Abstract
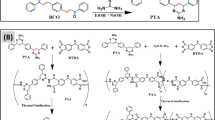
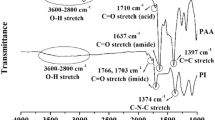
A novel tetraimide-dicarboxylic acid (I) was synthesized starting from the ring-opening addition of 4,4’-(hexafluoroisopropylidene)diphthalic anhydride (6FDA), 1,4-bis(4-aminophenoxy)benzene (TPEQ), and trimellitic anhydride (TMA) at a 1:2:2 molar ratio in N-methyl-2-pyrrolidone (NMP), followed by azeotropic condensation to the diacid I. A series of poly(amide-imide-imide)s (PAIIs) with inherent viscosities of 1.0–1.3 dL g-1 was prepared from the diacid I with various aromatic diamines by direct polycondensation. Most of the PAIIs were readily soluble in a variety of amide polar solvents, and even in less polar m-cresol and pyridine. Solvent-cast films had a tensile strength ranging from 80 to 101 MPa, elongation at break from 10 to 17%, and initial modulus from 2.0 to 2.4 GPa, and all of them exhibited clear yield points on their stress-strain curves. The glass transition temperature of these PAIIs was recorded at 260–289°C. They had 10% weight loss at a temperature above 520°C in air or nitrogen atmosphere.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
C. Feger, M. M. Khojasteh, and M. S. Htoo, “Advances in Polyimide Science and Technology”, Technomic Publishing Co. Inc., Lancaster, 1993.
M. J. M. Adadie and B. Sillion, “Polyimides and Other High—Temperature Polymers”, Elsevier Science Publishers B. V., Amsterdam, 1991.
K. L. Mittal, “Polyimide: Synthesis, Characterization, and Application”, vol. I & II, Plemnum Publishing Corporation, New York, N.Y., 1984.
C. Feger, M. M. Khojasteh, and J. E. McGrath, “Polyimides, Chemistry and Characterization”, Elsevier Science Publishers B. V., Amsterdam, 1989.
D. Wilson, H. D. Stenzenberger, and P. M. Hergenrother, “Polyimides”, Blackie, New York, N.Y., 1990.
P. E. Cassidy, “Thermally Stable Polymers”, Marcel Dekker, Inc., New York, N.Y., 1980.
H. H. Yang, “Aromatic High-Strength Fibers”, John Wiley & Sons, Inc., New York, N.Y., 1986.
V. L. Bell, B. L. Stump, and H. Gager, J. Polym. Sci., Polym. Chem. Ed., 14, 2275 (1976).
T. Takekoshi, J. G. Wirth, D. R. Heath, J. E. Kochanocoski, J. S. Manello, and M. T. Webber, J. Polym. Sci., Part A: Polym. Chem. , 18, 3069 (1980).
G. C. Eastmond and J. Paprotny, Macromolecules, 28, 2140 (1995).
G. C. Eastmond, J. Paprotny, and R. S. Irwin, Macromolecules, 29, 1382 (1996).
C. P. Yang and J. H. Lin, J. Polym. Sci., Part A: Polym. Chem., 31, 2153 (1993).
M. Sato and M. Yokoyama, Eur. Polym. J., 15, 733 (1979).
Q. Jin, T. Yamashita, and K. Horie, J. Polym. Sci., Part A: Polym. Chem., 32, 503 (1994).
C. P. Yang and S. H. Hsiao, Makromol. Chem., 190, 2119 (1989).
H. E. Frey, U. S. Patent 3 300 420 (Jan. 24, 1967).
Hitachi Chem. Co. Ltd., Fr. Patent 1 473 600 (Mar. 17, 1967).
J. Sambeth, Fr. Patent 1 498 015 (Oct. 13, 1967).
M. Kakimoto, R. Akiyama, Y. S. Negi, and Y. Imai, J. Polym. Sci., Part A: Polym. Chem., 26, 99 (1988).
Y. Imai, N. N. Maldar, and M. Kakimoto, J. Polym. Sci., Part A: Polym. Chem., 23, 2077 (1985).
J. F. Dezern, J. Polym. Sci., Part A: Polym. Chem., 26, 2157 (1988).
G. M. Bower and L. W. Frost, J. Polym. Sci., Part A-1: Polym. Chem., 3135 (1963).
C. P. Yang, S. H. Hsiao, and J. H. Lin, Makromol. Chem., 193, 1299 (1992).
S. H. Hsiao and C. P. Yang, Makromol. Chem., 191, 155 (1990).
C. P. Yang, J. M. Cheng, and S. H. Hsiao, Makromol. Chem., 193, 445 (1992).
C. P. Yang, J. Polym. Sci., Polym. Chem. Ed., 17, 3255 (1979).
C. P. Yang, S. H. Hsiao, and J. H. Lin., J. Polym. Sci., Part A: Polym. Chem., 30, 1865 (1992).
S. H. Hsiao and C. P. Yang, J. Polym. Sci., Part A: Polym. Chem., 28, 2169 (1990).
S. Maiti and A. Ray, J. Appl. Polym. Sci., 28, 225 (1983).
S. Maiti and A. Ray, J. Polym. Sci., Part A: Polym. Chem., 21, 999 (1983).
S. H. Hsiao and C. P. Yang, J. Polym. Sci., Part A: Polym. Chem., 28, 1149 (1990).
C. P. Yang, S. H. Hsiao, and W. L. Chou, J. Polym. Res., 2, 179 (1995).
C. P. Yang and J. J. Cherng, J. Polym. Sci., Part A: Polym. Chem., 33, 2209 (1995).
C. P. Yang and S. H. Hsiao, J. Appl. Polym. Sci., 30, 2883 (1985).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yang, CP., Chen, RS. & Wang, MJ. Synthesis and Properties of Organosoluble Poly(amide-imide-imide)s Based on Tetraimide-Dicarboxylic Acid Condensed from 4,4’-(Hexafluoroisopropylidene)diphthalic Anhydride, 1,4-Bis(4-aminophenoxy)benzene, and Trimellitic Anhydride, and Various Aromatic Diamines. Polym J 34, 194–202 (2002). https://doi.org/10.1295/polymj.34.194
Issue date:
DOI: https://doi.org/10.1295/polymj.34.194