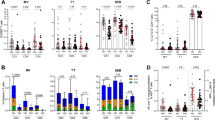
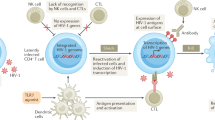
Abstract
Even before the partial success of a preventive HIV vaccine in a recent Phase III clinical trial, there had been an active research effort to determine one or more immune correlates of protection for HIV infection. This effort has been hampered by the lack of natural protective immunity against HIV. As a result, most of the studies have focused on long-term non-progressive infection or other clinical situations, none of which fully recapitulates protective immunity against HIV. Although this effort has been successful in defining characteristics of T cells in acute and non-progressive HIV infection, and has therefore greatly expanded our knowledge of the immunopathogenesis of AIDS, its success in defining immune correlates of protection is less clear. In this Opinion article we offer a perspective on how successful this effort has been in defining immune correlates of protection that have been, or will be, of use in the development of an HIV vaccine. Our view is that investing in an iterative approach to human vaccine efficacy trials of sufficient size and sampling frequency will improve the likelihood that an immune correlate of vaccine protection will be defined.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Buchbinder, S. P. et al. Efficacy assessment of a cell-mediated immunity HIV-1 vaccine (the Step Study): a double-blind, randomised, placebo-controlled, test-of-concept trial. Lancet 372, 1881–1893 (2008).
Flynn, N. M. et al. Placebo-controlled phase 3 trial of a recombinant glycoprotein 120 vaccine to prevent HIV-1 infection. J. Infect. Dis. 191, 654–665 (2005).
Pitisuttithum, P. et al. Randomized, double-blind, placebo-controlled efficacy trial of a bivalent recombinant glycoprotein 120 HIV-1 vaccine among injection drug users in Bangkok, Thailand. J. Infect. Dis. 194, 1661–1671 (2006).
Rerks-Ngarm, S. et al. Vaccination with ALVAC and AIDSVAX to prevent HIV-1 infection in Thailand. N. Engl. J. Med. 361, 2209–2220 (2009).
Pantaleo, G. & Koup, R. A. Correlates of immune protection in HIV-1 infection: what we know, what we don't know, what we should know. Nature Med. 10, 806–810 (2004).
Plotkin, S. A. Vaccines: correlates of vaccine-induced immunity. Clin. Infect. Dis. 47, 401–409 (2008).
Fowke, K. R. et al. Resistance to HIV-1 infection among persistently seronegative prostitutes in Nairobi, Kenya. Lancet 348, 1347–1351 (1996).
Baker, B. M., Block, B. L., Rothchild, A. C. & Walker, B. D. Elite control of HIV infection: implications for vaccine design. Expert Opin. Biol. Ther. 9, 55–69 (2009).
Leligdowicz, A. & Rowland-Jones, S. Tenets of protection from progression to AIDS: lessons from the immune responses to HIV-2 infection. Expert Rev. Vaccines 7, 319–331 (2008).
Koup, R. A. et al. Temporal association of cellular immune responses with the initial control of viremia in primary human immunodeficiency virus type 1 syndrome. J. Virol. 68, 4650–4655 (1994).
McMichael, A. J., Borrow, P., Tomaras, G. D., Goonetilleke, N. & Haynes, B. F. The immune response during acute HIV-1 infection: clues for vaccine development. Nature Rev. Immunol. 10, 11–23 (2010).
Price, H. et al. A TRIM5α exon 2 polymorphism is associated with protection from HIV-1 infection in the Pumwani sex worker cohort. AIDS 24, 1813–1821 (2010).
Rowland-Jones, S. L. et al. Cytotoxic T cell responses to multiple conserved HIV epitopes in HIV-resistant prostitutes in Nairobi. J. Clin. Invest. 102, 1758–1765 (1998).
Rosenberg, E. S. et al. Vigorous HIV-1-specific CD4+ T cell responses associated with control of viremia. Science 278, 1447–1450 (1997).
Edwards, B. H. et al. Magnitude of functional CD8+ T-cell responses to the Gag protein of human immunodeficiency virus type 1 correlates inversely with viral load in plasma. J. Virol. 76, 2298–2305 (2002).
Ogg, G. S. et al. Quantitation of HIV-1-specific cytotoxic T lymphocytes and plasma load of viral RNA. Science 279, 2103–2106 (1998).
Burgers, W. A. et al. Association of HIV-specific and total CD8+ T memory phenotypes in subtype C HIV-1 infection with viral set point. J. Immunol. 182, 4751–4761 (2009).
Addo, M. M. et al. Fully differentiated HIV-1 specific CD8+ T effector cells are more frequently detectable in controlled than in progressive HIV-1 infection. PLoS ONE 2, e321 (2007).
Hess, C. et al. HIV-1 specific CD8+ T cells with an effector phenotype and control of viral replication. Lancet 363, 863–866 (2004).
Day, C. L. et al. Proliferative capacity of epitope-specific CD8 T-cell responses is inversely related to viral load in chronic human immunodeficiency virus type 1 infection. J. Virol. 81, 434–438 (2007).
Hersperger, A. R. et al. Perforin expression directly ex vivo by HIV-specific CD8 T-cells is a correlate of HIV elite control. PLoS Pathog. 6, e1000917 (2010).
Migueles, S. A. et al. Lytic granule loading of CD8+ T cells is required for HIV-infected cell elimination associated with immune control. Immunity 29, 1009–1021 (2008).
Yang, O. O. et al. Efficient lysis of human immunodeficiency virus type 1-infected cells by cytotoxic T lymphocytes. J. Virol. 70, 5799–5806 (1996).
Blackbourn, D. J. et al. Suppression of HIV replication by lymphoid tissue CD8+ cells correlates with the clinical state of HIV-infected individuals. Proc. Natl Acad. Sci. USA 93, 13125–13130 (1996).
Spentzou, A. et al. Viral inhibition assay: a CD8 T cell neutralization assay for use in clinical trials of HIV-1 vaccine candidates. J. Infect. Dis. 201, 720–729 (2010).
Yang, O. O. et al. Suppression of human immunodeficiency virus type 1 replication by CD8+ cells: evidence for HLA class I-restricted triggering of cytolytic and noncytolytic mechanisms. J. Virol. 71, 3120–3128 (1997).
Betts, M. R. et al. HIV nonprogressors preferentially maintain highly functional HIV-specific CD8+ T cells. Blood 107, 4781–4789 (2006).
Geldmacher, C. et al. Minor viral and host genetic polymorphisms can dramatically impact the biologic outcome of an epitope-specific CD8 T-cell response. Blood 114, 1553–1562 (2009).
Simons, B. C. et al. Despite biased TRBV gene usage against a dominant HLA B57-restricted epitope, TCR diversity can provide recognition of circulating epitope variants. J. Immunol. 181, 5137–5146 (2008).
Day, C. L. et al. PD-1 expression on HIV-specific T cells is associated with T-cell exhaustion and disease progression. Nature 443, 350–354 (2006).
Petrovas, C. et al. PD-1 is a regulator of virus-specific CD8+ T cell survival in HIV infection. J. Exp. Med. 203, 2281–2292 (2006).
Trautmann, L. et al. Upregulation of PD-1 expression on HIV-specific CD8+ T cells leads to reversible immune dysfunction. Nature Med. 12, 1198–1202 (2006).
Kiepiela, P. et al. CD8+ T-cell responses to different HIV proteins have discordant associations with viral load. Nature Med. 13, 46–53 (2007).
Rolland, M. et al. Broad and Gag-biased HIV-1 epitope repertoires are associated with lower viral loads. PLoS ONE 3, e1424 (2008).
Zuniga, R. et al. Relative dominance of Gag p24-specific cytotoxic T lymphocytes is associated with human immunodeficiency virus control. J. Virol. 80, 3122–3125 (2006).
Gao, X. et al. Effect of a single amino acid change in MHC class I molecules on the rate of progression to AIDS. N. Engl. J. Med. 344, 1668–1675 (2001).
Hendel, H. et al. New class I and II HLA alleles strongly associated with opposite patterns of progression to AIDS. J. Immunol. 162, 6942–6946 (1999).
Kaslow, R. A. et al. Influence of combinations of human major histocompatibility complex genes on the course of HIV-1 infection. Nature Med. 2, 405–411 (1996).
Migueles, S. A. et al. HLA B*5701 is highly associated with restriction of virus replication in a subgroup of HIV-infected long term nonprogressors. Proc. Natl Acad. Sci. USA 97, 2709–2714 (2000).
Goulder, P. J. et al. Novel, cross-restricted, conserved, and immunodominant cytotoxic T lymphocyte epitopes in slow progressors in HIV type 1 infection. AIDS Res. Hum. Retroviruses 12, 1691–1698 (1996).
Turnbull, E. L. et al. HIV-1 epitope-specific CD8+ T cell responses strongly associated with delayed disease progression cross-recognize epitope variants efficiently. J. Immunol. 176, 6130–6146 (2006).
Martinez-Picado, J. et al. Fitness cost of escape mutations in p24 Gag in association with control of human immunodeficiency virus type 1. J. Virol. 80, 3617–3623 (2006).
Schneidewind, A. et al. Escape from the dominant HLA-B27-restricted cytotoxic T-lymphocyte response in Gag is associated with a dramatic reduction in human immunodeficiency virus type 1 replication. J. Virol. 81, 12382–12393 (2007).
Betts, M. R. et al. Characterization of functional and phenotypic changes in anti-Gag vaccine-induced T cell responses and their role in protection after HIV-1 infection. Proc. Natl Acad. Sci. USA 102, 4512–4517 (2005).
Streeck, H. et al. Human immunodeficiency virus type 1-specific CD8+ T-cell responses during primary infection are major determinants of the viral set point and loss of CD4+ T cells. J. Virol. 83, 7641–7648 (2009).
Goulder, P. J. et al. Substantial differences in specificity of HIV-specific cytotoxic T cells in acute and chronic HIV infection. J. Exp. Med. 193, 181–194 (2001).
Goonetilleke, N. et al. The first T cell response to transmitted/founder virus contributes to the control of acute viremia in HIV-1 infection. J. Exp. Med. 206, 1253–1272 (2009).
Leslie, A. J. et al. HIV evolution: CTL escape mutation and reversion after transmission. Nature Med. 10, 282–289 (2004).
Feeney, M. E. et al. HIV-1 viral escape in infancy followed by emergence of a variant-specific CTL response. J. Immunol. 174, 7524–7530 (2005).
Goepfert, P. A. et al. Transmission of HIV-1 Gag immune escape mutations is associated with reduced viral load in linked recipients. J. Exp. Med. 205, 1009–1017 (2008).
Goulder, P. J. et al. Evolution and transmission of stable CTL escape mutations in HIV infection. Nature 412, 334–338 (2001).
Gray, R. H. et al. Probability of HIV-1 transmission per coital act in monogamous, heterosexual, HIV-1-discordant couples in Rakai, Uganda. Lancet 357, 1149–1153 (2001).
Wawer, M. J. et al. Rates of HIV-1 transmission per coital act, by stage of HIV-1 infection, in Rakai, Uganda. J. Infect. Dis. 191, 1403–1409 (2005).
Salazar-Gonzalez, J. F. et al. Genetic identity, biological phenotype, and evolutionary pathways of transmitted/founder viruses in acute and early HIV-1 infection. J. Exp. Med. 206, 1273–1289 (2009).
Keele, B. F. et al. Low-dose rectal inoculation of rhesus macaques by SIVsmE660 or SIVmac251 recapitulates human mucosal infection by HIV-1. J. Exp. Med. 206, 1117–1134 (2009).
Hudgens, M. G. et al. Power to detect the effects of HIV vaccination in repeated low-dose challenge experiments. J. Infect. Dis. 200, 609–613 (2009).
Morgan, C. et al. The use of nonhuman primate models in HIV vaccine development. PLoS Med. 5, e173 (2008).
Nishimura, Y. et al. Highly pathogenic SHIVs and SIVs target different CD4+ T cell subsets in rhesus monkeys, explaining their divergent clinical courses. Proc. Natl Acad. Sci. USA 101, 12324–12329 (2004).
Shedlock, D. J., Silvestri, G. & Weiner, D. B. Monkeying around with HIV vaccines: using rhesus macaques to define 'gatekeepers' for clinical trials. Nature Rev. Immunol. 9, 717–728 (2009).
McElrath, M. J. et al. HIV-1 vaccine-induced immunity in the test-of-concept Step Study: a case-cohort analysis. Lancet 372, 1894–1905 (2008).
Shiver, J. W. et al. Replication-incompetent adenoviral vaccine vector elicits effective anti-immunodeficiency-virus immunity. Nature 415, 331–335 (2002).
Hansen, S. G. et al. Effector memory T cell responses are associated with protection of rhesus monkeys from mucosal simian immunodeficiency virus challenge. Nature Med. 15, 293–299 (2009).
D'Souza, M. P. & Altfeld, M. Measuring HIV-1-specific T cell immunity: how valid are current assays? J. Infect. Dis. 197, 337–339 (2008).
Streeck, H., Frahm, N. & Walker, B. D. The role of IFN-γ Elispot assay in HIV vaccine research. Nature Protoc. 4, 461–469 (2009).
Janetzki, S., Cox, J. H., Oden, N. & Ferrari, G. Standardization and validation issues of the ELISPOT assay. Methods Mol. Biol. 302, 51–86 (2005).
Baeten, J. M. et al. Trends in HIV-1 incidence in a cohort of prostitutes in Kenya: implications for HIV-1 vaccine efficacy trials. J. Acquir. Immune Defic. Syndr. 24, 458–464 (2000).
Kaul, R. et al. Reduced HIV risk-taking and low HIV incidence after enrollment and risk-reduction counseling in a sexually transmitted disease prevention trial in Nairobi, Kenya. J. Acquir. Immune Defic. Syndr. 30, 69–72 (2002).
van Loggerenberg, F. et al. Establishing a cohort at high risk of HIV infection in South Africa: challenges and experiences of the CAPRISA 002 acute infection study. PLoS ONE 3, e1954 (2008).
Qin, L., Gilbert, P. B., Corey, L., McElrath, M. J. & Self, S. G. A framework for assessing immunological correlates of protection in vaccine trials. J. Infect. Dis. 196, 1304–1312 (2007).
The Council of the Global HIV Vaccine Enterprise et al. The 2010 scientific strategic plan of the Global HIV Vaccine Enterprise. Nature Med. 16, 981–989 (2010).
Acknowledgements
We thank the many members of the Vaccine Research Center whose helpful discussions over the years have helped to frame the opinions expressed here.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Rights and permissions
About this article
Cite this article
Koup, R., Graham, B. & Douek, D. The quest for a T cell-based immune correlate of protection against HIV: a story of trials and errors. Nat Rev Immunol 11, 65–70 (2011). https://doi.org/10.1038/nri2890
Published:
Issue date:
DOI: https://doi.org/10.1038/nri2890
This article is cited by
-
Evolutionary dynamics of HIV at multiple spatial and temporal scales
Journal of Molecular Medicine (2012)
-
A 2020 vision for vaccines against HIV, tuberculosis and malaria
Nature (2011)
-
Novel cancer immunotherapy agents with survival benefit: recent successes and next steps
Nature Reviews Cancer (2011)