Abstract
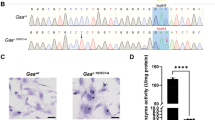
Glycogen-storage disease type II (GSDII) is an autosomal recessive disorder caused by a deficiency of acid α-glucosidase (GAA). The residual GAA activity is largely related to the severity of the clinical course. Most patients with infantile-onset GSDII do not show any enzyme activity, whereas patients with the late-onset forms of GSDII show various degrees of GAA activity. We performed a molecular genetic study on a Japanese boy with childhood-onset GSDII. The patient was a compound heterozygote for a newly discovered splice-site c.546G>T mutation and a recurrent missense p.R600C mutation, which usually causes the fatal infantile form in a homozygous state. The c.546G>T mutation, which did not alter the amino-acid sequence, was positioned at the last base of exon 2. cDNA-sequencing analysis revealed that c.546G>T was a leaky splice mutation, leading to the production of a normally spliced transcript, which was responsible for the low-level (approximately 10%) expression of the active enzyme in the patient's fibroblasts.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Accession codes
References
Hirschhorm, R. & Reuser, A. J. J. Glycogen storage disease type II. Acid alpha-glucosidase (acid maltase) deficiency In: Scriver CR, Beaudet AL, Sly WS, Valle D (eds) The metabolic and molecular bases of inherited disease, MacGraw-Hill: New York, pp 3389–3420 (2001).
Kuo, W. L., Hirschhorn, R., Huie, M. L. & Hirschhorn, K. Localization and ordering of acid alpha-glucosidase (GAA) and thymidine kinase (TK1) by fluorescence in situ hybridization. Hum. Genet. 97, 404–406 (1996).
Hoefsloot, L. H., Hoogeveen-Westerveld, M., Reuser, A. J. & Oostra, B. A. Characterization of the human lysosomal alpha-glucosidase gene. Biochem. J. 272, 493–497 (1990).
Moreland, R. J., Jin, X., Zhang, X. K., Decker, R. W., Albee, K. L. & Lee, K. L. Lysosomal acid alpha-glucosidase consists of four different peptides processed from a single chain precursor. J. Biol. Chem. 280, 6780–6791 (2005).
Hermans, M. M., Wisselaar, H. A., Kroos, M. A., Oostra, B. A. & Reuser, A. J. Human lysosomal alpha-glucosidase: functional characterization of the glycosylation sites. Biochem. J. 289, 681–686 (1993).
Wisselaar, H. A., Kroos, M. A., Hermans, M. M., van Beeumen, J. & Reuser, A. J. Structural and functional changes of lysosomal acid alpha-glucosidase during intracellular transport and maturation. J. Biol. Chem. 268, 2223–2231 (1993).
Kroos, M., Pomponio, R. J., van Vliet, L., Palmer, R. E., Phipps, M., Van der Helm, R. et al. Update of the Pompe disease mutation database with 107 sequence variants and a format for severity rating. Hum. Mutat. 29, E13–E26 (2008).
Tsujino, S., Huie, M., Kanazawa, N., Sugie, H., Goto, Y., Kawai, M. et al. Frequent mutations in Japanese patients with acid maltase deficiency. Neuromuscul. Disord. 10, 599–603 (2000).
Schoser, B., Hill, V. & Raben, N. Therapeutic approaches in glycogen storage disease type II/Pompe disease. Neurotherapeutics 5, 569–578 (2008).
Gort, L., Coll, M. J. & Chabás, A. Glycogen storage disease type II in Spanish patients: high frequency of c.1076-1G>C mutation. Mol. Genet. Metab. 92, 183–187 (2007).
Hermans, M. M., van Leenen, D., Kroos, M. A., Beesley, C. E., Van Der Ploeg, A. T., Sakuraba, H. et al. Twenty-two novel mutations in the lysosomal alpha-glucosidase gene (GAA) underscore the genotype-phenotype correlation in glycogen storage disease type II. Hum. Mutat. 23, 47–56 (2004).
Huie, M. L., Chen, A. S., Tsujino, S., Shanske, S., DiMauro, S., Engel, A. G. et al. Aberrant splicing in adult onset glycogen storage disease type II (GSDII): molecular identification of an IVS1 (-13T → G) mutation in a majority of patients and a novel IVS10 (+1GT → CT) mutation. Hum. Mol. Genet. 3, 2231–2236 (1994).
Nascimbeni, A. C., Fanin, M., Tasca, E. & Angelini, C. Molecular pathology and enzyme processing in various phenotypes of acid maltase deficiency. Neurology 70, 617–626 (2008).
Acknowledgements
We sincerely thank the members of the family for their participation, Dr Ikuya Nonaka and Dr Ichizo Nishino for their pathological examination of the muscle and Dr Hideo Sugie for his enzyme assay. This work was supported by a grant from Takeda Science Foundation for MM.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Maimaiti, M., Takahashi, S., Okajima, K. et al. Silent exonic mutation in the acid-α-glycosidase gene that causes glycogen storage disease type II by affecting mRNA splicing. J Hum Genet 54, 493–496 (2009). https://doi.org/10.1038/jhg.2009.66
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/jhg.2009.66
Keywords
This article is cited by
-
A generic assay for the identification of splicing variants that induce nonsense-mediated decay in Pompe disease
European Journal of Human Genetics (2021)
-
Splicing mutations in glycogen-storage disease type II: evaluation of the full spectrum of mutations and their relation to patients’ phenotypes
European Journal of Human Genetics (2011)