Abstract
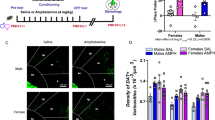
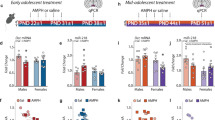
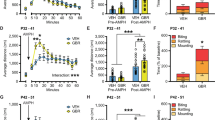
Initiation of drug use during adolescence is a strong predictor of both the incidence and severity of addiction throughout the lifetime. Intriguingly, adolescence is a period of dynamic refinement in the organization of neuronal connectivity, in particular medial prefrontal cortex (mPFC) dopamine circuitry. The guidance cue receptor, DCC (deleted in colorectal cancer), is highly expressed by dopamine neurons and orchestrates their innervation to the mPFC during adolescence. Furthermore, we have shown that amphetamine in adolescence regulates DCC expression in dopamine neurons. Drugs in adolescence may therefore induce their enduring behavioral effects via DCC-mediated disruption in mPFC dopamine development. In this study, we investigated the impact of repeated exposure to amphetamine during adolescence on both the development of mPFC dopamine connectivity and on salience attribution to drug context in adulthood. We compare these effects to those induced by adult exposure to an identical amphetamine regimen. Finally, we determine whether DCC signaling within dopamine neurons is necessary for these events. Exposure to amphetamine in adolescence, but not in adulthood, leads to an increase in the span of dopamine innervation to the mPFC, but a reduction of presynaptic sites present on these axons. Amphetamine treatment in adolescence, but not in adulthood, also produces an increase in salience attribution to a previously drug-paired context in adulthood. Remarkably, DCC signaling within dopamine neurons is required for both of these effects. Drugs of abuse in adolescence may therefore induce their detrimental behavioral consequences by disrupting mesocortical dopamine development through alterations in the DCC signaling cascade.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Anggård E, Gunne LM, Jönsson L-E, Niklasson F (1970). Pharmacokinetic and clinical studies on amphetamine dependent subjects. Eur J Clin Pharmacol 3: 3–11.
Anthony JC, Petronis KR (1995). Early-onset drug use and risk of later drug problems. Drug and Alcohol Depend 40: 9–15.
Antonopoulos J, Dori I, Dinopoulos A, Chiotelli M, Parnavelas JG (2002). Postnatal development of the dopaminergic system of the striatum in the rat. Neuroscience 110: 245–256.
Benes FM, Taylor JB, Cunningham MC (2000). Convergence and plasticity of monoaminergic systems in the medial prefrontal cortex during the postnatal period: implications for the development of psychopathology. Cereb Cortex 10: 1014–1027.
Benes FM, Vincent SL, Molloy R, Khan Y (1996). Increased interaction of dopamine-immunoreactive varicosities with GABA neurons of rat medial prefrontal cortex occurs during the postweanling period. Synapse 23: 237–245.
Cass DK, Thomases DR, Caballero A, Tseng KY (2013). Developmental disruption of gamma-aminobutyric acid function in the medial prefrontal cortex by noncontingent cocaine exposure during early adolescence. Biol Psychiatry 74: 490–501.
Cervo L, Samanin R (1996). Effects of dopaminergic and glutamatergic receptor antagonists on the establishment and expression of conditioned locomotion to cocaine in rats. Brain Res 731: 31–38.
Chao DL, Ma L, Shen K (2009). Transient cell-cell interactions in neural circuit formation. Nat Rev Neurosci 10: 262–271.
Colón-Ramos DA, Margeta MA, Shen K (2007). Glia promote local synaptogenesis through UNC-6 (netrin) signaling in C. elegans. Science 318: 103–106.
Currie CL, Wild TC (2012). Adolescent use of prescription drugs to get high in Canada. Can J Psychiatry 57: 745–751.
Deiner MS, Kennedy TE, Fazeli A, Serafini T, Tessier-Lavigne M, Sretavan DW (1997). Netrin-1 and DCC mediate axon guidance locally at the optic disc: loss of function leads to optic nerve hypoplasia. Neuron 19: 575–589.
Dent EW, Barnes AM, Tang F, Kalil K (2004). Netrin-1 and semaphorin 3A promote or inhibit cortical axon branching, respectively, by reorganization of the cytoskeleton. J Neurosci 24: 3002–3012.
Di Ciano P, Blaha CD, Phillips AG (1998). The relation between dopamine oxidation currents in the nucleus accumbens and conditioned increases in motor activity in rats following repeated administration of d-amphetamine or cocaine. Eur J Neurosci 10: 1113–1120.
Dickson BJ (2002). Molecular mechanisms of axon guidance. Science 298: 1959–1964.
Ersche KD, Barnes A, Jones PS, Morein-Zamir S, Robbins TW, Bullmore ET (2011). Abnormal structure of frontostriatal brain systems is associated with aspects of impulsivity and compulsivity in cocaine dependence. Brain 134: 2013–2024.
Flores C (2011). Role of netrin-1 in the organization and function of the mesocorticolimbic dopamine system. J Psychiatry Neurosci 36: 296–310.
Franklin TR, Druhan JP (2000). Involvement of the nucleus accumbens and medial prefrontal cortex in the expression of conditioned hyperactivity to a cocaine-associated environment in rats. Neuropsychopharmacology 23: 633–644.
Giedd JN, Blumenthal J, Jeffries NO, Castellanos FX, Liu H, Zijdenbos A et al (1999). Brain development during childhood and adolescence: a longitudinal MRI study. Nat Neurosci 2: 861–863.
Gogtay N, Giedd JN, Lusk L, Hayashi KM, Greenstein D, Vaituzis AC et al (2004). Dynamic mapping of human cortical development during childhood through early adulthood. Proc Natl Acad Sci USA 101: 8174–8179.
Goldman JS, Ashour MA, Magdesian MH, Tritsch NX, Harris SN, Christofi N et al (2013). Netrin-1 Promotes Excitatory Synaptogenesis between Cortical Neurons by Initiating Synapse Assembly. J Neurosci 33: 17278–17289.
Goldstein RZ, Volkow ND (2011). Dysfunction of the prefrontal cortex in addiction: neuroimaging findings and clinical implications. Nat Rev Neurosci 12: 652–669.
Grant A, Fathalli F, Rouleau G, Joober R, Flores C (2012). Association between schizophrenia and genetic variation in DCC: a case-control study. Schizophr Res 137: 26–31.
Grant A, Hoops D, Labelle-Dumais C, Prévost M, Rajabi H, Kolb B et al (2007). Netrin-1 receptor-deficient mice show enhanced mesocortical dopamine transmission and blunted behavioural responses to amphetamine. Eur J Neurosci 26: 3215–3228.
Grant BF, Dawson DA (2003). Age of onset of drug use and its association with DSM-IV drug abuse and dependence: results from the National Longitudinal Alcohol Epidemiologic Survey. J Subst Abuse 10: 163–173.
Gustavsen I, Mørland J, Bramness JG (2006). Impairment related to blood amphetamine and/or methamphetamine concentrations in suspected drugged drivers. Accid Anal Prev 38: 490–495.
Hall DA, Stanis JJ, Marquez Avila H, Gulley JM (2008). A comparison of amphetamine- and methamphetamine-induced locomotor activity in rats: evidence for qualitative differences in behavior. Psychopharmacology (Berl) 195: 469–478.
Kalivas PW (2008). Addiction as a pathology in prefrontal cortical regulation of corticostriatal habit circuitry. Neurotox Res 14: 185–189.
Kalsbeek A, Voorn P, Buijs RM, Pool CW, Uylings HB (1988). Development of the dopaminergic innervation in the prefrontal cortex of the rat. J Comp Neurol 269: 58–72.
Kohno M, Morales AM, Ghahremani DG, Hellemann G, London ED (2014). Risky decision making, prefrontal cortex, and mesocorticolimbic functional connectivity in methamphetamine dependence. JAMA Psychiatry 71: 812–820.
Kramer JC, Fischman VS, Littlefield DC (1967). Amphetamine abuse. Pattern and effects of high doses taken intravenously. JAMA 201: 305–309.
Kuczenski R, Segal DS (2005). Stimulant actions in rodents: implications for attention-deficit/hyperactivity disorder treatment and potential substance abuse. Biol Psychiatry 57: 1391–1396.
LaLumiere RT, Smith KC, Kalivas PW (2012). Neural circuit competition in cocaine-seeking: roles of the infralimbic cortex and nucleus accumbens shell. Eur J Neurosci 35: 614–622.
Laviola G, Macrì S, Morley-Fletcher S, Adriani W (2003). Risk-taking behavior in adolescent mice: psychobiological determinants and early epigenetic influence. Neurosci Biobehav Rev 27: 19–31.
Leatherdale ST, Burkhalter R (2012). The substance use profile of Canadian youth: exploring the prevalence of alcohol, drug and tobacco use by gender and grade. Addict Behav 37: 318–322.
Levitt P, Moore RY (1979). Development of the noradrenergic innervation of neocortex. Brain Res 162: 243–259.
Lidov HG, Grzanna R, Molliver ME (1980). The serotonin innervation of the cerebral cortex in the rat—an immunohistochemical analysis. Neuroscience 5: 207–227.
Madras BK (2010). Office of National Drug Control Policy: a scientist in drug policy in Washington, DC. Ann NY Acad Sci 1187: 370–402.
Mahmood OM, Goldenberg D, Thayer R, Migliorini R, Simmons AN, Tapert SF (2013). Adolescents' fMRI activation to a response inhibition task predicts future substance use. Addict Behav 38: 1435–1441.
Manitt C, Eng C, Pokinko M, Ryan RT, Torres-Berrío A, Lopez JP et al (2013). dcc orchestrates the development of the prefrontal cortex during adolescence and is altered in psychiatric patients. Transl Psychiatry 3: e338.
Manitt C, Labelle-Dumais C, Eng C, Grant A, Mimee A, Stroh T et al (2010). Peri-pubertal emergence of UNC-5 homologue expression by dopamine neurons in rodents. PLoS ONE 5: e11463.
Manitt C, Mimee A, Eng C, Pokinko M, Stroh T, Cooper HM et al (2011). The netrin receptor DCC is required in the pubertal organization of mesocortical dopamine circuitry. J Neurosci 31: 8381–8394.
Manitt C, Nikolakopoulou AM, Almario DR, Nguyen SA, Cohen-Cory S (2009). Netrin participates in the development of retinotectal synaptic connectivity by modulating axon arborization and synapse formation in the developing brain. J Neurosci 29: 11065–11077.
McCabe SE, West BT, Morales M, Cranford JA, Boyd CJ (2007). Does early onset of non-medical use of prescription drugs predict subsequent prescription drug abuse and dependence? Results from a national study. Addiction 102: 1920–1930.
McCutcheon JE, Conrad KL, Carr SB, Ford KA, McGehee DS, Marinelli M (2012). Dopamine neurons in the ventral tegmental area fire faster in adolescent rats than in adults. J Neurophysiol 108: 1620–1630.
Méneret A, Depienne C, Riant F, Trouillard O, Bouteiller D, Cincotta M et al (2014). Congenital mirror movements: Mutational analysis of RAD51 and DCC in 26 cases. Neurology 82: 1999–2002.
Miner LH, Schroeter S, Blakely RD, Sesack SR (2003). Ultrastructural localization of the norepinephrine transporter in superficial and deep layers of the rat prelimbic prefrontal cortex and its spatial relationship to probable dopamine terminals. J Comp Neurol 466: 478–494.
Naneix F, Marchand AR, Di Scala G, Pape J-R, Coutureau E (2012). Parallel maturation of goal-directed behavior and dopaminergic systems during adolescence. J Neurosci 32: 16223–16232.
O'Donnell P (2011). Adolescent onset of cortical disinhibition in schizophrenia: insights from animal models. Schizophr Bull 37: 484–492.
Osborne PB, Halliday GM, Cooper HM, Keast JR (2005). Localization of immunoreactivity for deleted in colorectal cancer (DCC), the receptor for the guidance factor netrin-1, in ventral tier dopamine projection pathways in adult rodents. Neuroscience 131: 671–681.
Paxinos G, Franklin KBJ (2008) The Mouse Brain in Stereotaxic Coordinates. 3rd edn. Amsterdam: Elselvier/Academic Press.
Peters J, Kalivas PW, Quirk GJ (2009). Extinction circuits for fear and addiction overlap in prefrontal cortex. Learn Mem 16: 279–288.
Peters J, LaLumiere RT, Kalivas PW (2008). Infralimbic prefrontal cortex is responsible for inhibiting cocaine seeking in extinguished rats. J Neurosci 28: 6046–6053.
Pfaffl MW (2001). A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29: e45.
Pokinko M, Moquin L, Gratton A, Flores C (2014). Mesocortical dopamine depletion reverses the protective phenotype of netrin-1 receptor deficient mice. Program No. 210.07/A51. 2014 Neuroscience Meeting Planner. Society for Neuroscience, 2014. Washington D.C. online.
Poon VY, Choi S, Park M (2013). Growth factors in synaptic function. Front Synaptic Neurosci 5: 6.
Reyes S, Fu Y, Double KL, Cottam V, Thompson LH, Kirik D et al (2013). Trophic factors differentiate dopamine neurons vulnerable to Parkinson's disease. Neurobiol Aging 34: 873–886.
Riffee WH, Ludden TM, Wilcox RE, Gerald MC (1978). Brain and plasma concentrations of amphetamine isomers in mice. J Pharmacol Exp Ther 206: 586–594.
Robbins TW, Roberts AC, Owen AM, Sahakian BJ, Everitt BJ, Wilkinson L et al (1994). Monoaminergic-dependent cognitive functions of the prefrontal cortex in monkey and man. Thierry A-M, Glowinski J, Goldman-Rakic PS (eds). Motor and Cognitive Functions of the Prefrontal Cortex. Springer-Verlag: Berlin, Heidelberg, 93–111.
Robins LN, Przybeck TR (1985). Age of onset of drug use as a factor in drug and other disorders. NIDA Res Monogr 56: 178–192.
Robinson TE, Berridge KC (1993). The neural basis of drug craving: an incentive-sensitization theory of addiction. Brain Res Brain Res Rev 18: 247–291.
Robinson TE, Berridge KC (2008). Review. The incentive sensitization theory of addiction: some current issues. Philos Trans R Soc Lond B Biol Sci 363: 3137–3146.
Rogers RD, Everitt BJ, Baldacchino A, Blackshaw AJ, Swainson R, Wynne K et al (1999). Dissociable deficits in the decision-making cognition of chronic amphetamine abusers, opiate abusers, patients with focal damage to prefrontal cortex, and tryptophan-depleted normal volunteers: evidence for monoaminergic mechanisms. Neuropsychopharmacology 20: 322–339.
Rosenberg DR, Lewis DA (1995). Postnatal maturation of the dopaminergic innervation of monkey prefrontal and motor cortices: a tyrosine hydroxylase immunohistochemical analysis. J Comp Neurol 358: 383–400.
SAMHSA (2012). Results from the 2011 National Survey on Drug Use and Health: Summary of national findings. NSDUH Series H-44, HHS Publication No (SMA) 12-4713. Substance Abuse and Mental Health Services Administration: Rockville, MD.
Schneider M (2013). Adolescence as a vulnerable period to alter rodent behavior. Cell Tissue Res 354: 99–106.
Séguéla P, Watkins KC, Descarries L (1988). Ultrastructural features of dopamine axon terminals in the anteromedial and the suprarhinal cortex of adult rat. Brain Res 442: 11–22.
Shaw P, Kabani NJ, Lerch JP, Eckstrand K, Lenroot R, Gogtay N et al (2008). Neurodevelopmental trajectories of the human cerebral cortex. J Neurosci 28: 3586–3594.
Sowell ER, Peterson BS, Thompson PM, Welcome SE, Henkenius AL, Toga AW (2003). Mapping cortical change across the human life span. Nat Neurosci 6: 309–315.
Sowell ER, Thompson PM, Holmes CJ, Jernigan TL, Toga AW (1999). In vivo evidence for post-adolescent brain maturation in frontal and striatal regions. Nat Neurosci 2: 859–861.
Spear LP (2000). The adolescent brain and age-related behavioral manifestations. Neurosci Biobehav Rev 24: 417–463.
Srour M, Rivière J-B, Pham JMT, Dubé M-P, Girard S, Morin S et al (2010). Mutations in DCC cause congenital mirror movements. Science 328: 592.
Stewart J, de Wit H, Eikelboom R (1984). Role of unconditioned and conditioned drug effects in the self-administration of opiates and stimulants. Psychol Rev 91: 251–268.
Swendsen J, Burstein M, Case B, Conway KP, Dierker L, He J et al (2012). Use and abuse of alcohol and illicit drugs in US adolescents: results of the National Comorbidity Survey-Adolescent Supplement. Arch Gen Psychiatry 69: 390–398.
Tirelli E, Laviola G, Adriani W (2003). Ontogenesis of behavioral sensitization and conditioned place preference induced by psychostimulants in laboratory rodents. Neurosci Biobehav Rev 27: 163–178.
Tomasi D, Goldstein RZ, Telang F, Maloney T, Alia-Klein N, Caparelli EC et al (2007). Widespread disruption in brain activation patterns to a working memory task during cocaine abstinence. Brain Res 1171: 83–92.
Tseng K-Y, O'Donnell P (2007). Dopamine modulation of prefrontal cortical interneurons changes during adolescence. Cereb Cortex 17: 1235–1240.
Van den Heuvel DMA, Pasterkamp RJ (2008). Getting connected in the dopamine system. Prog Neurobiol 85: 75–93.
Van Swearingen AED, Walker QD, Kuhn CM (2013). Sex differences in novelty- and psychostimulant-induced behaviors of C57BL/6 mice. Psychopharmacology (Berl) 225: 707–718.
Verdejo-García A, Bechara A, Recknor EC, Pérez-García M (2006). Executive dysfunction in substance dependent individuals during drug use and abstinence: an examination of the behavioral, cognitive and emotional correlates of addiction. J Int Neuropsychol Soc 12: 405–415.
Williams RW, Bartheld von CS, Rosen GD (2003). Counting cells in sectioned material: a suite of techniques, tools, and tips. Curr Protoc Neurosci Chapter 1: Unit 1.11.
Winberg ML, Mitchell KJ, Goodman CS (1998). Genetic analysis of the mechanisms controlling target selection: complementary and combinatorial functions of netrins, semaphorins, and IgCAMs. Cell 93: 581–591.
Woicik PA, Urban C, Alia-Klein N, Henry A, Maloney T, Telang F et al (2011). A pattern of perseveration in cocaine addiction may reveal neurocognitive processes implicit in the Wisconsin Card Sorting Test. Neuropsychologia 49: 1660–1669.
Yetnikoff L, Almey A, Arvanitogiannis A, Flores C (2011). Abolition of the behavioral phenotype of adult netrin-1 receptor deficient mice by exposure to amphetamine during the juvenile period. Psychopharmacology (Berl) 217: 505–514.
Yetnikoff L, Eng C, Benning S, Flores C (2010). Netrin-1 receptor in the ventral tegmental area is required for sensitization to amphetamine. Eur J Neurosci 31: 1292–1302.
Yetnikoff L, Labelle-Dumais C, Flores C (2007). Regulation of netrin-1 receptors by amphetamine in the adult brain. Neuroscience 150: 764–773.
Yetnikoff L, Pokinko M, Arvanitogiannis A, Flores C (2013a). Adolescence: a time of transition for the phenotype of dcc heterozygous mice. Psychopharmacology (Berl) 231: 1705–1714.
Yetnikoff L, Reichard RA, Schwartz ZM, Parsely KP, Zahm DS (2013b). Protracted maturation of forebrain afferent connections of the ventral tegmental area in the rat. J Comp Neurol 522: 1031–1047.
Acknowledgements
This work was funded by the Canadian Institutes of Health Research, Natural Science and Engineering Research Council of Canada, and the Fonds de Recherche du Québec—Santé. LMR is supported by a predoctoral STEM fellowship from Fulbright Canada.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Reynolds, L., Makowski, C., Yogendran, S. et al. Amphetamine in Adolescence Disrupts the Development of Medial Prefrontal Cortex Dopamine Connectivity in a dcc-Dependent Manner. Neuropsychopharmacol 40, 1101–1112 (2015). https://doi.org/10.1038/npp.2014.287
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/npp.2014.287
This article is cited by
-
Axo-glial interactions between midbrain dopamine neurons and oligodendrocyte lineage cells in the anterior corpus callosum
Brain Structure and Function (2023)
-
Guidance cues: linking drug use in adolescence with psychiatric disorders
Neuropsychopharmacology (2019)
-
MicroRNA Expression Profiling in the Prefrontal Cortex: Putative Mechanisms for the Cognitive Effects of Adolescent High Fat Feeding
Scientific Reports (2018)
-
Non-Contingent Exposure to Amphetamine in Adolescence Recruits miR-218 to Regulate Dcc Expression in the VTA
Neuropsychopharmacology (2018)
-
Adolescent cocaine exposure induces prolonged synaptic modifications in medial prefrontal cortex of adult rats
Brain Structure and Function (2017)