Key Points
-
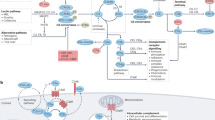
Complement is a key component of immunity with crucial inflammatory and opsonic properties; inappropriate activation of complement triggers or exacerbates inflammatory disease.
-
Complement dysregulation is a core feature of some diseases and contributes to pathology in many others.
-
Approved agents have been developed for and are highly effective in some orphan applications, but their progress to use in more common diseases has been slow.
-
Numerous challenges, such as target concentration or high turnover, limit the efficacy of these agents in humans.
-
Numerous novel agents targeting different parts of the complement system in different ways are now emerging from pre-clinical studies and are entering Phase I/II trials; these agents bring the potential for more-effective and more-specific anti-complement therapies in disease.
-
Other agents, both biologic and small molecule, are in Phase II or III trials for both rare and common diseases — administration routes include localized (for example, intravitreal) and systemic routes.
-
There is an urgent need to develop biomarkers and imaging methods that enable monitoring of the effects and efficacy of anti-complement agents.
Abstract
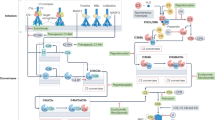
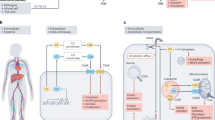
The complement system is a key innate immune defence against infection and an important driver of inflammation; however, these very properties can also cause harm. Inappropriate or uncontrolled activation of complement can cause local and/or systemic inflammation, tissue damage and disease. Complement provides numerous options for drug development as it is a proteolytic cascade that involves nine specific proteases, unique multimolecular activation and lytic complexes, an arsenal of natural inhibitors, and numerous receptors that bind to activation fragments. Drug design is facilitated by the increasingly detailed structural understanding of the molecules involved in the complement system. Only two anti-complement drugs are currently on the market, but many more are being developed for diseases that include infectious, inflammatory, degenerative, traumatic and neoplastic disorders. In this Review, we describe the history, current landscape and future directions for anti-complement therapies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Morgan, B. P. Fundamental Immunology 7th edn (ed. Paul, W. E.) 850–862 (Lippincott Williams & Wilkins, 2012).
Kim, D. D. & Song, W. C. Membrane complement regulatory proteins. Clin. Immunol. 118, 127–136 (2006).
Grumach, A. S. & Kirschfink, M. Are complement deficiencies really rare? Overview on prevalence, clinical importance and modern diagnostic approach. Mol. Immunol. 61, 110–117 (2014).
de Cordoba, S. R., Tortajada, A., Harris, C. L. & Morgan, B. P. Complement dysregulation and disease: from genes and proteins to diagnostics and drugs. Immunobiology 217, 1034–1046 (2012).
Barbour, T. D., Pickering, M. C. & Cook, T. H. Dense deposit disease and C3 glomerulopathy. Semin. Nephrol. 33, 493–507 (2013). A comprehensive and current review of the roles of complement in renal disease.
Risitano, A. M. Paroxysmal nocturnal hemoglobinuria and other complement-mediated hematological disorders. Immunobiology 217, 1080–1087 (2012).
Brodsky, R. A. Paroxysmal nocturnal hemoglobinuria. Blood 124, 2804–2811 (2014).
Linton, S. M. & Morgan, B. P. Complement activation and inhibition in experimental models of arthritis. Mol. Immunol. 36, 905–914 (1999).
Gehrs, K. M., Anderson, D. H., Johnson, L. V. & Hageman, G. S. Age-related macular degeneration — emerging pathogenetic and therapeutic concepts. Ann. Med. 38, 450–471 (2006).
Troutbeck, R., Al Qureshi, S. & Guymer, R. H. Therapeutic targeting of the complement system in age-related macular degeneration: a review. Clin. Experiment Ophthalmol. 40, 18–26 (2012).
Schifferli, J. A. Complement and immune complexes. Res. Immunol. 147, 109–110 (1996).
Truedsson, L., Bengtsson, A. A. & Sturfelt, G. Complement deficiencies and systemic lupus erythematosus. Autoimmunity 40, 560–566 (2007).
Pepys, M. B. Role of complement in induction of antibody production in vivo. Effect of cobra factor and other C3 reactive agents on thymus-dependent and thymus-independent antibody responses. J. Exp. Med. 140, 126–145 (1974).
Carroll, M. C. & Isenman, D. E. Regulation of humoral immunity by complement. Immunity 37, 199–207 (2012).
Clarke, E. V. & Tenner, A. J. Complement modulation of T cell immune responses during homeostasis and disease. J. Leukoc. Biol. 96, 745–756 (2014).
Ricklin, D., Hajishengallis, G., Yang, K. & Lambris, J. D. Complement — a key system for immune surveillance and homeostasis. Nat. Immunol. 11, 785–797 (2010). An excellent review of the important activities of complement.
Lewis, M. J. & Botto, M. Complement deficiencies in humans and animals: links to autoimmunity. Autoimmunity 39, 367–378 (2006).
Skattum, L., van Deuren, M., van der Poll, T. & Truedsson, L. Complement deficiency states and associated infections. Mol. Immunol. 48, 1643–1655 (2011).
Chi, Z. L., Yoshida, T., Lambris, J. D. & Iwata, T. Suppression of drusen formation by compstatin, a peptide inhibitor of complement C3 activation, on cynomolgus monkey with early-onset macular degeneration. Adv. Exp. Med. Biol. 703, 127–135 (2010).
Do, D. V. et al. A phase Ia dose-escalation study of the anti-factor D monoclonal antibody fragment FCFD4514S in patients with geographic atrophy. Retina 34, 313–320 (2014).
He, C., Imai, M., Song, H., Quigg, R. J. & Tomlinson, S. Complement inhibitors targeted to the proximal tubule prevent injury in experimental nephrotic syndrome and demonstrate a key role for C5b-9. J. Immunol. 174, 5750–5757 (2005).
Risitano, A. M. et al. The complement receptor 2/factor H fusion protein TT30 protects paroxysmal nocturnal hemoglobinuria erythrocytes from complement-mediated hemolysis and C3 fragment. Blood 119, 6307–6316 (2012). A clear description of a novel targeted complement inhibitor with potential for improved therapy.
Swaak, A. J. et al. Determination of the half-life of C3 in patients and its relation to the presence of C3 breakdown products and/or circulating immune complexes. Rheumatol. Int. 2, 161–166 (1982).
Rother, R. P., Rollins, S. A., Mojcik, C. F., Brodsky, R. A. & Bell, L. Discovery and development of the complement inhibitor Eculizumab for the treatment of paroxysmal nocturnal hemoglobinuria. Nat. Biotech. 25, 1256–1264 (2007). A useful overview of the developmental history of eculizumab.
Zuber, J. et al. Use of Eculizumab for atypical haemolytic uraemic syndrome and C3 glomerulopathies. Nat. Rev. Nephrol. 8, 643–657 (2012).
Ternant, D. et al. Pharmacokinetics and concentration-effect relationship of adalimumab in rheumatoid arthritis. Br. J. Clin. Pharmacol. 79, 286–297. (2015).
Sissons, J. G., Liebowitch, J., Amos, N. & Peters, D. K. Metabolism of the fifth component of complement, and its relation to metabolism of the third component, in patients with complement activation. J. Clin. Invest. 59, 704–715 (1977).
Noris, M. et al. Dynamics of complement activation in aHUS and how to monitor eculizumab therapy. Blood 124, 1715–1726 (2014).
Katschke, K. J. Jr et al. Structural and functional analysis of a C3b specific antibody that selectively inhibits the alternative pathway of complement. J. Biol. Chem. 284, 10473–10479 (2009).
Paixão-Cavalcante, D. et al. A humanized antibody that regulates the alternative pathway convertase: potential for therapy of renal disease associated with nephritic factors. J. Immunol. 192, 4844–4851 (2014).
Müller-Eberhard, H. J. Complement. Annu. Rev. Biochem. 44, 697–724 (1975).
Pascual, M. et al. Metabolism of complement factor D in renal failure. Kidney Int. 34, 529–536 (1988).
Loyet, K. M. et al. Complement inhibition in cynomolgus monkeys by anti-factor D antigen-binding fragment for the treatment of an advanced form of dry age-related macular degeneration. J. Pharmacol. Exp. Ther. 351, 527–537 (2014).
Kang, J. S., Deluca, P. P. & Lee, K. C. Emerging PEGylated drugs. Expert Opin. Emerg. Drugs 14, 363–380 (2009).
Jazayeri, J. A. & Carroll, G. J. Fc based cytokines: prospects for engineering superior therapeutics. BioDrugs 22, 11–26 (2008).
Zimmerman, J. L., Dellinger, R. P., Straube, R. C. & Levin, J. L. Phase I trial of the recombinant soluble complement receptor 1 in acute lung injury and acute respiratory distress syndrome. Crit. Care Med. 28, 3149–3154 (2000). An early clinical trial of the first recombinant complement regulator.
Lazar, H. L. et al. Soluble human complement receptor 1 limits ischemic damage in cardiac surgery patients at high risk requiring cardiopulmonary bypass. Circulation 110 (11 Suppl. 1), II274–II279 (2004).
Sacks, S. H. & Zhou, W. The role of complement in the early immune response to transplantation. Nat. Rev. Immunol. 12, 431–442 (2012).
Schwertz, H. et al. Serine protease inhibitor nafamostat given before reperfusion reduces inflammatory myocardial injury by complement and neutrophil inhibition. Cardiovasc. Pharmacol. 52, 151–160 (2008).
Gál, P., Dobó, J., Beinrohr, L., Pál, G. & Závodszky, P. Inhibition of the serine proteases of the complement system. Adv. Exp. Med. Biol. 735, 23–40 (2013).
Shi, J. et al. TNT003, an inhibitor of the serine protease C1s, prevents complement activation induced by cold agglutinins. Blood 123, 4015–4022 (2014). A study targeting classical pathway activation as a potential therapy for cold agglutinin disease.
Watkins, A. M. & Arora, P. S. Structure-based inhibition of protein–protein interactions. Eur. J. Med. Chem. 94, 480–488 (2014).
Ruiz-Gómez, G. et al. Structure–activity relationships for substrate-based inhibitors of human complement factor B. J. Med. Chem. 52, 6042–6052 (2009).
Forneris, F. et al. Structures of C3b in complex with factors B and D give insight into complement convertase formation. Science 330, 1816–1820 (2010).
Kocsis, A. et al. Selective inhibition of the lectin pathway of complement with phage display selected peptides against mannose-binding lectin-associated serine protease (MASP)-1 and -2: significant contribution of MASP 1 to lectin pathway activation. J. Immunol. 185, 4169–4178 (2010).
Woodruff, T. M. et al. Species dependence for binding of small molecule agonist and antagonists to the C5a receptor on polymorphonuclear leukocytes. Inflammation 25, 171–177 (2001).
Ricklin, D. & Lambris, J. D. Compstatin: a complement inhibitor on its way to clinical application. Adv. Exp. Med. Biol. 632, 273–292 (2008).
Jacobson, A. C. & Weis, J. H. Comparative functional evolution of human and mouse CR1 and CR2. J. Immunol. 181, 2953–2959 (2008).
Holers, V. M., Kinoshita, T. & Molina, H. The evolution of mouse and human complement C3 binding proteins: divergence of form but conservation of function. Immunol. Today 13, 231–236 (1992).
Bhatia, S. N. & Ingber, D. E. Microfluidic organs-on-chips. Nat. Biotech. 32, 760–772 (2014).
Roos, A., Ramwadhdoebé, T. H., Nauta, A. J., Hack, C. E. & Daha, M. R. Therapeutic inhibition of the early phase of complement activation. Immunobiology 205, 595–609 (2002).
Kjaer, T. R., Thiel, S. & Andersen, G. R. Toward a structure-based comprehension of the lectin pathway of complement. Mol. Imm. 56, 413–422 (2013).
Bertini, R. et al. Noncompetitive allosteric inhibitors of the inflammatory chemokine receptors CXCR1 and CXCR2: prevention of reperfusion injury. Proc. Natl Acad. Sci. USA 101, 11791–11796 (2004).
Cavalieri, B. et al. Neutrophil recruitment in the reperfused-injured rat liver was effectively attenuated by repertaxin, a novel allosteric noncompetitive inhibitor of CXCL8 receptors: a therapeutic approach for the treatment of post-ischemic hepatic syndromes. Int. J. Immunopathol. Pharmacol. 18, 475–486 (2005).
Phuan, P. W. et al. C1q targeted monoclonal antibody prevents complement-dependent cytotoxicity and neuropathology in in vitro and mouse models of neuromyelitis optica. Acta Neuropathol. 125, 829–840 (2013).
Roos, A. et al. Specific inhibition of the classical complement pathway by C1q binding peptides. J. Immunol. 167, 7052–7059 (2001).
Mauriello, C. T. et al. A novel peptide inhibitor of classical and lectin complement activation including ABO incompatibility. Mol. Immunol. 53, 132–139 (2013).
Sharp, J. A., Whitley, P. H., Cunnion, K. M. & Krishna, N. K. Peptide inhibitor of complement C1, a novel suppressor of classical pathway activation: mechanistic studies and clinical potential. Front. Immunol. 5, 406 (2014).
Gadek, J. E. et al. Replacement therapy in hereditary angioedema: successful treatment of acute episodes of angioedema with partly purified C1 inhibitor. N. Engl. J. Med. 302, 542–546 (1980). A classic paper demonstrating the lifesaving efficacy of C1INH in acute angioedema.
Zanichelli, A., Mansi, M., Periti, G. & Cicardi, M. Therapeutic management of hereditary angioedema due to C1 inhibitor deficiency. Expert Rev. Clin. Immunol. 9, 477–488 (2013).
Rosen, F. S., Alper, C. A., Pensky, J., Klemperer, M. R. & Donaldson, V. H. Genetically determined heterogeneity of the C1 esterase inhibitor in patients with hereditary angioneurotic edema. J. Clin. Invest. 50, 2143–2149 (1971).
Craig, T. J. et al. C1 esterase inhibitor concentrate in 1085 hereditary angioedema attacks — final results of the I.M.P.A.C.T.2 study. Allergy 66, 1604–1611 (2011).
Bork, K. & Witzke, G. Long-term prophylaxis with C1 inhibitor (C1 INH) concentrate in patients with recurrent angioedema caused by hereditary and acquired C1 inhibitor deficiency. J. Allergy Clin. Immunol. 83, 677–682 (1989).
Bernstein, J. A. et al. Escalating doses of C1 esterase inhibitor (CINRYZE) for prophylaxis in patients with hereditary angioedema. J. Allergy Clin. Immunol. Pract. 2, 77–84 (2014).
Bork, K. An evidence based therapeutic approach to hereditary and acquired angioedema. Curr. Opin. Allergy Clin. Immunol. 14, 354–362 (2014).
Caliezi, C. et al. C1 inhibitor in patients with severe sepsis and septic shock: beneficial effect on renal dysfunction. Crit. Care Med. 30, 1722–1728 (2002).
Igonin, A. A. et al. C1 esterase inhibitor infusion increases survival rates for patients with sepsis. Crit. Care Med. 40, 770–777 (2012).
Lu, F., Fernandes, S. M. & Davis, A. E. 3rd The effect of C1 inhibitor on myocardial ischemia and reperfusion injury. Cardiovasc. Pathol. 22, 75–80 (2013).
Lu, F. et al. The effect of C1 inhibitor on intestinal ischemia and reperfusion injury. Am. J. Physiol. Gastrointest. Liver Physiol. 295, G1042–G1049 (2008).
Subasinghe, N. L. et al. Design and synthesis of biphenylsulphonyl thiophene carbxamide inhibitors of the complement component C1s. Bioorg. Med. Chem. Lett. 22, 5303–5307 (2012).
Coppens, M., Eikelboom, J. W., Gustafsson, D., Weitz, J. I. & Hirsh, J. Translational success stories: development of direct thrombin inhibitors. Circ. Res. 111, 920–929 (2012).
Héja, D. et al. Monospecific inhibitors show that both mannan-binding lectin-associated serine protease 1 (MASP 1) and -2 are essential for lectin pathway activation and reveal structural plasticity of MASP 2. J. Biol. Chem. 287, 20290–20300 (2012).
Carroll, S. & Georgiou, G. Antibody-mediates inhibition of human C1s and the classical complement pathway. Immunobiology 218, 1041–1048 (2013).
Nydegger, U. E., Fearon, D. T. & Austen, K. F. The modulation of the alternative pathway of complement in C2 deficient human serum by changes in concentration of the component and control proteins. J. Immunol. 120, 1404–1408 (1978).
Heinen, S. et al. Hemolytic uremic syndrome: a factor H mutation (E1172Stop) causes defective complement control at the surface of endothelial cells. J. Am. Soc. Nephrol. 18, 506–514 (2007). This is among the first studies to describe an association of a C-terminal mutation in FH with aHUS.
Fakhouri, F. et al. Treatment with human complement factor H rapidly reverses renal complement deposition in factor H deficient mice. Kidney Int. 78, 279–286 (2010).
Kim, S. J. et al. Intravitreal human complement factor H in a rat model of laser-induced choroidal neovascularisation. Br. J. Ophthalmol. 97, 367–370 (2013).
Khandhadia, S. et al. Age-related macular degeneration and modification of systemic complement factor H production through liver transplantation. Ophthalmology 120, 1612–1618 (2013).
Schmidt, C. Q. et al. Rational engineering of a minimized immune inhibitor with unique triple-targeting properties. J. Immunol. 190, 5712–5721 (2013). Interesting account of the design, production and testing of mini FH.
Hebecker, M. et al. An engineered construct combining complement regulatory and surface-recognition domains represents a minimal-size functional factor H. J. Immunol. 191, 912–921 (2013).
Trapp, R. G., Fletcher, M., Forristal, J. & West, C. D. C4 binding protein deficiency in a patient with atypical Behçet's disease. J. Rheumatol. 14, 135–138 (1987).
Blom, A. M., Nandakumar, K. S. & Holmdahl, R. C4b binding protein (C4BP) inhibits development of experimental arthritis in mice. Ann. Rheum. Dis. 68, 136–142 (2009).
Grumach, A. S., Leitão, M. F., Arruk, V. G., Kirschfink, M. & Condino-Neto, A. Recurrent infections in partial complement factor I deficiency: evaluation of three generations of a Brazilian family. Clin. Exp. Immunol. 143, 297–304 (2006).
Møller Rasmussen, J. et al. Three cases of factor I deficiency: the effect of treatment with plasma. Clin. Exp. Immunol. 74, 131–136 (1988).
Lay, E. et al. Complotype affects the extent of down-regulation by Factor I of the C3b feedback cycle in vitro. Clin. Exp. Immunol. 181, 314–322 (2014).
Lesher, A. M., Nilsson, B. & Song, W. C. Properdin in complement activation and tissue injury. Mol. Immunol. 56, 191–198 (2013).
Ali, Y. M. et al. Low-dose recombinant properdin provides substantial protection against Streptococcus pneumoniae and Neisseria meningitidis infection. Proc. Natl Acad. Sci. USA 111, 5301–5306 (2014).
Weisman, H. F. et al. Soluble human complement receptor type 1: in vivo inhibitor of complement suppressing post-ischemic myocardial inflammation and necrosis. Science. 249, 146–151 (1990). Landmark paper describing the engineering of an effective fluid-phase regulator of complement.
Schmid, R. A., Hillinger, S., Hamacher, J. & Stammberger, U. TP20 is superior to TP10 in reducing ischemia/reperfusion injury in rat lung grafts. Transplant. Proc. 33, 948–949 (2001).
Linton, S. M. et al. Therapeutic efficacy of a novel membrane-targeted complement regulator in antigen-induced arthritis in the rat. Arthritis Rheum. 43, 2590–2597 (2000).
Banz, Y. et al. Attenuation of myocardial reperfusion injury in pigs by Mirococept, a membrane-targeted complement inhibitor derived from human CR1. Cardiovasc. Res. 76, 482–493 (2007). Demonstration that membrane targeting of a complement inhibitor increases efficacy.
Sprong, T. et al. Deficient alternative complement pathway activation due to factor D deficiency by 2 novel mutations in the complement factor D gene in a family with meningococcal infections. Blood 107, 4865–4870 (2006).
Katschke, K. J. Jr et al. Inhibiting alternative pathway complement activation by targeting the factor D exosite. J. Biol. Chem. 287, 12886–12892 (2012).
Cole, L. B., Kilpatrick, J. M., Chu, N. & Babu, Y. S. Structure of 3,4 dichloro isocoumarin-inhibited factor D. Acta Crystallogr. D Biol. Crystallogr. 54, 711–717 (1998).
Sukuru, S. C. et al. A lead discovery strategy driven by a comprehensive analysis of proteases in the peptide substrate space. Protein Sci. 19, 2096–2109 (2010).
Abdel-Magid, A. F. Inhibitors of factor D may provide a treatment for age-related macular degeneration. ACS Med. Chem. Lett. 5, 106–107 (2014).
Hu, X. et al. Therapeutic inhibition of the alternative complement pathway attenuates chronic EAE. Mol. Immunol. 54, 302–308 (2013).
Grossman, T. R. et al. Inhibition of the alternative complement pathway by antisense oligonucleotides targeting complement factor B improves lupus nephritis in mice. Immunobiology http://dx.doi.org/10.1016/j.imbio.2015.08.001 (2015).
Oh, S., Kweon, M. H., Rhee, K. H., Ho Lee, K. & Sung, H. C. Inhibition of complement by recombinant Sh-CRIT-ed1 analogues. Immunology 110, 73–79 (2003).
Maillard, J. L. & Zarco, R. M. Decomplementation par un facteur extrait du venin de cobra. Ann. Inst. Pasteur 114, 756–780 (in French) (1968).
Wright, M. C., Nelson, R. A. & Finch, S. C. The effects of a cobra venom factor and ethyl palmitate on the prolongation of survival of heterologous erythrocytes. Yale J. Biol. Med. 43, 173–176 (1970).
Till, G. O., Johnson, K. J., Kunkel, R. & Ward, P. A. Intravascular activation of complement and acute lung injury. Dependency on neutrophils and toxic oxygen metabolites. J. Clin. Invest. 69, 1126–1135 (1982).
Vogel, C. W., Finnegan, P. W. & Fritzinger, D. C. Humanized cobra venom factor: activity and therapeutic efficacy in preclinical disease models. Mol. Immunol. 61, 191–203 (2014).
Kinnunen, K. & Yla-Herttuala, S. Gene therapy in age-related macular degeneration and hereditary macular disorders. Front. Biosci. 4, 2546–2557 (2012).
Fluiter, K., Opperhuizen, A. L., Morgan, B. P., Baas, F. & Ramaglia, V. Inhibition of the membrane attack complex of the complement system reduces secondary neuroaxonal loss and promotes neurologic recovery after traumatic brain injury in mice. J. Immunol. 192, 2339–2348 (2014).
Klos, A. et al. The role of the anaphylatoxins in health and disease. Mol. Immunol. 46, 2753–2766 (2009).
Engelke, C. et al. Distinct roles of the anaphylatoxins C3a and C5a in dendritic cell-mediated allergic asthma. J. Immunol. 193, 5387–5401 (2014).
Solari, R., Pease, J. E. & Begg, M. Chemokine receptors as therapeutic targets: why aren't there more drugs? Eur. J. Pharmacol. 746, 363–367 (2014).
Woodruff, T. M. et al. Therapeutic activity of C5a receptor antagonists in a rat model of neurodegeneration. FASEB J. 20, 1407–1417 (2006).
Vergunst, C. E. et al. Blocking the receptor for C5a in patients with rheumatoid arthritis does not reduce synovial inflammation. Rheumatology (Oxford) 46, 1773–1778 (2007).
Wu, M. C. et al. The receptor for complement component C3a mediates protection from intestinal ischemia-reperfusion injuries by inhibiting neutrophil mobilization. Proc. Natl Acad. Sci. USA 110, 9439–9444 (2013).
Hoehlig, K. et al. A novel C5a neutralizing mirror-image (l-)aptamer prevents organ failure and improves survival in experimental sepsis. Mol. Ther. 21, 2236–2246 (2013).
Khan, M. A. et al. Targeting complement component 5a promotes vascular integrity and limits airway remodeling. Proc. Natl Acad. Sci. USA 110, 6061–6066 (2013).
Singer, M. Advances in the management of macular degeneration. F1000Prime Rep. 6, 29 (2014).
Nunn, M. A. et al. Complement inhibitor of C5 activation from the soft tick Ornithodoros moubata. J. Immunol. 174, 2084–2091 (2005).
Fredslund, F. et al. Structure of and influence of a tick complement inhibitor on human complement component 5. Nat. Immunol. 9, 753–760 (2008).
Hepburn, N. J. et al. In vivo characterization and therapeutic efficacy of a C5 specific inhibitor from the soft tick Ornithodoros moubata. J. Biol. Chem. 282, 8292–8299 (2007).
Vakeva, A. et al. High-density lipoproteins can act as carriers of glycophosphoinositol lipid anchored CD59 in human plasma. Immunology 82, 28–33 (1994).
Fraser, D. A. et al. Generation of a recombinant, membrane-targeted form of the complement regulator CD59; activity in vitro and in vivo. J. Biol. Chem. 278, 48921–48927 (2003).
Hill, A. et al. Protection of erythrocytes from human complement-mediated lysis by membrane-targeted recombinant soluble CD59: a new approach to PNH therapy. Blood 107, 2131–2137 (2006). Demonstration that restoration of CD59 protects PNH erythrocytes from lysis.
Lee, M., Narayanan, S., McGeer, E. G. & McGeer, P. L. Aurin tricarboxylic acid protects against red blood cell hemolysis in patients with paroxysmal nocturnal hemoglobinemia. PLoS ONE 29, e87316 (2014).
Harris, C. L. et al. Generation of anti-complement 'prodrugs': cleavable reagents for specific delivery of complement regulators to disease sites. J. Biol. Chem. 278, 36068–36076 (2003).
Bagshawe, K. D., Sharma, S. K. & Begent, R. H. Antibody-directed enzyme prodrug therapy (ADEPT) for cancer. Expert Opin. Biol. Ther. 4, 1777–1789 (2004).
Atkinson, C. et al. Targeted complement inhibition by C3d recognition ameliorates tissue injury without apparent increase in susceptibility to infection. J. Clin. Invest. 115, 2444–2453 (2005). First demonstration that complement activation products can be used to localize therapy.
Atkinson, C. et al. Targeted complement inhibitors protect against posttransplant cardiac ischemia and reperfusion injury and reveal an important role for the alternative pathway of complement activation. J. Immunol. 185, 7007–7013 (2010).
Fridkis-Hareli, M. et al. Design and development of TT30, a novel C3d targeted C3/C5 convertase inhibitor for treatment of human complement alternative pathway-mediated diseases. Blood 118, 4705–4713 (2011).
Thurman, J. M. et al. Detection of complement activation using monoclonal antibodies against C3d. J. Clin. Invest. 123, 2218–2230 (2013). Describe novel reagents that enable imaging of complement activation in vivo.
Thurman, J. M. & Rohrer, B. Noninvasive detection of complement activation through radiologic imaging. Adv. Exp. Med. Biol. 735, 271–282 (2013).
Harris, C. L., Heurich, M., Cordoba, S. R. & Morgan, B. P. The complotype: dictating risk for inflammation and infection. Trends Immunol. 33, 513–521 (2012). Describes the development of the concept of the complotype as predictor of disease risk.
Heurich, M. et al. Common polymorphisms in C3, factor B, and factor H collaborate to determine systemic complement activity and disease risk. Proc. Natl Acad. Sci. USA 108, 8761–8766 (2011).
Hageman, G. S. et al. A common haplotype in the complement regulatory gene factor H (HF1/CFH) predisposes individuals to age-related macular degeneration. Proc. Natl Acad. Sci. USA 102, 7227–7232 (2005). Identification of the strong association of FH polymorphism with AMD.
Sofat, R. et al. Complement factor H genetic variant and age-related macular degeneration: effect size, modifiers and relationship to disease subtype. Int. J. Epidemiol. 41, 250–262 (2012).
Gold, B. et al. Variation in factor B (BF) and complement component 2 (C2) genes is associated with age-related macular degeneration. Nat. Genet. 38, 458–462 (2006).
Yates, Y. R. et al. Complement C3 variant and the risk of age-related macular degeneration. N. Engl. J. Med. 357, 19–27 (2007).
Jager, R. D., Mieler, W. F. & Miller, J. W. Age-related macular degeneration. N. Engl. J. Med. 358, 2606–2617 (2008).
Kavanagh, D., Goodship, T. H. & Richards, A. Atypical hemolytic uremic syndrome. Semin. Nephrol. 33, 508–530 (2013).
Manuelian, T. et al. Mutations in factor H reduce binding affinity to C3b and heparin and surface attachment to endothelial cells in hemolytic uremic syndrome. J. Clin. Invest. 111, 1181–1190 (2003).
Moore, I. et al. Association of factor H autoantibodies with deletions of CFHR1, CFHR3, CFHR4, and with mutations in CFH, CFI, CD46, and C3 in patients with atypical hemolytic uremic syndrome. Blood. 115, 379–387 (2010). Important description of link between anti FH autoantibodies, deletions in FH-related (FHR) proteins and aHUS.
Frei, Y., Lambris, J. D. & Stockinger, B. Generation of a monoclonal antibody to mouse C5; application in an ELISA assay for detection of anti C5 antibodies. Mol. Cell. Probes 1, 141–149 (1987).
Wang, Y., Rollins, S. A., Madri, J. A. & Matis, L. A. Anti C5 monoclonal antibody therapy prevents collagen-induced arthritis and ameliorates established disease. Proc. Natl Acad. Sci. USA 92, 8955–8959 (1995).
Thomas, T. C. et al. Inhibition of complement activity by humanized anti C5 antibody and single-chain Fv. Mol. Immunol. 33, 1389–1140 (1996).
Adis International Limited Eculizumab: 5G1.1, h5G1.1, long-acting anti C5 monoclonal antibody 5G1 1, long-acting anti C5 monoclonal antibody 5G1.1. Drugs R. D. 8, 61–68 (2007).
Kaplan, M. Eculizumab (Alexion). Curr. Opin. Investig. Drugs 3, 1017–1023 (2002).
Hillmen, P. et al. Effect of eculizumab on hemolysis and transfusion requirements in patients with paroxysmal nocturnal hemoglobinuria. N. Engl. J. Med. 350, 552–559 (2004). First description of profound effect of eculizumab on PNH outcomes.
Brodsky, R. A. et al. Multicenter phase 3 study of the complement inhibitor eculizumab for the treatment of patients with paroxysmal nocturnal hemoglobinuria. Blood 111, 1840–1847 (2008).
Hillmen, P. et al. Long-term safety and efficacy of sustained eculizumab treatment in patients with paroxysmal nocturnal haemoglobinuria. Br. J. Haematol. 162, 62–73 (2013).
Legendre, C. M. et al. Terminal complement inhibitor eculizumab in atypical hemolytic-uremic syndrome. N. Engl. J. Med. 368, 2169–2181 (2013). Demonstration of efficacy of eculizumab in therapy of aHUS.
Ricklin, D. et al. A molecular insight into complement inhibition by the Staphylococcal complement inhibitor family. J. Immunol. 183, 2565–2574 (2009).
Parker, C. J., Kar, S. & Kirkpatrick, P. Eculizumab. Nat. Rev. Drug Disc. 6, 515–516 (2007).
Nishimura, J. et al. Genetic variants in C5 and poor response to Eculizumab. N. Engl. J. Med. 370, 632–639 (2014).
Mullard, A. Protein–protein interaction inhibitors get into the groove. Nat. Rev. Drug Disc. 11, 173–175 (2012).
Wu, J. et al. Structure of complement fragment C3b–factor H and implications for host protection by complement regulators. Nat. Immunol. 10, 728–733 (2009).
Sahu, A., Kay, B. K. &, Lambris, J. D. Inhibition of complement by a C3 binding peptide isolated from a phage-displayed random peptide library. J. Immunol. 157, 884–891 (1996).
Qu, H. et al. New analogs of the clinical complement inhibitor compstatin with subnanomolar affinity and enhanced pharmacokinetic properties. Immunobiology 218, 496–505 (2013).
Mastellos, D. C. et al. Compstatin: a C3 targeted complement inhibitor reaching its prime for bedside intervention. Eur. J. Clin. Invest. 45, 423–440 (2015).
Leung, E. & Landa, G. Update on current and future novel therapies for dry age-related macular degeneration. Expert Rev. Clin. Pharmacol. 6, 565–579 (2013).
Lachmann, P. J. The amplification loop of the complement pathways. Adv. Immunol. 104, 115–149 (2009).
Martinez-Saguer, I. et al. Pharmacokinetics of plasma-derived C1 esterase inhibitor after subcutaneous versus intravenous administration in subjects with mild or moderate hereditary angioedema: the PASSION study. Transfusion 54, 1552–1561 (2014).
van Doorn, M. B. et al. A phase I study of recombinant human C1 inhibitor in asymptomatic patients with hereditary angioedema. J. Allergy Clin. Immunol. 116, 876–883 (2005).
Riedl, M. A. et al. Recombinant human C1 esterase inhibitor relieves symptoms of hereditary angioedema attacks: phase 3, randomized, placebo-controlled trial. Ann. Allergy Asthma Immunol. 112, 163–169 (2014).
Riedl, M. et al. Spectrum of complement-mediated thrombotic microangiopathies: pathogenetic insights identifying novel treatment approaches. Semin. Thromb. Hemost. 40, 444–464 (2014).
Pittock, S. J. et al. Eculizumab in AQP4 IgG-positive relapsing neuromyelitis optica spectrum disorders: an open-label pilot study. Lancet Neurol. 12, 554–562 (2013).
Ingram, G., Hakobyan, S., Robertson, N. P. & Morgan, B. P. Complement in multiple sclerosis: its role in disease and potential as a biomarker. Clin. Exp. Immunol. 155, 128–139 (2009).
Ingram, G. et al. Systemic complement profiling in multiple sclerosis as a biomarker of disease state. Mult. Scler. 18, 1401–1411 (2012).
Willison, H. J. et al. The role of complement and complement regulators in mediating motor nerve terminal injury in murine models of Guillain–Barré syndrome. J. Neuroimmunol. 201–202, 172–182 (2008).
Walgaard, C., Jacobs, B. C. & van Doorn, P. A. Emerging drugs for Guillain–Barré syndrome. Expert Opin. Emerg. Drugs 16, 105–120 (2011).
Morgan, B. P. et al. The membrane attack pathway of complement drives pathology in passively induced experimental autoimmune myasthenia gravis in mice. Clin. Exp. Immunol. 146, 294–302 (2006).
Howard, J. F. Jr et al. A randomized, double-blind, placebo-controlled phase II study of eculizumab in patients with refractory generalized myasthenia gravis. Muscle Nerve 48, 76–84 (2013).
Coppo, R. et al. Dramatic effects of eculizumab in a child with diffuse proliferative lupus nephritis resistant to conventional therapy. Pediatr. Nephrol. 30, 167–172 (2015).
Kiryluk, K. & Novak, J. The genetics and immunobiology of IgA nephropathy. J. Clin. Invest. 124, 2325–2332 (2014).
Rosenblad, T. et al. Eculizumab treatment for rescue of renal function in IgA nephropathy. Pediatr. Nephrol. 29, 2225–2228 (2014).
Okroj, M., Heinegård, D., Holmdahl, R. & Blom, A. M. Rheumatoid arthritis and the complement system. Ann. Med. 39, 517–530 (2007).
Sivaram, G. et al. Association of genetic variants of mannan-binding (MBL) lectin 2 gene, MBL levels and function in ulcerative colitis and Crohn's disease. Innate Immun. 17, 526–531 (2011).
Elvington, M., Schepp-Berglind, J. & Tomlinson, S. Regulation of the alternative pathway of complement modulates injury and immunity in a chronic model of DSS-induced colitis. Clin Exp Immunol. 179, 500–508 (2014).
Gupta, N. & Wang, E. S. Long-term response of refractory primary cold agglutinin disease to eculizumab therapy. Ann. Hematol. 93, 343–344 (2014).
Lessey, E., Li, N., Diaz, L. & Liu, Z. Complement and cutaneous autoimmune blistering diseases. Immunol. Res. 41, 223–232 (2008).
Lim, W. Complement and the antiphospholipid syndrome. Curr. Opin. Hematol. 18, 361–365 (2011).
Lonze, B. E. et al. Eculizumab prevents recurrent antiphospholipid antibody syndrome and enables successful renal transplantation. Am. J. Transplant. 14, 459–465 (2014).
Yang, M. M. et al. Association of C2 and CFB polymorphisms with anterior uveitis. Invest. Ophthalmol. Vis. Sci. 53, 4969–4974 (2012).
Kallenberg, C. G., Stegeman, C. A., Abdulahad, W. H. & Heeringa, P. Pathogenesis of ANCA-associated vasculitis: new possibilities for intervention. Am. J. Kidney Dis. 62, 1176–1187 (2013).
Lewis, R. D., Jackson, C. L., Morgan, B. P. & Hughes, T. R. The membrane attack complex of complement drives the progression of atherosclerosis in apolipoprotein E knockout mice. Mol. Immunol. 47, 1098–1105 (2010).
Hertle, E., Stehouwer, C. D. & van Greevenbroek, M. M. The complement system in human cardiometabolic disease. Mol. Immunol. 61, 135–148 (2014).
Li, Y. et al. Label-free quantitative proteomic analysis reveals dysfunction of complement pathway in peripheral blood of schizophrenia patients: evidence for the immune hypothesis of schizophrenia. Mol. Biosyst. 8, 2664–2671 (2012).
Håvik, B. et al. The complement control-related genes CSMD1 and CSMD2 associate to schizophrenia. Biol. Psychiatry 70, 35–42 (2011).
Zhang, X. & Köhl, J. A complex role for complement in allergic asthma. Expert Rev. Clin. Immunol. 6, 269–277 (2010).
Khan, M. A., Nicolls, M. R., Surguladze, B. & Saadoun, I. Complement components as potential therapeutic targets for asthma treatment. Respir. Med. 108, 543–549 (2014).
Marc, M. M. et al. Complement factors C3a, C4a, and C5a in chronic obstructive pulmonary disease and asthma. Am. J. Respir. Cell. Mol. Biol. 31, 216–219 (2004).
Kodama, T. et al. Role of complement in a murine model of peanut-induced anaphylaxis. Immunobiology 218, 844–850 (2013).
Khodoun, M. et al. Peanuts can contribute to anaphylactic shock by activating complement. J. Allergy Clin. Immunol. 123, 342–351 (2009).
Rittirsch, D., Redl, H. & Huber-Lang, M. Role of complement in multiorgan failure. Clin. Dev. Immunol. 2012, 962927 (2012).
Huber-Lang, M. et al. Double blockade of CD14 and complement C5 abolishes the cytokine storm and improves morbidity and survival in polymicrobial sepsis in mice. J. Immunol. 192, 5324–5331 (2014).
Ramos-Summerford, T. N. & Barnum, S. R. Experimental cerebral malaria: the murine model provides crucial insight into the role of complement. Trends Parasitol. 30, 215–216 (2014).
Chimenti, M. S. et al. Imunomodulation in psoriatic arthritis: focus on cellular and molecular pathways. Autoimmun. Rev. 12, 599–606 (2013).
Chimenti, M. S. et al. Complement system in psoriatic arthritis: a useful marker in response prediction and monitoring of anti-TNF treatment. Clin. Exp. Rheumatol. 30, 23–30 (2012).
De Bleecker, J. L., Creus, K. K. & De Paepe, B. Potential therapeutic targets for idiopathic inflammatory myopathies. Drug News Perspect. 19, 549–557 (2006).
Gordon, P. A., Winer, J. B., Hoogendijk, J. E. & Choy, E. H. Immunosuppressant and immunomodulatory treatment for dermatomyositis and polymyositis. Cochrane Database Syst. Rev. 8, CD003643 (2012).
Wang, Q. et al. Identification of a central role for complement in osteoarthritis. Nat. Med. 17, 1674–1679 (2011). A thorough analysis in mouse models implicating complement in degenerative joint disease.
Lepus, C. M. et al. Brief report: carboxypeptidase B serves as a protective mediator in osteoarthritis. Arthritis Rheumatol. 66, 101–106 (2014).
Eikelenboom, P. et al. Innate immunity and the etiology of late-onset Alzheimer's disease. Neurodegener. Dis. 10, 271–273 (2012).
Wyss-Coray, T. & Rogers, J. Inflammation in Alzheimer disease a brief review of the basic science and clinical literature. Cold Spring Harb Perspect. Med. 2, a006346 (2012).
Rosen, A. M. & Stevens, B. The role of the classical complement cascade in synapse loss during development and glaucoma. Adv. Exp. Med. Biol. 703, 75–93 (2010). This paper describes a novel role of complement in synaptic pruning relevant to development and disease.
Flyvbjerg, A. Diabetic angiopathy, the complement system and the tumor necrosis factor superfamily. Nat. Rev. Endocrinol. 6, 94–101 (2010).
Yanai, R., Thanos, A. & Connor, K. M. Complement involvement in neovascular ocular diseases. Adv. Exp. Med. Biol. 946, 161–183 (2012).
Lindberg, S. et al. Soluble form of membrane attack complex independently predicts mortality and cardiovascular events in patients with ST elevation myocardial infarction treated with primary percutaneous coronary intervention. Am. Heart J. 164, 786–792 (2012).
Orsini, F. et al. Targeting mannose-binding lectin confers long-lasting protection with a surprisingly wide therapeutic window in cerebral ischemia. Circulation 126, 1484–1494 (2012).
Heydenreich, N. et al. C1 inhibitor protects from brain ischemia-reperfusion injury by combined antiinflammatory and antithrombotic mechanisms. Stroke 43, 2457–2467 (2012).
Huber-Lang, M., Kovtun, A. & Ignatius, A. The role of complement in trauma and fracture healing. Semin. Immunol. 25, 73–78 (2013).
Gorsuch, W. B., Chrysanthou, E., Schwaeble, W. J. & Stahl, G. L. The complement system in ischemia-reperfusion injuries. Immunobiology 217, 1026–1033 (2012).
DeAngelis, R. A., Reis, E. S., Ricklin, D. & Lambris, J. D. Targeted complement inhibition as a promising strategy for preventing inflammatory complications in hemodialysis. Immunobiology 217, 1097–1105 (2012).
Reis, E. S. et al. Therapeutic C3 inhibitor Cp40 abrogates complement activation induced by modern hemodialysis filters. Immunobiology 220, 476–482 (2014).
Noris, M., Mescia, F. & Remuzzi, G. STEC-HUS, atypical HUS and TTP are all diseases of complement activation. Nat. Rev. Nephrol. 8. 622–633 (2012).
Yehoshua, Z. et al. Systemic complement inhibition with eculizumab for geographic atrophy in age-related macular degeneration: the COMPLETE study. Ophthalmology 121, 693–701 (2014).
Stegall, M. D. et al. Terminal complement inhibition decreases antibody-mediated rejection in sensitized renal transplant recipients. Am. J. Transplant. 11, 2405–2413 (2011).
Acknowledgements
The authors thank K. Miller of GlaxoSmithKline, for her thoughtful review and discussion of the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
B.P.M. has undertaken paid consultancy work and sponsored research for GlaxoSmithKline, Swedish Orphan Biovitrum and Achillion Pharma. C.L.H. is employed by GlaxoSmithKline.
Related links
FURTHER INFORMATION
Glossary
- Tickover
-
The spontaneous low-level complement activation that occurs continuously in the plasma of normal individuals and assists in the rapid response to pathogens.
- Opsonins
-
Proteins and protein fragments that coat pathogens and dead or dying host cells to label them for recognition and elimination by phagocytes. The principle complement opsonins are C3b and iC3b.
- Short consensus repeats
-
Structural units comprising approximately 60 amino acids held in a globular domain by 2 invariant disulfide bonds; they are the key building blocks for the assembly of proteins in the regulators of complement activation (RCA) family — including Factor H (FH) and the FH-related (FHR) proteins, C4b-binding protein (C4BP), complement receptor type 1 (CR1) and CR2 — but are also found in other complement proteins and proteins unrelated to complement.
- Oligosaccharide sialyl Lewis X epitopes
-
Carbohydrate structures that are expressed on cell surfaces and act as ligands for cell adhesion molecules of the selectin family.
Rights and permissions
About this article
Cite this article
Morgan, B., Harris, C. Complement, a target for therapy in inflammatory and degenerative diseases. Nat Rev Drug Discov 14, 857–877 (2015). https://doi.org/10.1038/nrd4657
Published:
Issue date:
DOI: https://doi.org/10.1038/nrd4657
This article is cited by
-
Inhibition of acute complement responses towards bolus-injected nanoparticles using targeted short-circulating regulatory proteins
Nature Nanotechnology (2024)
-
Soluble terminal complement complex blood levels are elevated in schizophrenia
European Archives of Psychiatry and Clinical Neuroscience (2024)
-
Discovering covalent inhibitors of protein–protein interactions from trillions of sulfur(VI) fluoride exchange-modified oligonucleotides
Nature Chemistry (2023)
-
Complement therapeutics are coming of age in rheumatology
Nature Reviews Rheumatology (2023)
-
Bifico relieves irritable bowel syndrome by regulating gut microbiota dysbiosis and inflammatory cytokines
European Journal of Nutrition (2023)