Abstract
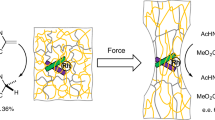
We measured the single-molecule interaction force between various hydroxybenzoic acids and a Si3N4 cantilever by atomic force microscopy in force curve mode. The hydroxybenzoic acids have different numbers/positions of hydroxyl groups covalently attached to the aromatic ring. The adhesion properties of these hydroxybenzoic acids were compared to determine the effect of the hydroxyl groups on single-molecule interactions. As the number of hydroxyl group increased, the number of single-molecule interactions increased and the interaction force also increased. The catechol system with two hydroxyl groups on the aromatic ring, similar to that found in mussel adhesive, has been well studied. Here, we demonstrate that gallic acid (GA) with three hydroxyl groups on the aromatic ring showed good adhesion and a large number of single-molecule interactions. We synthesized GA adhesives that exhibited sufficiently strong adhesion (10 MPa) to be of practical use.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Waite, J. H. & Tanzer, M. L. Polyphenolic substance of mytilus edulis: novel adhesive containing L-Dopa and hydroxyproline. Science 212, 1038–1040 (1981).
Waite, J. H. Surface chemistry - Mussel power. Nat. Mater. 7, 8–9 (2008).
Lee, H., Lee, P. B. & Messersmith, B. P. A reversible wet/dry adhesive inspired by mussels and geckos. Nature 448, 338 (2007).
Gunawan, R. C., King, A. J., Lee, P. B., Messersmith, B. P. & Miller, M. W. Surface presentation of bioactive ligands in a nonadhesive background using DOPA-tethered biotinylated poly(ethylene glycol). Langmuir 23, 10635–10643 (2007).
Lee, H., Dellatore, M. S., Miller, M. W. & Messersmith, B. P. Mussel-inspired surface chemistry for multifunctional coatings. Science 318, 426–430 (2007).
Lee, H., Lee, Y., Statz, R. A., Rho, J., Park, G. T. & Messersmith, B. P. Substrate-independent layer-by-layer assembly by using mussel-adhesive-inspired polymers. Adv. Mater. 20, 1619 (2008).
Lee, H., Rho, J. & Messersmith, B. P. Facile conjugation of biomolecules onto surfaces via mussel adhesive protein inspired coatings. Adv. Mater. 21, 431 (2009).
Saiz-Poseu, J., Sedo, J., Garcia, B., Benaiges, C., Parella, T., Alibes, R., Hernando, J., Busque, F. & Ruiz-Molina, D. Versatile nanostructured materials via direct reaction of functionalized catechols. Adv. Mater. 25, 2066–2070 (2013).
Darshan, D. G., Michael, L., Yu, Z., Amit, P. S., Saroj, N., Ulrike, T., Moshe, E. & Ganpati, R. Annealing-induced interfacial toughening using a molecular nanolayer. Nature 447, 299–302 (2007).
Matos-Perez, C. R., White, J. D. & Wilker, J. J. Polymer composition and substrate influences on the adhesive bonding of a biomimetic, cross-linking polymer. J. Am. Chem. Soc. 134, 9498–9505 (2012).
Lee, H., Scherer, N. F. & Messersmith, P. B. Single-molecule mechanics of mussel adhesion. Proc. Natl Acad. Sci. USA 103, 12999–13003 (2006).
Li, Y., Qin, M., Li, Y., Cao, Y. & Wang, W. Single molecule evidence for the adaptive binding of DOPA to different wet surfaces. Langmuir 30, 4358–4366 (2014).
Trosa, A. & Pizzi, A. Industrial hardboard and other panels binder from waste lignocellulosic liquors phenol-formaldehyde resins. Holz Roh. Werkstoff 56, 220–233 (1998).
GarroGalvez, J. M. & Riedl, B. Pyrogallol-formaldehyde thermosetting adhesives. J. Appl. Polym. Sci. 65, 399–408 (1997).
Sileika, T. S., Barrett, D. G., Zhang, R., Lau, K. H. & Messersmith, P. B. Colorless multifunctional coatings inspired by polyphenols found in tea, chocolate, and wine. Angew. Chem. Int. Ed. 52, 10766–10770 (2013).
Szekeres, M., Illés, E., Janko, C., Farkas, K., Tóth, I. Y., Nesztor, D., Zupkó, I., Földesi, I., Alexiou, C. & Tombácz, E. Hemocompatibility and biomedical potential of poly(gallic acid) coated iron oxide nanoparticles for theranostic use. J. Nanomed. Nanotechnol. 6, 1000252 (2015).
Pollastri, M. P., Porter, N. A., McIntosh, T. J. & Simon, S. A. Synthesis, structure, and thermal properties of 1, 2-dipalmitoylgalloylglycerol (DPGG), a novel self-adhering lipid. Chem. Phys. Lipids 104, 67–74 (2000).
Do Kang, H., Jung, H.-S., Ahn, N., Yang, S. M., Seo, S., Suh, K.-Y., Chang, P.-S., Jeon, N. L., Kim, J. & Kim., K. Janus-compartmental alginate microbeads having polydiacetylene liposomes and magnetic nanoparticles for visual lead(II) detection. ACS Appl. Mater. Interfaces 6, 10631–10637 (2014).
Schmidt, R. & DeWolf, C. E. Monolayer behavior of 1,2-dipalmitoylgalloylglycerol, a synthetic lipid with strong cohesive properties. Langmuir 20, 3284–3288 (2004).
Kaneko, D., Kinugawa, S., Matsumoto, K. & Kaneko, T. Terminally-catecholized hyper-branched polymers with high performance adhesive characteristics. Plant Biotechnol. 27, 293–296 (2010).
Kaneko, D., Wang, S., Matsumoto, K., Kinugawa, S., Yasaki, K., Chi, D. H. & Kaneko, T. Mussel-mimetic strong adhesive resin from bio-base polycoumarates. Polymer J. 43, 855–858 (2011).
Wang, S., Kaneko, D., Kan, K., Jin, X. & Kaneko, T. Syntheses of hyperbranched liquid-crystalline biopolymers with strong adhesion from phenolic phytomonomers. Pure Appl. Chem. 84, 2559–2568 (2012).
Wang, S., Tateyama, S., Kaneko, D., Ohki, S.-Y. & Kaneko, T. Synthesis of well-defined hyperbranched polymers bio-based on multifunctional phenolic acids and their structure-thermal property relationships. Polym. Degrad. Stabil. 96, 2048–2054 (2011).
Kaneko, D., Matsumoto, K., Kinugawa, S., Tateyama, S. & Kaneko, T. Effects of adhesive characteristics of the catechol group on fiber-reinforced plastics. Polymer J. 43, 944–947 (2011).
Chauzar, M., Tateyama, S., Ishikura, T., Matsumoto, K., Kaneko, D., Ebitani, K. & Kaneko, T. Hydrotalcites catalyze the acidolysis polymerization of phenolic acid to create highly heat-resistant bioplastics. Adv. Func. Mater 22, 3438–3444 (2012).
Hiraishi, N., Kaneko, D., Taira, S., Wang, S., Otsuki, M. & Tagami, J. Mussel-mimetic, bioadhesive polymers from plant-derived materials. J. Investig. Clin. Dent. 6, 59–62 (2013).
Kang, M.-S., Oh, J.-S., Kang, I.-C., Hong, S.-J. & Choi, C.-H. Inhibitory effect of methyl gallate and gallic acid on oral bacteria. J. Microbiol. 46, 744–750 (2008).
Kim, S.-H., Jun, C.-D., Suk, K., Choi, B.-J., Lim, H., Park, S. & Lee, S. H. Gallic acid inhibits histamine release and pro-inflammatory cytokine production in mast cells. Toxicol. Sci. 91, 123–131 (2006).
Tamura, T. Pocket Manual of Bioreagent Preparation 183–184 (Yodosha Co., Ltd, Tokyo, Japan, 2004).
Senden, T. J. Force microscopy and surface interactions. Curr. Opin. Colloid Interface Sci. 6, 85–101 (2001).
Oesterhelt, F., Rief, M. & Gaub, H. E. Single molecule force spectroscopy by AFM indicates helical structure of poly(ethylene-glycol) in water. New J. Phys. 1, 6.1–6.11 (1999).
Kienberger, F., Pastushenko, V. P., Kada, G., Gruber, H. J., Riener, C., Schindler, H. & Hinterdorfer, P. Static and dynamical propertiesof single poly(ethylene glycol) molecules investigated by force spectroscopy. Single Mol. 2, 123–128 (2000).
Smith, E. A. & Chen, W. How to prevent the loss of surface functionality derived from aminosilanes. Langmuir 24, 12405–12409 (2008).
Fadeev, A. Y. & McCarthy, T. J. Self-assembly is not the only reaction possible between alkyltrichlorosilanes and surfaces: monomolecular and oligomeric covalently attached layers of dichloro- and trichloroalkylsilanes on silicon. Langmuir 16, 7268–7274 (2000).
Barhoumi, H., Maaref, A. & Jaffrezic-Renault, N. Experimental study of thermodynamic surface characteristics and pH sensitivity of silicon dioxide and silicon nitride. Langmuir 26, 7165–7173 (2010).
Yang, Z., Yang, Y., Yan, W., Tu, Q., Wang, J. & Huang, N. Construction of polyfunctional coatings assisted by gallic acid to facilitate co-immobilization of diverse biomolecules. ACS Appl. Mater. Interfaces 5, 10495–10501 (2013).
Araujo, P. Z., Morando, P. J. & Blesa, M. A. Interaction of catechol and gallic acid with titanium dioxide in aqueous suspensions. 1. Equilibrium studies. Langmuir 21, 3470–3474 (2005).
Razvag, Y., Gutkin, V. & Reches, M. Probing the interaction of individual amino acids with inorganic surfaces using atomic force spectroscopy. Langmuir 29, 10102–10109 (2013).
Acknowledgements
This research was financially supported by the New Energy and Industrial Technology Development Organization and the Kaneko project (11D16002d).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Kinugawa, S., Wang, S., Taira, S. et al. Single-molecule interaction force measurements of catechol analog monomers and synthesis of adhesive polymer using the results. Polym J 48, 715–721 (2016). https://doi.org/10.1038/pj.2015.140
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/pj.2015.140
This article is cited by
-
Gallol-Containing Polymers: Synthesis and Applications
Chemistry Africa (2023)