Abstract
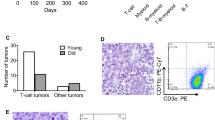
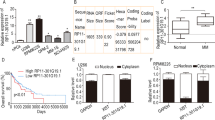
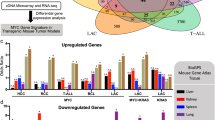
The multifaceted oncogene c-Myc plays important roles in the development and progression of human cancer. Recent in vitro and in vivo studies have shown that the p19Arf–Mdm2–p53 and the ribosomal protein (RP)–Mdm2–p53 pathways are both essential in preventing oncogenic c-Myc-induced tumorigenesis. Disruption of each pathway individually by p19Arf deletion or by Mdm2C305F mutation, which disrupts RP-Mdm2 binding, accelerates Eμ-myc transgene-induced pre-B/B-cell lymphoma in mice at seemingly similar paces with median survival around 10 and 11 weeks, respectively, compared to 20 weeks for Eμ-myc transgenic mice. Because p19Arf can inhibit ribosomal biogenesis through its interaction with nucleophosmin (NPM/B23), RNA helicase DDX5 and RNA polymerase I transcription termination factor (TTF-I), it has been speculated that the p19Arf–Mdm2–p53 and the RP–Mdm2–p53 pathways might be a single p19Arf–RP–Mdm2–p53 pathway, in which p19Arf activates p53 by inhibiting RP biosynthesis; thus, p19Arf deletion or Mdm2C305F mutation would result in similar consequences. Here, we generated mice with concurrent p19Arf deletion and Mdm2C305F mutation and investigated the compound mice for tumorigenesis in the absence and the presence of oncogenic c-Myc overexpression. In the absence of Eμ-myc transgene, the Mdm2C305F mutation did not elicit spontaneous tumors in mice, nor did it accelerate spontaneous tumors in mice with p19Arf deletion. In the presence of Eμ-myc transgene, however, Mdm2C305F mutation significantly accelerated p19Arf deletion-induced lymphomagenesis and promoted rapid metastasis. We found that when p19Arf–Mdm2–p53 and RP–Mdm2–p53 pathways are independently disrupted, oncogenic c-Myc-induced p53 stabilization and activation is only partially attenuated. When both pathways are concurrently disrupted, however, c-Myc-induced p53 stabilization and activation are essentially obliterated. Thus, the p19Arf–Mdm2–p53 and the RP–Mdm2–p53 are non-redundant pathways possessing similar capabilities to activate p53 upon c-Myc overexpression.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Meyer N, Penn LZ . Reflecting on 25 years with MYC. Nat Rev Cancer 2008; 8: 976–990.
Arabi A, Wu S, Ridderstrale K, Bierhoff H, Shiue C, Fatyol K et al. c-Myc associates with ribosomal DNA and activates RNA polymerase I transcription. Nat Cell Biol 2005; 7: 303–310.
Menssen A, Hermeking H . Characterization of the c-MYC-regulated transcriptome by SAGE: identification and analysis of c-MYC target genes. Proc Natl Acad Sci USA 2002; 99: 6274–6279.
Gomez-Roman N, Grandori C, Eisenman RN, White RJ . Direct activation of RNA polymerase III transcription by c-Myc. Nature 2003; 421: 290–294.
Miliani de Marval PL, Zhang Y . The RP-Mdm2-p53 pathway and tumorigenesis. Oncotarget 2011; 2: 234–238.
Mateyak MK, Obaya AJ, Adachi S, Sedivy JM . Phenotypes of c-Myc-deficient rat fibroblasts isolated by targeted homologous recombination. Cell Growth Differ 1997; 8: 1039–1048.
Evan GI, Wyllie AH, Gilbert CS, Littlewood TD, Land H, Brooks M et al. Induction of apoptosis in fibroblasts by c-myc protein. Cell 1992; 69: 119–128.
Packham G, Cleveland JL . c-Myc and apoptosis. Biochim Biophys Acta 1995; 1242: 11–28.
Lane DP . Cancer. p53, guardian of the genome. Nature 1992; 358: 15–16.
Meng X, Leslie P, Zhang Y, Dong J . Stem cells in a three-dimensional scaffold environment. Springerplus 2014; 3: 80.
Wu X, Bayle JH, Olson D, Levine AJ . The p53-mdm-2 autoregulatory feedback loop. Genes Dev 1993; 7: 1126–1132.
Sherr CJ . Divorcing ARF and p53: an unsettled case. Nat Rev Cancer 2006; 6: 663–673.
Eischen CM, Weber JD, Roussel MF, Sherr CJ, Cleveland JL . Disruption of the ARF-Mdm2-p53 tumor suppressor pathway in Myc-induced lymphomagenesis. Genes Dev 1999; 13: 2658–2669.
Zhang Y, Lu H . Signaling to p53: ribosomal proteins find their way. Cancer Cell 2009; 16: 369–377.
Macias E, Jin A, Deisenroth C, Bhat K, Mao H, Lindstrom MS et al. An ARF-independent c-MYC-activated tumor suppression pathway mediated by ribosomal protein-Mdm2 Interaction. Cancer Cell 2010; 18: 231–243.
Adams JM, Harris AW, Pinkert CA, Corcoran LM, Alexander WS, Cory S et al. The c-myc oncogene driven by immunoglobulin enhancers induces lymphoid malignancy in transgenic mice. Nature 1985; 318: 533–538.
Itahana K, Bhat KP, Jin A, Itahana Y, Hawke D, Kobayashi R et al. Tumor suppressor ARF degrades B23, a nucleolar protein involved in ribosome biogenesis and cell proliferation. Mol Cell 2003; 12: 1151–1164.
Sugimoto M, Kuo ML, Roussel MF, Sherr CJ . Nucleolar Arf tumor suppressor inhibits ribosomal RNA processing. Mol Cell 2003; 11: 415–424.
Lessard F, Morin F, Ivanchuk S, Langlois F, Stefanovsky V, Rutka J et al. The ARF tumor suppressor controls ribosome biogenesis by regulating the RNA polymerase I transcription factor TTF-I. Mol Cell 2010; 38: 539–550.
Saporita AJ, Chang HC, Winkeler CL, Apicelli AJ, Kladney RD, Wang J et al. RNA helicase DDX5 is a p53-independent target of ARF that participates in ribosome biogenesis. Cancer Res 2011; 71: 6708–6717.
Kamijo T, Bodner S, van de Kamp E, Randle DH, Sherr CJ . Tumor spectrum in ARF-deficient mice. Cancer Res 1999; 59: 2217–2222.
Lindstrom MS, Jin A, Deisenroth C, White Wolf G, Zhang Y . Cancer-associated mutations in the MDM2 zinc finger domain disrupt ribosomal protein interaction and attenuate MDM2-induced p53 degradation. Mol Cell Biol 2007; 27: 1056–1068.
Hoag WG . Spontaneous cancer in mice. Ann NY Acad Sci 1963; 108: 805–831.
Smith GS, Walford RL, Mickey MR . Lifespan and incidence of cancer and other diseases in selected long-lived inbred mice and their F 1 hybrids. J Natl Cancer Inst 1973; 50: 1195–1213.
White P, Liebhaber SA, Cooke NE . 129X1/SvJ mouse strain has a novel defect in inflammatory cell recruitment. J Immunol 2002; 168: 869–874.
Shetty S, Bruns T, Weston CJ, Stamataki Z, Oo YH, Long HM et al. Recruitment mechanisms of primary and malignant B cells to the human liver. Hepatology 2012; 56: 1521–1531.
Sidman CL, Marshall JD, Harris AW . Genetic studies on Emu-myc transgenic mice. Curr Top Microbiol Immunol 1988; 141: 94–99.
Aoudjit F, Potworowski EF, St-Pierre Y . The metastatic characteristics of murine lymphoma cell lines in vivo are manifested after target organ invasion. Blood 1998; 91: 623–629.
Eischen CM, Alt JR, Wang P . Loss of one allele of ARF rescues Mdm2 haploinsufficiency effects on apoptosis and lymphoma development. Oncogene 2004; 23: 8931–8940.
Toth P, Tucsek Z, Tarantini S, Sosnowska D, Gautam T, Mitschelen M et al. IGF-1 deficiency impairs cerebral myogenic autoregulation in hypertensive mice. J Cereb Blood Flow Metab 2014; 34: 1887–1897.
van Riggelen J, Yetil A, Felsher DW . MYC as a regulator of ribosome biogenesis and protein synthesis. Nat Rev Cancer 2010; 10: 301–309.
Zindy F, Eischen CM, Randle DH, Kamijo T, Cleveland JL, Sherr CJ et al. Myc signaling via the ARF tumor suppressor regulates p53-dependent apoptosis and immortalization. Genes Dev 1998; 12: 2424–2433.
Kamijo T, Zindy F, Roussel MF, Quelle DE, Downing JR, Ashmun RA et al. Tumor suppression at the mouse INK4a locus mediated by the alternative reading frame product p19ARF. Cell 1997; 91: 649–659.
Qi Y, Gregory MA, Li Z, Brousal JP, West K, Hann SR . p19ARF directly and differentially controls the functions of c-Myc independently of p53. Nature 2004; 431: 712–717.
Zhang Q, Spears E, Boone DN, Li Z, Gregory MA, Hann SR . Domain-specific c-Myc ubiquitylation controls c-Myc transcriptional and apoptotic activity. Proc Natl Acad Sci USA 2013; 110: 978–983.
Dai MS, Arnold H, Sun XX, Sears R, Lu H . Inhibition of c-Myc activity by ribosomal protein L11. EMBO J 2007; 26: 3332–3345.
Liao JM, Zhou X, Gatignol A, Lu H . Ribosomal proteins L5 and L11 co-operatively inactivate c-Myc via RNA-induced silencing complex. Oncogene 2014; 33: 4916–4923.
Acknowledgements
We thank Congying Wu, Yizhou He, Yong Liu, Aiwen Jin and Laura Tollini for their helpful advice and technical assistance. We are in debt to Yue Xiong, Guillermina Lozano, Gerard Evan, Charles Sherr and Norman Sharpless for their generosity in sharing mouse strains and reagents. This research was supported by grants from the National Institutes of Health (CA127770, CA100302 and CA167637) to YZ. This research was also supported by grants from the NSFC and Jiangsu Center for the Collaboration and Innovation of Cancer Biotherapy to YZ, the National S&T Major Project for Infectious Diseases of China (No. 2012ZX10002-017) to JD. NRC was supported in part by a grant from the National Institute of General Medical Sciences under award 5T32 GM007092.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Meng, X., Carlson, N., Dong, J. et al. Oncogenic c-Myc-induced lymphomagenesis is inhibited non-redundantly by the p19Arf–Mdm2–p53 and RP–Mdm2–p53 pathways. Oncogene 34, 5709–5717 (2015). https://doi.org/10.1038/onc.2015.39
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/onc.2015.39