Key Points
-
The generation of protective B-cell responses is limited both in early life and in elderly individuals. Infants are vulnerable to infections soon after the disappearance of IgG antibodies of maternal origin, and elderly individuals have problems in dealing with new pathogens and in responding to pathogens that they have previously overcome.
-
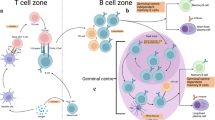
Numerous factors concur to limit plasma-cell differentiation in early life, including decreased strength of signals from the B-cell receptor (BCR) and co-stimulatory molecules, decreased antigen availability through binding of maternal antibodies, delayed follicular dendritic cell (FDC) maturation limiting germinal centre reactions and B-cell competition for simultaneous access to limited germinal centre resources. The duration of IgG responses elicited in early life is also shorter than in mature individuals, probably as a result of exposure to a large load of environmental antigens and the restriction of numerous plasmablasts towards a limited set of plasma-cell survival niches in the bone marrow.
-
By contrast, this combination of factors results in the preferential differentiation of early-life B cells towards memory B cells. This does not imply that the magnitude and persistence of these memory responses are mature, as infant-triggered memory B cells might not be life-long.
-
Elderly individuals also have an inability to raise brisk and sustained responses to new antigens. But at the same time they have increasing titres of organ-specific and organ-non-specific autoantibodies, possibly resulting from their generation as by-products of earlier responses to other antigens. The elderly also have an over-representation of specific classes of antibody from individual B-cell clones.
-
Factors that contribute to the inability to raise sustained antibody responses include an age-related decrease in the production of new B cells from their precursors, a progressive shift from mostly naive B cells to mostly memory cells in later years, limitations of B-cell proliferation and/or retention of immune complexes by FDCs resulting in fewer and smaller germinal centre responses, and suboptimal CD40–CD40L interactions between B and T cells.
-
At both extremes of age, potential strategies towards improved vaccine efficacy include the development of delivery systems and immunomodulators that increase the recruitment and production of naive B cells (for example, higher antigen doses, increased antigen retention, improved B-cell targeting and increased stem cell differentiation) and that induce B-cell activation and/or their differentiation into plasma cells, as well as providing boosters as often as necessary to recall antigen-specific memory B cells.
Abstract
Infants and the elderly share a high vulnerability to infections and therefore have specific immunization requirements. Inducing potent and sustained B-cell responses is as challenging in infants as it is in older subjects. Several mechanisms to explain the decreased B-cell responses at the extremes of age apply to both infants and the elderly. These include intrinsic B-cell limitations as well as numerous microenvironmental factors in lymphoid organs and the bone marrow. This Review describes the mechanisms that shape B-cell responses at the extremes of age and how they could be taken into account to design more effective immunization strategies for these high-risk age groups.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Bryce, J., Boschi-Pinto, C., Shibuya, K. & Black, R. E. WHO estimates of the causes of death in children. Lancet 365, 1147–1152 (2005).
Levy, O. Innate immunity of the newborn: basic mechanisms and clinical correlates. Nature Rev. Immunol. 7, 379–390 (2007).
Zinkernagel, R. M. Maternal antibodies, childhood infections, and autoimmune diseases. N. Engl. J. Med. 345, 1331–1335 (2001).
Yoshikawa, T. T. Epidemiology and unique aspects of aging and infectious diseases. Clin. Infect. Dis. 30, 931–933 (2000).
Yoshikawa, T. T. Perspective: aging and infectious diseases: past, present, and future. J. Infect. Dis. 176, 1053–1057 (1997).
Thompson, W. W. et al. Mortality associated with influenza and respiratory syncytial virus in the United States. JAMA 289, 179–186 (2003).
Gardner, P. & Pabbatireddy, S. Vaccines for women age 50 and older. Emerg. Infect. Dis. 10, 1990–1995 (2004).
Streilein, J. W. Neonatal tolerance: towards an immunogenetic definition of self. Immunol. Rev. 46, 123–146 (1979).
Siegrist, C. Neonatal and early life vaccinology. Vaccine 19, 3331–3346 (2001).
Adkins, B., Leclerc, C. & Marshall-Clarke, S. Neonatal adaptive immunity comes of age. Nature Rev. Immunol. 4, 553–564 (2004).
Hannoun, C., Megas, F. & Piercy, J. Immunogenicity and protective efficacy of influenza vaccination. Virus Res. 103, 133–138 (2004).
Looney, R. J. et al. Hepatitis B immunization of healthy elderly adults: relationship between naive CD4+ T cells and primary immune response and evaluation of GM-CSF as an adjuvant. J. Clin. Immunol. 21, 30–36 (2001).
Pollard, A., Perrett, K. P. & Beverley, P. C. Maintaining protection against invasive bacteria with protein– polysaccharide conjugate vaccines. Nature Rev. Immunol. 13 Feb 2009 (doi: 10.1038/nri2494).
Wright, P. F. et al. Evaluation of a live, cold-passaged, temperature-sensitive, respiratory syncytial virus vaccine candidate in infancy. J. Infect. Dis. 182, 1331–1342 (2000).
Halsey, N. & Galazka, A. The efficacy of DPT and oral poliomyelitis immunization schedules initiated from birth to 12 weeks of age. Bull. World Health Organ. 63, 1151–1169 (1985).
Einhorn, M. S., Weinberg, G. A., Anderson, E. L., Granoff, P. D. & Granoff, D. M. Immunogenicity in infants of Haemophilus influenzae type B polysaccharide in a conjugate vaccine with Neisseria meningitidis outer-membrane protein. Lancet 2, 299–302 (1986).
Booy, R. et al. Immunogenicity of combined diphtheria, tetanus, and pertussis vaccine given at 2, 3, and 4 months versus 3, 5, and 9 months of age. Lancet 339, 507–510 (1992).
Tiru, M., Hallander, H. O., Gustafsson, L., Storsaeter, J. & Olin, P. Diphtheria antitoxin response to DTP vaccines used in Swedish pertussis vaccine trials, persistence and projection for timing of booster. Vaccine 18, 2295–2306 (2000).
Olin, P., Hallander, H. O., Gustafsson, L., Barreto, L. & Podda, A. Measuring protection; a case study of pertussis vaccines — Swedish Trial II: secondary non-randomized comparisons between two schedules of infant vaccination. Dev. Biol. Stand. 95, 211–220 (1998).
Johnson, C. E. et al. Measles vaccine immunogenicity in 6- versus 15-month-old infants born to mothers in the measles vaccine era. Pediatrics 93, 939–944 (1994).
Gans, H. A. et al. Deficiency of the humoral immune response to measles vaccine in infants immunized at age 6 months. JAMA 280, 527–532 (1998). This report shows the slow acquisition of the antibody-response capacity: only 36% of 6-month-old infants have seroprotective neutralizing antibody titres of 120 or higher after measles vaccination, compared with 100% of 9-month-old infants.
Letson, G. W. et al. Effect of maternal antibody on immunogenicity of hepatitis A vaccine in infants. J. Pediatr. 144, 327–332 (2004). This study shows that the slow acquisition of the antibody response capacity also occurs in response to potent non-live vaccines, such as hepatitis A.
Giuliano, M. et al. Antibody responses and persistence in the two years after immunization with two acellular vaccines and one whole-cell vaccine against pertussis. J. Pediatr. 132, 983–988 (1998).
Whittle, H. C. et al. Effect of subclinical infection on maintaining immunity against measles in vaccinated children in West Africa. Lancet 353, 98–102 (1999).
Trotter, C. L., Andrews, N. J., Kaczmarski, E. B., Miller, E. & Ramsay, M. E. Effectiveness of meningococcal serogroup C conjugate vaccine 4 years after introduction. Lancet 364, 365–367 (2004).
Roduit, C. et al. Immunogenicity and protective efficacy of neonatal vaccination against Bordetella pertussis in a murine model: evidence for early control of pertussis. Infect. Immun. 70, 3521–3528 (2002).
Kaur, K., Chowdhury, S., Greenspan, N. S. & Schreiber, J. R. Decreased expression of tumor necrosis factor family receptors involved in humoral immune responses in preterm neonates. Blood 110, 2948–2954 (2007).
Timens, W., Boes, A., Rozeboom-Uiterwijk, T. & Poppema, S. Immaturity of the human splenic marginal zone in infancy. Possible contribution to the deficient infant immune response. J. Immunol. 143, 3200–3206 (1989). This seminal work identifies crucial maturation stages for the development of the marginal zone of the spleen.
Griffioen, A. W., Rijkers, G. T., Janssens-Korpela, P. & Zegers, B. J. Pneumococcal polysaccharides complexed with C3d bind to human B lymphocytes via complement receptor type 2. Infect. Immun. 59, 1839–1845 (1991).
Kanswal, S., Katsenelson, N., Selvapandiyan, A., Bram, R. J. & Akkoyunlu, M. Deficient TACI expression on B lymphocytes of newborn mice leads to defective Ig secretion in response to BAFF or APRIL. J. Immunol. 181, 976–990 (2008).
Siegrist, C. A. Mechanisms by which maternal antibodies influence infant vaccine responses: review of hypotheses and definition of main determinants. Vaccine 21, 3406–3412 (2003).
Johnston, R. B. Jr, Altenburger, K. M., Atkinson, A. W. Jr & Curry, R. H. Complement in the newborn infant. Pediatrics 64, 781–786 (1979).
Pihlgren, M. et al. Influence of complement C3 amount on IgG responses in early life: immunization with C3b-conjugated antigen increases murine neonatal antibody responses. Vaccine 23, 329–335 (2004).
Landers, C. D., Chelvarajan, R. L. & Bondada, S. The role of B cells and accessory cells in the neonatal response to TI-2 antigens. Immunol. Res. 31, 25–36 (2005).
Pihlgren, M. et al. Unresponsiveness to lymphoid-mediated signals at the neonatal follicular dendritic cell precursor level contributes to delayed germinal center induction and limitations of neonatal antibody responses to T-dependent antigens. J. Immunol. 170, 2824–2832 (2003). This work provides a mechanistic explanation for the delayed and limited antibody responses in early life by identifying the slow postnatal maturation of FDCs as a crucial factor that decreases germinal centre responses.
Kruschinski, C., Zidan, M., Debertin, A. S., von Horsten, S. & Pabst, R. Age-dependent development of the splenic marginal zone in human infants is associated with different causes of death. Hum. Pathol. 35, 113–121 (2004).
Blanchard Rohner, G. et al. The magnitude of the antibody and memory B cell responses during priming with a protein–polysaccharide conjugate vaccine in human infants is associated with the persistence of antibody and the intensity of booster response. J. Immunol. 180, 2165–2173 (2008).
Kovarik, J. et al. Adjuvant effects of CpG oligodeoxynucleotides on responses against T-independent type 2 antigens. Immunology 102, 67–76 (2001).
Pihlgren, M. et al. Delayed and deficient establishment of the long-term bone marrow plasma cell pool during early life. Eur. J. Immunol. 31, 939–946 (2001).
Pihlgren, M. et al. Reduced ability of neonatal and early-life bone marrow stromal cells to support plasmablast survival. J. Immunol. 176, 165–172 (2006).
Belnoue, E. et al. APRIL is critical for plasmablast survival in the bone marrow and poorly expressed by early-life bone marrow stromal cells. Blood 111, 2755–2764 (2008). This work identifies a unique pro-survival effect of APRIL on antigen-specific plasmablasts and shows that its limited production by bone marrow stromal cells is associated with the defective establishment of the plasma-cell pool.
Newport, M. J. et al. Genetic regulation of immune responses to vaccines in early life. Genes Immun. 5, 122–129 (2004).
Marchant, A. et al. Predominant influence of environmental determinants on the persistence and avidity maturation of antibody responses to vaccines in infants. J. Infect. Dis. 193, 1598–1605 (2006).
Dong, D. X. et al. Immunization of neonates with trivalent oral poliomyelitis vaccine (Sabin). Bull. World Health Organ. 64, 853–860 (1986).
Pichichero, M. E. et al. Impact of a birth dose of hepatitis B vaccine on the reactogenicity and immunogenicity of diphtheria–tetanus–acellular pertussis–hepatitis B–inactivated poliovirus–Haemophilus influenzae type b combination vaccination. Pediatr. Infect. Dis. J. 21, 854–859 (2002).
Belloni, C. et al. Immunogenicity of a three-component acellular pertussis vaccine administered at birth. Pediatrics 111, 1042–1045 (2003).
Knuf, M. et al. Neonatal vaccination with an acellular pertussis vaccine accelerates the acquisition of pertussis antibodies in infants. J. Pediatr. 152, 655–660 (2008).
Dengrove, J. et al. IgG and IgG subclass specific antibody responses to diphtheria and tetanus toxoids in newborns and infants given DTP immunization. Pediatr. Res. 20, 735–739 (1986).
Halasa, N. B., O'Shea, A., Shi, J. R., LaFleur, B. J. & Edwards, K. M. Poor immune responses to a birth dose of diphtheria, tetanus, and acellular pertussis vaccine. J. Pediatr. 153, 327–332 (2008).
Siegrist, C. A. Blame vaccine interference, not neonatal immunization, for suboptimal responses after neonatal diphtheria, tetanus, and acellular pertussis immunization. J. Pediatr. 153, 305–307 (2008).
Ridings, J., Dinan, L., Williams, R., Roberton, D. & Zola, H. Somatic mutation of immunoglobulin VH6 genes in human infants. Clin. Exp. Immunol. 114, 33–39 (1998).
Lucas, A. H., Azmi, F. H., Mink, C. M. & Granoff, D. M. Age-dependent V region expression in the human antibody response to the Haemophilus influenzae type b polysaccharide. J. Immunol. 150, 2056–2061 (1993).
Schallert, N. et al. Generation of adult-like antibody avidity profiles after early-life immunization with protein vaccines. Eur. J. Immunol. 32, 752–760 (2002).
Hetherington, S. V. & Rutkowski, A. F. Antibody affinity in infants after immunization with conjugated capsular polysaccharide from Haemophilus influenzae type b. J. Infect. Dis. 162, 1185–1188 (1990).
Weitkamp, J. H. et al. Infant and adult human B cell responses to rotavirus share common immunodominant variable gene repertoires. J. Immunol. 171, 4680–4688 (2003).
Zemlin, M. et al. The postnatal maturation of the immunoglobulin heavy chain IgG repertoire in human preterm neonates is slower than in term neonates. J. Immunol. 178, 1180–1188 (2007).
Kolar, G. R., Yokota, T., Rossi, M. I., Nath, S. K. & Capra, J. D. Human fetal, cord blood, and adult lymphocyte progenitors have similar potential for generating B cells with a diverse immunoglobulin repertoire. Blood 104, 2981–2987 (2004).
Nair, N. et al. Age-dependent differences in IgG isotype and avidity induced by measles vaccine received during the first year of life. J. Infect. Dis. 196, 1339–1345 (2007).
Smith, K. G., Light, A., Nossal, G. J. & Tarlinton, D. M. The extent of affinity maturation differs between the memory and antibody-forming cell compartments in the primary immune response. EMBO J. 16, 2996–3006 (1997).
Gatto, D. et al. Complement receptors regulate differentiation of bone marrow plasma cell precursors expressing transcription factors Blimp-1 and XBP-1. J. Exp. Med. 201, 993–1005 (2005).
Bryant, V. L. et al. Cytokine-mediated regulation of human B cell differentiation into Ig-secreting cells: predominant role of IL-21 produced by CXCR5+ T follicular helper cells. J. Immunol. 179, 8180–8190 (2007).
Benson, M. J., Erickson, L. D., Gleeson, M. W. & Noelle, R. J. Affinity of antigen encounter and other early B-cell signals determine B-cell fate. Curr. Opin. Immunol. 19, 275–280 (2007).
Latz, E., Franko, J., Golenbock, D. T. & Schreiber, J. R. Haemophilus influenzae type b–outer membrane protein complex glycoconjugate vaccine induces cytokine production by engaging human toll-like receptor 2 (TLR2) and requires the presence of TLR2 for optimal immunogenicity. J. Immunol. 172, 2431–2438 (2004).
Decker, M. D., Edwards, K. M., Bradley, R. & Palmer, P. Comparative trial in infants of four conjugate Haemophilus influenzae type b vaccines. J. Pediatr. 120, 184–189 (1992).
Borrow, R. et al. Immunogenicity of, and immunologic memory to, a reduced primary schedule of meningococcal C–tetanus toxoid conjugate vaccine in infants in the United Kingdom. Infect. Immun. 71, 5549–5555 (2003).
Samandari, T. et al. Differences in response to a hepatitis B vaccine booster dose among Alaskan children and adolescents vaccinated during infancy. Pediatrics 120, e373–e381 (2007).
Lu, C. Y. et al. Humoral and cellular immune responses to a hepatitis B vaccine booster 15–18 years after neonatal immunization. J. Infect. Dis. 197, 1419–1426 (2008).
Amanna, I. J., Carlson, N. E. & Slifka, M. K. Duration of humoral immunity to common viral and vaccine antigens. N. Engl. J. Med. 357, 1903–1915 (2007). An excellent paper that measures the duration of antiviral antibody responses and shows that for most of the responses tested there was no correlation between peripheral memory B-cell numbers and antibody levels.
Ochsenbein, A. F. et al. Protective long-term antibody memory by antigen-driven and T help-dependent differentiation of long-lived memory B cells to short-lived plasma cells independent of secondary lymphoid organs. Proc. Natl Acad. Sci. USA 97, 13263–13268 (2000).
Goodwin, K., Viboud, C. & Simonsen, L. Antibody response to influenza vaccination in the elderly: a quantitative review. Vaccine 24, 1159–1169 (2006).
Melegaro, A. & Edmunds, W. J. The 23-valent pneumococcal polysaccharide vaccine. Part I. Efficacy of PPV in the elderly: a comparison of meta-analyses. Eur. J. Epidemiol. 19, 353–363 (2004).
Weinberger, B., Herndler-Brandstetter, D., Schwanninger, A., Weiskopf, D. & Grubeck-Loebenstein, B. Biology of immune responses to vaccines in elderly persons. Clin. Infect. Dis. 46, 1078–1084 (2008).
Hallgren, H. M., Buckley, C. E. III, Gilbertsen, V. A. & Yunis, E. J. Lymphocyte phytohemagglutinin responsiveness, immunoglobulins and autoantibodies in aging humans. J. Immunol. 111, 1101–1107 (1973).
Weksler, M. E. & Szabo, P. The effect of age on the B-cell repertoire. J. Clin. Immunol. 20, 240–249 (2000).
Welch, M. J., Fong, S., Vaughan, J. & Carson, D. Increased frequency of rheumatoid factor precursor B lymphocytes after immunization of normal adults with tetanus toxoid. Clin. Exp. Immunol. 51, 299–304 (1983).
Mariotti, S. et al. Thyroid and other organ-specific autoantibodies in healthy centenarians. Lancet 339, 1506–1508 (1992).
Kyle, R. A. et al. Prevalence of monoclonal gammopathy of undetermined significance. N. Engl. J. Med. 354, 1362–1369 (2006).
Stoop, J. W., Zegers, B. J., Sander, P. C. & Ballieux, R. E. Serum immunoglobulin levels in healthy children and adults. Clin. Exp. Immunol. 4, 101–112 (1969).
Kirkland, J. L., Tchkonia, T., Pirtskhalava, T., Han, J. & Karagiannides, I. Adipogenesis and aging: does aging make fat go MAD? Exp. Gerontol. 37, 757–767 (2002).
Johnson, S. A. & Cambier, J. C. Ageing, autoimmunity and arthritis: senescence of the B cell compartment — implications for humoral immunity. Arthritis Res. Ther. 6, 131–139 (2004).
Bohnhorst, J. O., Bjorgan, M. B., Thoen, J. E., Natvig, J. B. & Thompson, K. M. Bm1–Bm5 classification of peripheral blood B cells reveals circulating germinal center founder cells in healthy individuals and disturbance in the B cell subpopulations in patients with primary Sjogren's syndrome. J. Immunol. 167, 3610–3618 (2001).
Wei, C. et al. A new population of cells lacking expression of CD27 represents a notable component of the B cell memory compartment in systemic lupus erythematosus. J. Immunol. 178, 6624–6633 (2007).
Sanz, I., Wei, C., Lee, F. E. & Anolik, J. Phenotypic and functional heterogeneity of human memory B cells. Semin. Immunol. 20, 67–82 (2008).
Mamani-Matsuda, M. et al. The human spleen is a major reservoir for long-lived vaccinia virus-specific memory B cells. Blood 111, 4653–4659 (2008). This paper reports the differential distribution of memory B cells between blood and spleen, and the lack of any significant correlation between specific memory B cells in the blood and the serum titres of their specific antibody.
Fairfax, K. A., Kallies, A., Nutt, S. L. & Tarlinton, D. M. Plasma cell development: from B-cell subsets to long-term survival niches. Semin. Immunol. 20, 49–58 (2008).
Tarlinton, D., Radbruch, A., Hiepe, F. & Dorner, T. Plasma cell differentiation and survival. Curr. Opin. Immunol. 20, 162–169 (2008).
Radbruch, A. et al. Competence and competition: the challenge of becoming a long-lived plasma cell. Nature Rev. Immunol. 6, 741–750 (2006).
Han, S. et al. Enhanced differentiation of splenic plasma cells but diminished long-lived high-affinity bone marrow plasma cells in aged mice. J. Immunol. 170, 1267–1273 (2003).
Kyle, R. A. & Rajkumar, S. V. Monoclonal gammopathy of undetermined significance. Br. J. Haematol. 134, 573–589 (2006).
Luscieti, P., Hubschmid, T., Cottier, H., Hess, M. W. & Sobin, L. H. Human lymph node morphology as a function of age and site. J. Clin. Pathol. 33, 454–461 (1980).
Pawelec, G. et al. T cells and aging, January 2002 update. Front. Biosci. 7, d1056–d1183 (2002).
Frasca, D., Nguyen, D., Riley, R. L. & Blomberg, B. B. Effects of aging on proliferation and E47 transcription factor activity induced by different stimuli in murine splenic B cells. Mech. Ageing Dev. 124, 361–369 (2003).
Weng, N. P., Levine, B. L., June, C. H. & Hodes, R. J. Human naive and memory T lymphocytes differ in telomeric length and replicative potential. Proc. Natl Acad. Sci. USA 92, 11091–11094 (1995).
Weng, N. P., Granger, L. & Hodes, R. J. Telomere lengthening and telomerase activation during human B cell differentiation. Proc. Natl Acad. Sci. USA 94, 10827–10832 (1997).
Son, N. H., Murray, S., Yanovski, J., Hodes, R. J. & Weng, N. Lineage-specific telomere shortening and unaltered capacity for telomerase expression in human T and B lymphocytes with age. J. Immunol. 165, 1191–1196 (2000).
Aydar, Y., Wu, J., Song, J., Szakal, A. K. & Tew, J. G. FcγRII expression on follicular dendritic cells and immunoreceptor tyrosine-based inhibition motif signaling in B cells. Eur. J. Immunol. 34, 98–107 (2004).
Aydar, Y., Balogh, P., Tew, J. G. & Szakal, A. K. Follicular dendritic cells in aging, a “bottle-neck” in the humoral immune response. Ageing Res. Rev. 3, 15–29 (2004).
Rogerson, B. J., Harris, D. P., Swain, S. L. & Burgess, D. O. Germinal center B cells in Peyer's patches of aged mice exhibit a normal activation phenotype and highly mutated IgM genes. Mech. Ageing Dev. 124, 155–165 (2003).
Lumsden, J. M., Williams, J. A. & Hodes, R. J. Differential requirements for expression of CD80/86 and CD40 on B cells for T-dependent antibody responses in vivo. J. Immunol. 170, 781–787 (2003).
Eaton, S. M., Burns, E. M., Kusser, K., Randall, T. D. & Haynes, L. Age-related defects in CD4 T cell cognate helper function lead to reductions in humoral responses. J. Exp. Med. 200, 1613–1622 (2004).
Haynes, L. & Eaton, S. M. The effect of age on the cognate function of CD4+ T cells. Immunol. Rev. 205, 220–228 (2005).
Kolar, G. R., Mehta, D., Wilson, P. C. & Capra, J. D. Diversity of the Ig repertoire is maintained with age in spite of reduced germinal centre cells in human tonsil lymphoid tissue. Scand. J. Immunol. 64, 314–324 (2006).
Banerjee, M., Mehr, R., Belelovsky, A., Spencer, J. & Dunn-Walters, D. K. Age- and tissue-specific differences in human germinal center B cell selection revealed by analysis of IgVH gene hypermutation and lineage trees. Eur. J. Immunol. 32, 1947–1957 (2002).
Howard, W. A., Gibson, K. L. & Dunn-Walters, D. K. Antibody quality in old age. Rejuvenation Res. 9, 117–125 (2006).
Guerrettaz, L. M., Johnson, S. A. & Cambier, J. C. Acquired hematopoietic stem cell defects determine B-cell repertoire changes associated with aging. Proc. Natl Acad. Sci. USA 105, 11898–11902 (2008).
Gibson, K. L. et al. B-cell diversity decreases in old age and is correlated with poor health status. Aging Cell 8, 18–25 (2009). A spectratype analysis across the complementarity-determining region 3 in individuals of different ages that shows the decline in the B-cell repertoire in older individuals and at the same time maps their health status on the analysis, thereby linking frailty with repertoire distortion.
Healy, C. M. & Baker, C. J. Maternal immunization. Pediatr. Infect. Dis. J. 26, 945–948 (2007).
De Bruijn, I. et al. Antibody induction by virosomal, MF59-adjuvanted, or conventional influenza vaccines in the elderly. Vaccine 26, 119–127 (2007).
Sutherland, J. S. et al. Activation of thymic regeneration in mice and humans following androgen blockade. J. Immunol. 175, 2741–2753 (2005).
Dixit, V. D. et al. Ghrelin promotes thymopoiesis during aging. J. Clin. Invest. 117, 2778–2790 (2007).
Aspinall, R. et al. Old rhesus macaques treated with interleukin-7 show increased TREC levels and respond well to influenza vaccination. Rejuvenation Res. 10, 5–18 (2007).
Szabo, P. et al. Maturation of B cell precursors is impaired in thymic-deprived nude and old mice. J. Immunol. 161, 2248–2253 (1998).
Acknowledgements
The authors are grateful to all who participated in or supported their research projects.
Author information
Authors and Affiliations
Corresponding author
Related links
FURTHER INFORMATION
Glossary
- Infant
-
A ≤ 12-month-old human baby or a ≤ 3-week-old mouse (experimental definition).
- Neonate
-
A ≤ 28-day-old human infant or a ≤7-day-old mouse.
- T-cell-independent B-cell response
-
A B-cell response that is elicited without the requirement for CD4+ T-cell co-stimulation, which is a characteristic of responses to most polysaccharide antigens.
- T-cell-dependent B-cell response
-
A B-cell response that requires co-stimulatory signals provided by CD4+ T cells, a characteristic of responses to all protein or conjugate vaccines.
- Seroconversion
-
The development of detectable specific antibodies in the serum as a result of infection or immunization.
- Rheumatoid factor
-
An autoantibody specific for the Fc portion of IgG, which is most relevant in rheumatoid arthritis.
- Benign monoclonal gammopathy
-
A condition in which a low or non-quantifiable level of a monoclonal paraprotein is detected in the blood by protein electrophoresis.
- Telomere
-
A region of repetitive DNA at the end of chromosomes, which protects the end of the chromosome from destruction.
Rights and permissions
About this article
Cite this article
Siegrist, CA., Aspinall, R. B-cell responses to vaccination at the extremes of age. Nat Rev Immunol 9, 185–194 (2009). https://doi.org/10.1038/nri2508
Issue date:
DOI: https://doi.org/10.1038/nri2508
This article is cited by
-
Pneumokokkenimpfung im Säuglings- und im Kleinkindalter
Monatsschrift Kinderheilkunde (2024)
-
Dilemmas and options for COVID-19 vaccination in children
Italian Journal of Pediatrics (2023)
-
Antibody deficiencies in children are associated with prematurity and a family history of infections
Pediatric Research (2023)
-
Prediction of long-term humoral response induced by the two-dose heterologous Ad26.ZEBOV, MVA-BN-Filo vaccine against Ebola
npj Vaccines (2023)
-
T cell repertoire breadth is associated with the number of acute respiratory infections in the LoewenKIDS birth cohort
Scientific Reports (2023)