Abstract
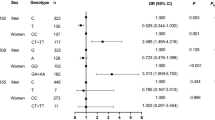
Several polymorphisms in the ApoA5 gene emerged as important candidate genes in triglyceride metabolism. The aim of this study was to determine the associations between ApoA5 polymorphisms, plasma triglyceride concentrations and the presence of cardiovascular disease (CVD) in three ethnic origins. Genotypes for 15 single nucleotide polymorphisms (SNPs) were determined in 659 older adults (mean age 71±7 years) who immigrated to Israel or whose ancestors originated from East Europe (Ashkenazi), North Africa, Asia (Sephardic) or Yemen (Yemenite). The minor alleles of the four common SNPs (rs662799, rs651821, rs2072560 and rs2266788) are associated with an increase of 27–38% in triglyceride concentration among Ashkenazi and Yemenite Jews compared with the major alleles, but not among those of Sephardic origin. Conversely, among the Sephardic group, the presence of the minor allele in SNP rs3135506 compared with the major allele was associated with an increase of 34% in triglyceride concentration. The four SNPs were in significant linkage disequilibrium (D′=0.96–0.99), resulting in three haplotypes H1, H2 and H3, representing 98–99% of the population. Haplotype H2 was significantly associated with triglyceride concentration among Ashkenazi and Yemenite but not among Sephardic Jews. Conversely, haplotype H3 was associated with triglyceride concentration in Sephardic but not in Ashkenazi and Yemenite Jews. Ashkenazi carriers of H2 haplotype had a CVD odds ratio of 2.19 (95% CI: 1.05–4.58) compared with H1 (the most frequent), after adjustment for all other risk factors. These results suggest that different SNPs in ApoA5 polymorphisms may be associated with triglyceride concentration and CVD in each of these ethnic origins.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Pennacchio, L. A., Olivier, M., Hubacek, J. A., Cohen, J. C., Cox, D. R., Fruchart, J. C. et al. An apolipoprotein influencing triglycerides in humans and mice revealed by comparative sequencing. Science 294, 169–173 (2001).
van Dijk, K. W., Rensen, P. C., Voshol, P. J. & Havekes, L. M. The role and mode of action of apolipoproteins CIII and AV: synergistic actors in triglyceride metabolism? Curr. Opin. Lipidol. 15, 239–246 (2004).
van der Vliet, H. N., Sammels, M. G., Leegwater, A. C., Levels, J. H., Reitsma, P. H., Boers, W. et al. Apolipoprotein A-V: a novel apolipoprotein associated with an early phase of liver regeneration. J. Biol. Chem. 276, 44512–44520 (2001).
Pennacchio, L. A., Olivier, M., Hubacek, J. A., Krauss, R. M., Rubin, E. M. & Cohen, J. C. Two independent apolipoprotein A5 haplotypes influence human plasma triglyceride levels. Hum. Mol. Genet. 11, 3031–3038 (2002).
Lai, C. Q., Demissie, S., Cupples, L. A., Zhu, Y., Adiconis, X., Parnell, L. D. et al. Influence of the APOA5 locus on plasma triglyceride, lipoprotein subclasses, and CVD risk in the Framingham Heart Study. J. Lipid. Res. 45, 2096–2105 (2004).
Ribalta, J., Figuera, L., Fernandez-Ballart, J., Vilella, E., Castro Cabezas, M., Masana, L. et al. Newly identified apolipoprotein AV gene predisposes to high plasma triglycerides in familial combined hyperlipidemia. Clin. Chem. 48, 1597–1600 (2002).
Talmud, P. J., Hawe, E., Martin, S., Olivier, M., Miller, G. J., Rubin, E. M. et al. Relative contribution of variation within the APOC3/A4/A5 gene cluster in determining plasma triglycerides. Hum. Mol. Genet. 11, 3039–3046 (2002).
Masana, L., Ribalta, J., Salazar, J., Fernandez-Ballart, J., Joven, J. & Cabezas, M. C. The apolipoprotein AV gene and diurnal triglyceridaemia in normolipidaemic subjects. Clin. Chem. Lab. Med. 41, 517–521 (2003).
Klos, K. L., Hamon, S., Clark, A. G., Boerwinkle, E., Liu, K. & Sing, C. F. APOA5 polymorphisms influence plasma triglycerides in young, healthy African Americans and whites of the CARDIA Study. J. Lipid. Res. 46, 564–571 (2005).
Lai, C. Q., Tai, E. S., Tan, C. E., Cutter, J., Chew, S. K., Zhu, Y. P. et al. The APOA5 locus is a strong determinant of plasma triglyceride concentrations across ethnic groups in Singapore. J. Lipid. Res. 44, 2365–2373 (2003).
Chandak, G. R., Ward, K. J., Yajnik, C. S., Pandit, A. N., Bavdekar, A., Joglekar, C. V. et al. Triglyceride associated polymorphisms of the APOA5 gene have very different allele frequencies in Pune, India compared to Europeans. BMC Med. Genet. 7, 76 (2006).
Kobyliansky, E., Micle, S., Goldschmidt-Nathan, M., Arensburg, B. & Nathan, H. Jewish populations of the world: genetic likeness and differences. Ann. Hum. Biol. 9, 1–34 (1982).
Bonne-Tamir, B., Karlin, S. & Kenett, R. Analysis of genetic data on Jewish populations. I. Historical background, demographic features, and genetic markers. Am. J. Hum. Genet. 31, 324–340 (1979).
Thomas, M. G., Parfitt, T., Weiss, D. A., Skorecki, K., Wilson, J. F., le Roux, M. et al. Y chromosomes traveling south: the Cohen modal haplotype and the origins of the Lemba—the ‘Black Jews of Southern Africa’. Am. J. Hum. Genet. 66, 674–686 (2000).
Goldbourt, U. Geographic origin of Jewish migrants, period of immigration and diversity of acculturation: is Israel a cardiovascular melting pot? Isr. Med. Assoc. J. 4, 370–372 (2002).
Karlin, S., Kenett, R. & Bonne-Tamir, B. Analysis of biochemical genetic data on Jewish populations: II. Results and interpretations of heterogeneity indices and distance measures with respect to standards. Am. J. Hum. Genet. 31, 341–365 (1979).
Green, M. S., Jucha, E. & Luz, Y. Ethnic differences in selected cardiovascular disease risk factors in Israeli workers. Isr. J. Med. Sci. 21, 808–816 (1985).
Toor, M., Katchalsky, A., Agmon, J. & Allalouf, D. Atherosis and related factors in immigrants to Israel. Circulation 22, 265–279 (1960).
Goldbourt, U. & Kark, J. D. The epidemiology of coronary heart disease in the ethnically and culturally diverse population of Israel. A review. Isr. J. Med. Sci. 18, 1077–1097 (1982).
Kallner, G. & Groen, J. J. Mortality from coronary (arteriosclerotic) heart disease and cerebrovascular accidents among eastern immigrants in Israel. J. Chronic Dis. 21, 25–35 (1968).
Medalie, J. H., Kahn, H. A., Neufeld, H. N., Riss, E., Goldbourt, U., Perlstein, T. et al. Myocardial infarction over a five-year period. I. Prevalence, incidence and mortality experience. J. Chronic Dis. 26, 63–84 (1973).
Hokanson, J. E. & Austin, M. A. Plasma triglyceride level is a risk factor for cardiovascular disease independent of high-density lipoprotein cholesterol level: a meta-analysis of population-based prospective studies. J. Cardiovasc. Risk 3, 213–219 (1996).
Prineas, R. J., Crow, R. S. & Blackburn, H The Minnesota Code Manual of Electocardiographic Findings: Standards and Procedures for Measurement and Classification (John Wright, Boston, Massachusetts; Bristol, 1982).
Department of Health Education and Welfare. Lipid Research Clinics Program: Manual of Laboratory Operations, Lipid and Lipoprotein Analysis (NIH Publication No. 76-628. US Government Printing Office, Washington, DC, 1976 ).
Jurinke, C., Denissenko, M. F., Oeth, P., Ehrich, M., van den Boom, D. & Cantor, C. R. A single nucleotide polymorphism based approach for the identification and characterization of gene expression modulation using MassARRAY. Mutat. Res. 573, 83–95 (2005).
Stephens, M., Smith, N. J. & Donnelly, P. A new statistical method for haplotype reconstruction from population data. Am. J. Hum. Genet. 68, 978–989 (2001).
Stephens, M. & Scheet, P. Accounting for decay of linkage disequilibrium in haplotype inference and missing-data imputation. Am. J. Hum. Genet. 76, 449–462 (2005).
Graham, R. R., Langefeld, C. D., Gaffney, P. M., Ortmann, W. A., Selby, S. A., Baechler, E. C. et al. Genetic linkage and transmission disequilibrium of marker haplotypes at chromosome 1q41 in human systemic lupus erythematosus. Arthritis Res. 3, 299–305 (2001).
Rothman, K. J. No adjustments are needed for multiple comparisons. Epidemiology 1, 43–46 (1990).
Eichenbaum-Voline, S., Olivier, M., Jones, E. L., Naoumova, R. P., Jones, B., Gau, B. et al. Linkage and association between distinct variants of the APOA1/C3/A4/A5 gene cluster and familial combined hyperlipidemia. Arterioscler. Thromb. Vasc. Biol. 24, 167–174 (2004).
Nabika, T., Nasreen, S., Kobayashi, S. & Masuda, J. The genetic effect of the apoprotein AV gene on the serum triglyceride level in Japanese. Atherosclerosis 165, 201–204 (2002).
Endo, K., Yanagi, H., Araki, J., Hirano, C., Yamakawa-Kobayashi, K. & Tomura, S. Association found between the promoter region polymorphism in the apolipoprotein A-V gene and the serum triglyceride level in Japanese schoolchildren. Hum. Genet. 111, 570–572 (2002).
Lee, K. W., Ayyobi, A. F., Frohlich, J. J. & Hill, J. S. APOA5 gene polymorphism modulates levels of triglyceride, HDL cholesterol and FERHDL but is not a risk factor for coronary artery disease. Atherosclerosis 176, 165–172 (2004).
Talmud, P. J., Martin, S., Taskinen, M. R., Frick, M. H., Nieminen, M. S., Kesaniemi, Y. A. et al. APOA5 gene variants, lipoprotein particle distribution, and progression of coronary heart disease: results from the LOCAT study. J. Lipid. Res. 45, 750–756 (2004).
Martin, S., Nicaud, V., Humphries, S. E. & Talmud, P. J. Contribution of APOA5 gene variants to plasma triglyceride determination and to the response to both fat and glucose tolerance challenges. Biochim. Biophys. Acta. 1637, 217–225 (2003).
Aouizerat, B. E., Kulkarni, M., Heilbron, D., Drown, D., Raskin, S., Pullinger, C. R. et al. Genetic analysis of a polymorphism in the human apoA-V gene: effect on plasma lipids. J. Lipid Res. 44, 1167–1173 (2003).
Baum, L., Tomlinson, B. & Thomas, G. N. APOA5-1131T>C polymorphism is associated with triglyceride levels in Chinese men. Clin. Genet. 63, 377–379 (2003).
Austin, M. A., Talmud, P. J., Farin, F. M., Nickerson, D. A., Edwards, K. L., Leonetti, D. et al. Association of apolipoprotein A5 variants with LDL particle size and triglyceride in Japanese Americans. Biochim. Biophys. Acta. 1688, 1–9 (2004).
Groenendijk, M., Cantor, R. M., de Bruin, T. W. & Dallinga-Thie, G. M. The apoAI-CIII-AIV gene cluster. Atherosclerosis 157, 1–11 (2001).
Wong, W. M., Hawe, E., Li, L. K., Miller, G. J., Nicaud, V., Pennacchio, L. A. et al. Apolipoprotein AIV gene variant S347 is associated with increased risk of coronary heart disease and lower plasma apolipoprotein AIV levels. Circ. Res. 92, 969–975 (2003).
Schaap, F. G., Rensen, P. C., Voshol, P. J., Vrins, C., van der Vliet, H. N., Chamuleau, R. A. et al. ApoAV reduces plasma triglycerides by inhibiting very low density lipoprotein-triglyceride (VLDL-TG) production and stimulating lipoprotein lipase-mediated VLDL-TG hydrolysis. J. Biol. Chem. 279, 27941–27947 (2004).
Dallongeville, J., Cottel, D., Montaye, M., Codron, V., Amouyel, P. & Helbecque, N. Impact of APOA5/A4/C3 genetic polymorphisms on lipid variables and cardiovascular disease risk in French men. Int. J. Cardiol. 106, 152–156 (2006).
Szalai, C., Keszei, M., Duba, J., Prohaszka, Z., Kozma, G. T., Csaszar, A. et al. Polymorphism in the promoter region of the apolipoprotein A5 gene is associated with an increased susceptibility for coronary artery disease. Atherosclerosis 173, 109–114 (2004).
Hubacek, J. A., Skodova, Z., Adamkova, V., Lanska, V. & Poledne, R. The influence of APOAV polymorphisms (T-1131>C and S19>W) on plasma triglyceride levels and risk of myocardial infarction. Clin. Genet. 65, 126–130 (2004).
Burchard, E. G., Ziv, E., Coyle, N., Gomez, S. L., Tang, H., Karter, A. J. et al. The importance of race and ethnic background in biomedical research and clinical practice. N. Engl. J. Med. 348, 1170–1175 (2003).
Acknowledgements
We thank Professor Chao-Qiang Lai for his excellent technical assistance. Genotyping was performed at the Genome Knowledge Center at the Weizmann Institute of Science, supported by the Israel Ministry of Science and the Crown Human Genome Center.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest
Rights and permissions
About this article
Cite this article
Ken-Dror, G., Goldbourt, U. & Dankner, R. Different effects of apolipoprotein A5 SNPs and haplotypes on triglyceride concentration in three ethnic origins. J Hum Genet 55, 300–307 (2010). https://doi.org/10.1038/jhg.2010.27
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/jhg.2010.27
Keywords
This article is cited by
-
SNPs in apolipoproteins contribute to sex-dependent differences in blood lipids before and after a high-fat dietary challenge in healthy U.S. adults
BMC Nutrition (2022)
-
Effects of apolipoprotein A5 haplotypes on the ratio of triglyceride to high-density lipoprotein cholesterol and the risk for metabolic syndrome in Koreans
Lipids in Health and Disease (2014)
-
Genetic association of lipid metabolism related SNPs with myocardial infarction in the Pakistani population
Molecular Biology Reports (2014)