, S R, and Sibly, R M. 1990. The effect of new environment on adapted genetic architecture. Heredity, 64, 323–330.
Janssen, G M, De Jong, G, Joosse, E N G, and Scharloo, W. 1988. A negative maternal effect in springtails. Evolution, 42, 828–834.
Kaplan, R H, and Cooper, W S. 1984. The evolution of developmental plasticity in reproductive characteristics: an application of the ‘adaptive coin-flipping’ principle. Am Nat, 123, 393–410.
Kirkpatrick, M, and Lande, R. 1989. The evolution of maternal characters. Evolution, 43, 485–503.
Lande, R. 1979. Quantitative genetic analysis of multivariate evolution, applied to brain: body size allometry. Evolution, 33, 402–416.
Levins, R. 1968. Evolution in Changing Environments. Princeton University Press, Princeton, NJ.
Mousseau, T A. 1991. Geographic variation in maternalage effects on diapause in a cricket. Evolution, 45, 1053–1059.
Mousseau, T A, and Dingle, H. 1991a. Maternal effects in insect life histories. Ann Rev Entomol, 36, 511–535.
Mousseau, T A, and Dingle, H. 1991b. Maternal effects in insects: examples, constraints and geographic variation. In: Dudley, E. C. (ed.) The Unity of Evolutionary Biology, The Unity of Evolutionary Biology: Proceedings of the ICSEB IV Meeting, pp. 745–761. Dioscorides Press, Portland, OR.
Newman, R A. 1988. Adaptive plasticity in development of Scaphiopus couchii tadpoles in desert ponds. Evolution, 42, 774–783.
Rausher, M D. 1985. Competition, frequency-independent selection and diapause in Battus philenor butterflies. Flor Entomol, 69, 62–78.
Roff, D A. 1986. The genetic basis of wing dimorphism in the sand cricket, Gryllus firmus and its relevance to the evolution of wing dimorphism in insects. Heredity, 57, 221–231.
Roff, D A. 1990. Selection for changes in the incidence of wing dimorphism in Gryllus firmus. Heredity, 65, 163–168.
Roff, D A. 1994. Evolution of dimorphic traits: effect of directional selection on heritability. Heredity, (in press).
Rossiter, M C. 1991. Maternal effects generate variation in life history: consequences of egg weight plasticity in the gypsy moth. Funct Ecol, 5, 386–393.
SAS Institute Inc. 1988. SAS Users' Guide: Statistics Version 603 SAS Institute Ine, Cary, NC.
Scheiner, S M, Caplan, R L, and Lyman, R F. 1989. A search for trade-offs among life history traits in Drosophila melanogaster. Evol Ecol, 3, 51–63.
Schlichting, C D. 1986. The evolution of phenotypic plasticity in plants. Ann Rev Ecol Syst, 17, 667–693.
Schmalhausen, I I. 1949. Factors of Evolution: The Theory of Stabilizing Selection. Blakiston, Philadelphia.
Seger, J, and Brockmann, H J. 1987. What is bet-hedging?. In: Harvey, P. H. and Partridge, L. (eds) Oxford Surveys in Evolutionary Biology, 4, pp. 182–211. Oxford University Press, Oxford.
Shorey, H H, and Hale, R C. 1965. Mass rearing of the larvae of nine noctuid species on a simple artificial medium. J Econ Entomol, 58, 522–524.
Sokal, R R, and Rohlf, F J. 1981. Biometry, 2nd edn. Freeman and Co., New York.
Stearns, S C. 1989. The evolutionary significance of reaction norms. Bioscience, 39, 436–446.
Stearns, S C. 1992. The Evolution of Life Histories. Oxford University Press, Oxford.
Tauber, C A, and Tauber, M J. 1986. Genetic variation in all-or-none life-history traits of the lacewing Chrysoperla carnea. Can J Zool, 64, 1542–1544.
Van Noordwijk, A J, and Gebhardt, M. 1987. Reflections on the genetics of quantitative traits with continuous environmental variation. In: V. Loeschcke (ed.) Genetic Constraints on Adaptive Evolution, pp. 73–90. Springer Verlag, Berlin.
Van Tienderen, P H. 1991. Evolution of generalists and specialists in spatially heterogeneous environments. Evolution, 45, 1317–1331.
Via, S. 1987. Genetic constraints on the evolution of phenotypic plasticity. In: V. Loeschcke, (ed.) Genetic Constraints on Adaptive Evolution, pp. 47–71. Springer Verlag, Berlin.
Via, S. 1993. Adaptive phenotypic plasticity: target or byproduct of selection in a variable environment? Am Nat, 142, 352–365.
Via, S, and Lande, R. 1985. Genotype-environment interaction and the evolution of phenotypic plasticity. Evolution, 39, 505–523.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Carrière, Y. Evolution of phenotypic variance: non-Mendelian parental influences on phenotypic and genotypic components of life-history traits in a generalist herbivore. Heredity 72, 420–430 (1994). https://doi.org/10.1038/hdy.1994.58
Received:
Issue date:
DOI: https://doi.org/10.1038/hdy.1994.58
Keywords
This article is cited by
-
Summer diapause induced by high temperatures in the oriental tobacco budworm: ecological adaptation to hot summers
Scientific Reports (2016)
-
Effects of the larval host plant on the supercooling capacity and physiological characteristics of beet armyworm pupae, Spodoptera exigua (Lepidoptera: Noctuidae)
Journal of Plant Diseases and Protection (2014)
-
10.1007/BF02383552
CrossRef Listing of Deleted DOIs (2011)
-
The quantitative genetics of wing dimorphism under laboratory and ‘field’ conditions in the cricket Gryllus pennsylvanicus
Heredity (1997)
-
Change in genetic architecture resulting from the evolution of insecticide resistance: a theoretical and empirical analysis
Heredity (1995)