Abstract
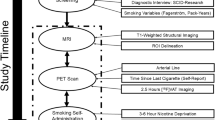
Cigarette smoking leads to upregulation of brain nicotinic acetylcholine receptors (nAChRs), including the common α4β2* nAChR subtype. Although a substantial percentage of smokers receive treatment for tobacco dependence with counseling and/or medication, the effect of a standard course of these treatments on nAChR upregulation has not yet been reported. In the present study, 48 otherwise healthy smokers underwent positron emission tomography (PET) scanning with the radiotracer 2-FA (for labeling α4β2* nAChRs) before and after treatment with either cognitive-behavioral therapy, bupropion HCl, or pill placebo. Specific binding volume of distribution (VS/fP), a measure proportional to α4β2* nAChR density, was determined for regions known to have nAChR upregulation with smoking (prefrontal cortex, brainstem, and cerebellum). In the overall study sample, significant decreases in VS/fP were found for the prefrontal cortex, brainstem, and cerebellum of −20 (±35), −25 (±36), and −25 (±31)%, respectively, which represented movement of VS/fP values toward values found in non-smokers (mean 58.2% normalization of receptor levels). Participants who quit smoking had significantly greater reductions in VS/fP across regions than non-quitters, and correlations were found between reductions in cigarettes per day and decreases in VS/fP for brainstem and cerebellum, but there was no between-group effect of treatment type. Thus, smoking reduction and cessation with commonly used treatments (and pill placebo) lead to decreased α4β2* nAChR densities across brain regions. Study findings could prove useful in the treatment of smokers by providing encouragement with the knowledge that decreased smoking leads to normalization of specific brain receptors.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Abrams DB, Niaura RS, Brown RA, Emmons KM, Goldstein MG, Monti PM (2003) The Tobacco Dependence Treatment Handbook: A Guide to Best Practices. The Guilford Press: New York.
Agboola S, McNeill A, Coleman T, Leonardi Bee J (2010). A systematic review of the effectiveness of smoking relapse prevention interventions for abstinent smokers. Addiction 105: 1362–1380.
Al’Absi M, Carr SB, Bongard S (2007). Anger and psychobiological changes during smoking abstinence and in response to acute stress: prediction of smoking relapse. Int J Psychophysiol 66: 109–115.
Batra A, Collins SE, Torchalla I, Schroter M, Buchkremer G (2008). Multidimensional smoker profiles and their prediction of smoking following a pharmacobehavioral intervention. J Subst Abuse Treat 35: 41–52.
Beck A, Steer R (1996) Manual for BDI-II. Psychological Corporation: San Antonio, TX, USA.
Benwell ME, Balfour DJK, Anderson JM (1988). Evidence that tobacco smoking increases the density of (-)-[3H]nicotine binding sites in human brain. J Neurochem 50: 1243–1247.
Bergen AW, Javitz HS, Krasnow R, Nishita D, Michel M, Conti DV et al (2013). Nicotinic acetylcholine receptor variation and response to smoking cessation therapies. Pharmacogenet Genomics 23: 94–103.
Breese CR, Marks MJ, Logel J, Adams CE, Sullivan B, Collins AC et al (1997). Effect of smoking history on [3H]nicotine binding in human postmortem brain. J Pharmacol Exp Ther 282: 7–13.
Brody AL, Mandelkern MA, Costello MR, Abrams AL, Scheibal D, Farahi J et al (2009). Brain nicotinic acetylcholine receptor occupancy: effect of smoking a denicotinized cigarette. Int J Neuropsychopharmacol 12: 305–316.
Brody AL, Mandelkern MA, London ED, Khan A, Kozman D, Costello MR et al (2011). Effect of secondhand smoke on occupancy of nicotinic acetylcholine receptors in brain. Arch Gen Psychiatry 68: 953–960.
Brody AL, Mandelkern MA, London ED, Olmstead RE, Farahi J, Scheibal D et al (2006). Cigarette smoking saturates brain alpha4beta2 nicotinic acetylcholine receptors. Arch Gen Psychiatry 63: 907–915.
Brody AL, Mukhin AG, La Charite J, Ta K, Farahi J, Sugar CA et al (2013). Up-regulation of nicotinic acetylcholine receptors in menthol cigarette smokers. Int J Neuropsychopharmacol (e-pub ahead of print).
Cahill K, Stead LF, Lancaster T (2011). Nicotine receptor partial agonists for smoking cessation. Cochrane Database Syst Rev 2: CD006103.
Carmody TP (1990). Preventing relapse in the treatment of nicotine addiction: current issues and future directions. J Psychoactive Drugs 22: 211–238.
CDC (2009). Cigarette smoking among adults and trends in smoking cessation—United States 2008. MMWR Morb Mortal Wkly Rep 58: 1227–1232.
Cosgrove KP, Batis J, Bois F, Maciejewski PK, Esterlis I, Kloczynski T et al (2009). beta2-Nicotinic acetylcholine receptor availability during acute and prolonged abstinence from tobacco smoking. Arch Gen Psychiatry 66: 666–676.
Cosgrove KP, Esterlis I, McKee SA, Bois F, Seibyl JP, Mazure CM et al (2012). Sex differences in availability of beta2*-nicotinic acetylcholine receptors in recently abstinent tobacco smokers. Arch Gen Psychiatry 69: 418–427.
Dale LC, Glover ED, Sachs DP, Schroeder DR, Offord KP, Croghan IT et al (2001). Bupropion for smoking cessation: predictors of successful outcome. Chest 119: 1357–1364.
Dolle F, Valette H, Bottlaender M, Hinnen F, Vaufrey F, Guenther I et al (1998). Synthesis of 2-[F-18]fluoro-3-[2(S)-2-azetidinylmethoxy]pyridine, a highly potent radioligand for in vivo imaging central nicotinic acetylcholine receptors. J Labelled Compounds Radiopharm 41: 451–463.
Fant RV, Buchhalter AR, Buchman AC, Henningfield JE (2009). Pharmacotherapy for tobacco dependence. Handbook Exp Pharmacol 192: 487–510.
Fiore MC, Bailey WC, Cohen SJ, Dorfman SF, Goldstein MG, Gritz ER et al (2000) Treating Tobacco Use and Dependence. Clinical Practice Guideline. U.S. Department of Health and Human Services. Public Health Service: Rockville, MD, USA.
Fiore MC, Jaen CR, Baker TB (2008) Treating Tobacco Use and Dependence: 2008 Update. Department of Health and Human Services: Rockville, MD, USA.
Govind AP, Vezina P, Green WN (2009). Nicotine-induced upregulation of nicotinic receptors: underlying mechanisms and relevance to nicotine addiction. Biochem Pharmacol 78: 756–765.
Gwaltney CJ, Shiffman S, Balabanis MH, Paty JA (2005). Dynamic self-efficacy and outcome expectancies: prediction of smoking lapse and relapse. J Abnorm Psychol 114: 661–675.
Haaga DA, Stewart BL (1992). Self-efficacy for recovery from a lapse after smoking cessation. J Consult Clin Psychol 60: 24–28.
Hamilton M (1967). Development of a rating scale for primary depressive illness. Br J Soc Psychol 6: 278–296.
Hamilton M (1969). Diagnosis and rating of anxiety. Br J Psychiatry 3: 76–79.
Heatherton TF, Kozlowski LT, Frecker RC, Fagerstrom KO (1991). The Fagerstrom Test for Nicotine Dependence: a revision of the Fagerstrom Tolerance Questionnaire. Br J Addict 86: 1119–1127.
Hughes JR, Keely JP, Niaura RS, Ossip-Klein DJ, Richmond RL, Swan GE (2003). Measures of abstinence in clinical trials: issues and recommendations. Nicotine Tob Res 5: 13–25.
Hymowitz N, Cummings KM, Hyland A, Lynn WR, Pechacek TF, Hartwell TD (1997). Predictors of smoking cessation in a cohort of adult smokers followed for five years. Tob Control 6 (Suppl 2): S57–S62.
Innis RB, Cunningham VJ, Delforge J, Fujita M, Gjedde A, Gunn RN et al (2007). Consensus nomenclature for in vivo imaging of reversibly binding radioligands. J Cereb Blood Flow Metab 27: 1533–1539.
Jamal A, Dube SR, Malarcher AM, Shaw L, Engstrom MC (2012). Tobacco use screening and counseling during physician office visits among adults—National Ambulatory Medical Care Survey and National Health Interview Survey, United States, 2005-2009. MMWR Morb Mortal Wkly Rep 61 (Suppl): 38–45.
Japuntich SJ, Leventhal AM, Piper ME, Bolt DM, Roberts LJ, Fiore MC et al (2011). Smoker characteristics and smoking-cessation milestones. Am J Prev Med 40: 286–294.
Japuntich SJ, Smith SS, Jorenby DE, Piper ME, Fiore MC, Baker TB (2007). Depression predicts smoking early but not late in a quit attempt. Nicotine Tob Res 9: 677–686.
Jarvik ME, Madsen DC, Olmstead RE, Iwamoto-Schaap PN, Elins JL, Benowitz NL (2000). Nicotine blood levels and subjective craving for cigarettes. Pharmacol Biochem Behav 66: 553–558.
Kenford SL, Fiore MC, Jorenby DE, Smith SS, Wetter D, Baker TB (1994). Predicting smoking cessation. Who will quit with and without the nicotine patch. JAMA 271: 589–594.
Kimes AS, Chefer SI, Matochik JA, Contoreggi CS, Vaupel DB, Stein EA et al (2008). Quantification of nicotinic acetylcholine receptors in the human brain with PET: bolus plus infusion administration of 2-[18F]F-A85380. Neuroimage 39: 717–727.
King DP, Paciga S, Pickering E, Benowitz NL, Bierut LJ, Conti DV et al (2012). Smoking cessation pharmacogenetics: analysis of varenicline and bupropion in placebo-controlled clinical trials. Neuropsychopharmacology 37: 641–650.
Kozlowski LT, Porter CQ, Orleans CT, Pope MA, Heatherton T (1994). Predicting smoking cessation with self-reported measures of nicotine dependence: FTQ, FTND, and HSI. Drug Alcohol Depend 34: 211–216.
Lee W, Ray R, Bergen AW, Swan GE, Thomas P, Tyndale RF et al (2012). DRD1 associations with smoking abstinence across slow and normal nicotine metabolizers. Pharmacogenet Genomics 22: 551–554.
Lester HA, Xiao C, Srinivasan R, Son CD, Miwa J, Pantoja R et al (2009). Nicotine is a selective pharmacological chaperone of acetylcholine receptor number and stoichiometry. Implications for drug discovery. Am Assoc Pharm Scientists J 11: 167–177.
Levin ED, Westman EC, Stein RM, Carnahan E, Sanchez M, Herman S et al (1994). Nicotine skin patch treatment increases abstinence, decreases withdrawal symptoms, and attenuates rewarding effects of smoking. J Clin Psychopharmacol 14: 41–49.
Li WW, Froelicher ES (2008). Predictors of smoking relapse in women with cardiovascular disease in a 30-month study: extended analysis. Heart Lung 37: 455–465.
Mamede M, Ishizu K, Ueda M, Mukai T, Iida Y, Kawashima H et al (2007). Temporal change in human nicotinic acetylcholine receptor after smoking cessation: 5IA SPECT study. J Nucl Med 48: 1829–1835.
Marks MJ, McClure-Begley TD, Whiteaker P, Salminen O, Brown RW, Cooper J et al (2011). Increased nicotinic acetylcholine receptor protein underlies chronic nicotine-induced up-regulation of nicotinic agonist binding sites in mouse brain. J Pharmacol Exp Ther 337: 187–200.
Marubio LM, Gardier AM, Durier S, David D, Klink R, Arroyo-Jimenez MM et al (2003). Effects of nicotine in the dopaminergic system of mice lacking the alpha4 subunit of neuronal nicotinic acetylcholine receptors. Eur J Neurosci 17: 1329–1337.
McGranahan TM, Patzlaff NE, Grady SR, Heinemann SF, Booker TK (2011). alpha4beta2 nicotinic acetylcholine receptors on dopaminergic neurons mediate nicotine reward and anxiety relief. J Neurosci 31: 10891–10902.
McNair DM, Lorr D, Droppelman LF (1988) Manual for the Profile of Mood States. Educational Testing Services: San Diego, CA, USA.
Mitsis EM, Cosgrove KP, Staley JK, Frohlich EB, Bois F, Tamagnan GD et al (2007). [123I]5-IA-85380 SPECT imaging of beta2-nicotinic acetylcholine receptor availability in the aging human brain. Ann N Y Acad Sci 1097: 168–170.
Mukhin AG, Kimes AS, Chefer SI, Matochik JA, Contoreggi CS, Horti AG et al (2008). Greater nicotinic acetylcholine receptor density in smokers than in nonsmokers: a PET study with 2-18F-FA-85380. J Nucl Med 49: 1628–1635.
Paluck EC, McCormack JP, Ensom MH, Levine M, Soon JA, Fielding DW (2006). Outcomes of bupropion therapy for smoking cessation during routine clinical use. Ann Pharmacother 40: 185–190.
Quick MW, Lester RA (2002). Desensitization of neuronal nicotinic receptors. J Neurobiol 53: 457–478.
Rigotti NA, Gonzales D, Dale LC, Lawrence D, Chang Y (2009). A randomized controlled trial of adding the nicotine patch to rimonabant for smoking cessation: efficacy, safety and weight gain. Addiction 104: 266–276.
Rogers SW, Gahring LC, Collins AC, Marks M (1998). Age-related changes in neuronal nicotinic acetylcholine receptor subunit α4 expression are modified by long-term nicotine administration. J Neurosci 18: 4825–4832.
Schnoll RA, James C, Malstrom M, Rothman RL, Wang H, Babb J et al (2003). Longitudinal predictors of continued tobacco use among patients diagnosed with cancer. Ann Behav Med 25: 214–222.
Schnoll RA, Patterson F, Wileyto EP, Tyndale RF, Benowitz N, Lerman C (2009). Nicotine metabolic rate predicts successful smoking cessation with transdermal nicotine: a validation study. Pharmacol Biochem Behav 92: 6–11.
Shiffman S, Balabanis MH, Gwaltney CJ, Paty JA, Gnys M, Kassel JD et al (2007). Prediction of lapse from associations between smoking and situational antecedents assessed by ecological momentary assessment. Drug Alcohol Depend 91: 159–168.
Shiffman S, Balabanis MH, Paty JA, Engberg J, Gwaltney CJ, Liu KS et al (2000). Dynamic effects of self-efficacy on smoking lapse and relapse. Health Psychol 19: 315–323.
Shiffman SM, Jarvik ME (1976). Smoking Withdrawal Symptoms in Two Weeks of Abstinence. Psychopharmacology (Berl) 50: 35–39.
Slemmer JE, Martin BR, Damaj MI (2000). Bupropion is a nicotinic antagonist. J Pharmacol Exp Ther 295: 321–327.
Staley JK, Krishnan-Sarin S, Cosgrove KP, Krantzler E, Frohlich E, Perry E et al (2006). Human tobacco smokers in early abstinence have higher levels of beta2* nicotinic acetylcholine receptors than nonsmokers. J Neurosci 26: 8707–8714.
Tobey KM, Walentiny DM, Wiley JL, Carroll FI, Damaj MI, Azar MR et al (2012). Effects of the specific alpha4beta2 nAChR antagonist, 2-fluoro-3-(4-nitrophenyl) deschloroepibatidine, on nicotine reward-related behaviors in rats and mice. Psychopharmacology (Berl) 223: 159–168.
Walters CL, Brown S, Changeux JP, Martin B, Damaj MI (2006). The beta2 but not alpha7 subunit of the nicotinic acetylcholine receptor is required for nicotine-conditioned place preference in mice. Psychopharmacology (Berl) 184: 339–344.
Waters AJ, Shiffman S, Sayette MA, Paty JA, Gwaltney CJ, Balabanis MH (2004). Cue-provoked craving and nicotine replacement therapy in smoking cessation. J Consult Clin Psychol 72: 1136–1143.
West R, Hajek P, Stead L, Stapleton J (2005). Outcome criteria in smoking cessation trials: proposal for a common standard. Addiction 100: 299–303.
Westman EC, Behm FM, Simel DL, Rose JE (1997). Smoking behavior on the first day of a quit attempt predicts long-term abstinence. Arch Intern Med 157: 335–340.
Whiting PJ, Lindstrom JM (1988). Characterization of bovine and human neuronal nicotinic acetylcholine receptors using monoclonal antibodies. J Neurosci 8: 3395–3404.
Wiggers LC, Stalmeier PF, Oort FJ, Smets EM, Legemate DA, de Haes JC (2005). Do patients' preferences predict smoking cessation? Prev Med 41: 667–675.
Wullner U, Gundisch D, Herzog H, Minnerop M, Joe A, Warnecke M et al (2008). Smoking upregulates alpha4beta2* nicotinic acetylcholine receptors in the human brain. Neurosci Lett 430: 34–37.
Yates SL, Bencherif M, Fluhler EN, Lippiello PM (1995). Up-regulation of nicotinic acetylcholine receptors following chronic exposure of rats to mainstream cigarette smoke or alpha 4 beta 2 receptors to nicotine. Biochem Pharmacol 50: 2001–2008.
Zhang X, Tian JY, Svensson AL, Gong ZH, Meyerson B, Nordberg A (2002). Chronic treatments with tacrine and (-)-nicotine induce different changes of nicotinic and muscarinic acetylcholine receptors in the brain of aged rat. J Neural Transm 109: 377–392.
Acknowledgements
This study was supported by the National Institute on Drug Abuse (ALB (R01 DA20872)), the Tobacco-Related Disease Research Program (ALB (19XT-0135)), and the Department of Veterans Affairs, Office of Research and Development (CSR&D Merit Review Award I01 CX000412 (ALB)). The sponsors had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; or preparation, review, or approval of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
ALB and MAM were previously co-investigators on a grant funded by Pfizer, Inc. that was unrelated to this study. AGM is a co-investigator on a grant from Phillip Morris, Inc. for research unrelated to this study. All the other authors report no biomedical financial disclosures or potential conflicts of interest.
PowerPoint slides
Rights and permissions
About this article
Cite this article
Brody, A., Mukhin, A., Stephanie Shulenberger et al. Treatment for Tobacco Dependence: Effect on Brain Nicotinic Acetylcholine Receptor Density. Neuropsychopharmacol 38, 1548–1556 (2013). https://doi.org/10.1038/npp.2013.53
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/npp.2013.53
Keywords
This article is cited by
-
Cholinergic system adaptations are associated with cognitive function in people recently abstinent from smoking: a (-)-[18F]flubatine PET study
Neuropsychopharmacology (2023)
-
Cholinergic receptor binding in unimpaired older adults, mild cognitive impairment, and Alzheimer’s disease dementia
Alzheimer's Research & Therapy (2022)
-
Tabakentwöhnung
MMW - Fortschritte der Medizin (2020)
-
Bidirectional Associations among Nicotine and Tobacco Smoke, NeuroHIV, and Antiretroviral Therapy
Journal of Neuroimmune Pharmacology (2020)
-
Tobacco smoking and dopaminergic function in humans: a meta-analysis of molecular imaging studies
Psychopharmacology (2019)