Abstract
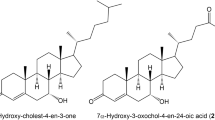
Unusual bile acids, such as unsaturated ketonic and 7β-hydroxylated bile acids, have been detected in urine early in life. To elucidate the normal profiles of usual and unusual urinary bile acids in the neonatal and pediatric periods, we measured the concentrations of 28 kinds in urine from normal newborns, infants, and children by gas chromatography-mass spectrometry. The mean total bile acid/Cr ratio in 7-d-old infants was significantly higher than in subjects of other age groups (birth, 2-4 mo, 5-7 mo, 11-12 mo, 2-3 y, 9-14 y, and adult) (p < 0.05). Relatively large amounts of unusual bile acids were detected during infancy, especially during the period up to 1 mo of age. At that time, 1β,3α,7α,12α-tetrahydroxy-5β-cholan-24-oic, 7α,12α-dihydroxy-3-oxo-5β-chol-1-en-24-oic, and 7α,12α-dihydroxy-3-oxo-4-cholen-24-oic acids were predominant among the unusual urinary bile acids present. Moreover, the levels of 3α,7β,12α-trihydroxy-5β-cholan-24-oic acid increased significantly after 2-4 mo of age. These results indicate that bile acid synthesis and metabolism in the liver of developing infants are significantly different from that occurring in the liver of adults. Significant amounts of urinary isomerized 7β-hydroxylated bile acids were detected after late infancy, probably because of changes in the intestinal bacterial flora response to a change in nutrition. We describe, for the first time, evidence of the epimerization of the 7α-hydroxyl group of cholic acid, which may be unique to human development.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Abbreviations
- GC-MS:
-
gas chromatography-mass spectrometry
- SIM:
-
selected ion monitoring
- Me-DMES:
-
methyl ester-dimethylethylsilyl ether
- HSDH:
-
hydroxysteroid dehydrogenase
References
Clayton PT, Leonard JV, Lawson AM, Setchell KDR, Andersson S, Egestad B, Sjövall J 1987 Familial giant cell hepatitis associated with synthesis of 3β,7α-dihydroxy- and 3β,7α,12α-trihydroxy-5-cholenoic acids. J Clin Invest 79: 1031–1038.
Setchell KDR, Suchy FJ, Weish MB, Zimmer-Nechemias L, Heubi J, Balistreri WF 1988 Δ4-3-Oxosteroid 5β-reductase deficiency described in identical twins with neonatal hepatitis: a new inborn error in bile acid synthesis. J Clin Invest 82: 2148–2157.
Almé B, Bremmelgaard A, Sjövall J, Thomassen P 1977 Analysis of metabolic profiles of bile acids in urine using a lipophilic anion exchanger and computerized gas-liquid chromatography-mass spectrometry. J Lipid Res 18: 339–362.
Shoda J, Mahara R, Osuga T, Tohma M, Ohnishi S, Miyazaki H, Tanaka N, Matsuzaki Y 1988 Similarity of unusual bile acids in human umbilical cord blood and amniotic fluid from newborns and in sera and urine from adult patients with cholestatic liver diseases. J Lipid Res 29: 847–858.
Shoda J, Osuga T, Matsuura K, Mahara R, Tohma M, Tanaka N, Matsuzaki Y, Miyazaki H 1989 Concurrent occurrence of 3β,12α-dihydroxy-5-cholenoic acid associated with 3β-hydroxy-5-cholenoic acid and their preferential urinary excretion in liver disease. J Lipid Res 30: 1233–1242.
Shoda J, Tanaka N, Ousuga T, Matsuura K, Miyazaki H 1990 Altered bile acid metabolism in liver disease. concurrent occurrence of C-1 and C-6 hydroxylated bile acid metabolites and their preferential excretion into urine. J Lipid Res 31: 249–259.
Tohma M, Mahara R, Takeshita H, Krosawa T, Ikegawa S, Nittono H 1985 Synthesis of the 1β-hydroxylated bile acids and identification of 1β,3α,7α-trihydroxy- and 1β,3α,12α-tetrahydroxy-5β-cholan-24-oic acids in human meconium. Chem Pharm Bull 33: 3071–3073.
Kimura A, Yamakawa R, Ushijima K, Fujisawa T, Kuriya N, Kato H, Inokuchi T, Mahara R, Kurosawa T, Tohma M 1994 Fetal bile acid metabolism during infancy: analysis of 1β-hydroxylated bile acids in urine, meconium, and feces. Hepatology 20: 819–824.
Mahara R, Kurosawa T, Tohma M 1989 Profile analysis of bile acids in human fetal and neonatal periods by gas chromatography-mass spectrometry. Proc Jap Soc Biomed Mass Spectrom 14: 67–74.
Setchell KDR, Dumaswala R, Colombo C, Ronchi M 1988 Hepatic bile acid metabolism during early development revealed from the analysis of human fetal gallbladder bile. J Biol Chem 263: 16637–16644.
Dumaswala R, Setchell KDR, Zimmer-Nechemias L, Iida J, Goto J, Nambara T 1989 Identification of 3α,4β,7α-trihydroxy-5β-cholanoic acid in human bile: reflection of a new pathway in bile acid metabolism in humans. J Lipid Res 30: 847–856.
Kurosawa T, Nomura Y, Mahara R, Yoshimura T, Kimura A, Ikegawa S, Tohma M 1995 Synthesis of 19-hydroxylated bile acids and identification of 3α,7α,12α,19-tetrahydroxy-5β-cholan-24-oic acid in human neonatal urine. Chem Pharm Bull 43: 1551–1557.
Murai T, Mahara R, Kurosawa T, Kimura A, Tohma M 1997 Determination of fetal bile acids in biological fluids from neonates by gas chromatography-negative ion chemical ionization mass spectrometry. J Chromatogr B 691: 13–22.
Kimura A, Suzuki M, Murai T, Inoue T, Kato H, Hori D, Nomura Y, Yoshimura T, Kurosawa T, Tohma M 1997 Perinatal bile acid metabolism: analysis of urinary bile acids in pregnant women and newborns. J Lipid Res 38: 1954–1962.
Tohma M, Mahara R, Takeshita H, Kurosawa T, Ikegawa S 1986 Synthesis of the 1β-hydroxylated bile acids, unusual bile acids in human biological fluids. Chem Pharm Bull 34: 2890–2899.
Tohma M, Mahara R, Takeshita H, Kurosawa T 1986 A convenient synthesis of 3β,12α-, 3β,7α-, and 3β,7β-dihydroxy-5-cholen-24-oic acids: unusual bile acids in human biological fluids. Steroids 48: 331–338.
Kurosawa T, Mahara R, Nittono H, Tohma 1989 Synthesis of 6-hydroxylated bile acids and identification of 3α,6α,7α,12α-tetrahydroxy-5β-cholan-24-oic acid in human meconium and neonatal urine. Chem Pharm Bull 37: 557–559.
Yoshimura T, Mahara R, Kurosawa T, Ikegawa S, Tohma M 1993 An efficient synthesis of 4β- and 6α-hydroxylated bile acids. Steroids 58: 52–58.
Suzuki M, Murai T, Yoshimura T, Kimura A, Kurosawa T, Tohma M 1997 Determination of 3-oxo-Δ4- and 3-oxo-Δ 4:6-bile acids and related compounds in biological fluids of infants with cholestasis by gas chromatography-mass spectrometry. J Chromatogr B 693: 11–21.
Lester R 1980 Physiological cholestasis. Gastroenterology 78: 864–865.
Suchy FJ, Balistreri WF, Heubi JE, Searcy JE, Levin RS 1981 Physiologic cholestasis: elevation of the primary serum bile acid concentration in normal infants. Gastroenterology 80: 1037–1041.
Balistreri WF, Heubi JE, Suchy FJ 1983 Immaturity of the enterohepatic circulation in early life: factors predisposing to "physiologic" maldigestion and cholestasis. J Pediatr Gastroenterol Nutr 2: 346–354.
Kimura A, Mahara R, Tohma M, Ushijima K, Yuge K, Ono E, Yamashita F 1989 Unusual 1β-hydroxylated bile acids in children with a paucity in interlobular bile ducts. Clin Chim Acta 185: 215–218.
Nakagawa M, Setchell KDR 1990 Bile acid metabolism in early life: studies of amniotic fluid. J Lipid Res 31: 1089–1098.
Wahlén E, Egestad B, Strandvik B, Sjövall J 1989 Ketonic bile acids in urine of infants during the neonatal period. J Lipid Res 30: 1847–1857.
Strandvik B, Wahlen E, Wikstrom S-A 1994 The urinary bile acid excretion in healthy premature and full-term infants during the neonatal period. Scand J Clin Lab Invest 54: 1–10.
Björkhem I, Einarsson K, Melone P, Hylemon P 1989 Mechanism of intestinal formation of deoxycholic acid from cholic acid in humans: evidence for a 3-oxo-Δ4-steroid intermediate. J Lipid Res 30: 1033–1039.
Wahlén E, Stradvik B 1993 Effects of different formula feeds on the developmental pattern of urinary bile acid excretion in infants. J Pediatr Gastroenterol Nutr 18: 9–19.
Jönsson G, Midtvedt AC, Norman A, Midtvedt T 1995 Intestinal microbial bile acid transformation in healthy infants. J Pediatr Gastroenterol Nutr 20: 394–402.
Macdonald IA, Sutherland JD 1983 Further studies on the bile salt induction of 7α-and 7β-hydroxysteroid dehydrogenases in Clostridium absonum.. Biochim Biophys Acta 750: 397–403.
Edenharder R, Knaflic T 1981 Epimerization of chenodeoxycholic acid to ursodeoxycholic acid by human intestinal lecithinase-lipase-negative Clostridia.. J Lipid Res 22: 652–658.
Hirano S, Masuda N 1982 Characterization of NADP-dependent 7β-hydroxysteroid dehydrogenases from Peptostreptococcus productus and Eubacterium aerofaciens. Appl Environ Microbiol 43: 1057–1063.
Hirano S, Masuda N 1981 Epimerization of the 7-hydroxy group of bile acids by combination of two kinds of microorganisms with 7α- and 7β-hydroxysteroid dehydrogenase activity, respectively. J Lipid Res 22: 1060–1068.
Sutherland JD, Holdeman LV, Williams CN, Macdonald IA 1984 Formation of urso-and ursodeoxy-cholic acids from primary bile acids by Clostridium limosum soil isolate. J Lipid Res 25: 1084–1089.
Armstrong MJ, Carey MC 1982 The hydrophobic-hydrophilic balance of bile salts. Inverse correlation between reverse-phase high performance liquid chromatographic mobilities and micellar cholesterol-solubilizing capacities. J Lipid Res 23: 70–80.
Gustafsson BE, Norman A, Sjövall J 1960 Influence of E. coli infection on turnover and metabolism of cholic acid in germ-free rats. Arch Biochem Biophys 91: 83–100.
Fromm H, Carlson EL, Hofmann AF, Farivar S, Amin P 1980 Metabolism in man of 7-keto lithocholic acid, precursor of cheno- and urso-deoxycholic acids. Am J Physiol 239:G161–G166.
Macdonald IA, Bokkenheuser VD, Winter J, McLernon AM, Mosbach EH 1983 Degradation of steroids in the human gut. J Lipid Res 24: 675–700.
Author information
Authors and Affiliations
Additional information
Supported in part by Grant-in-Aid 07670924 for Scientific Research from the Ministry of Education, Science, and Culture of Japan.
Rights and permissions
About this article
Cite this article
Kimura, A., Mahara, R., Inoue, T. et al. Profile of Urinary Bile Acids in Infants and Children: Developmental Pattern of Excretion of Unsaturated Ketonic Bile Acids and 7 β-Hydroxylated Bile Acids. Pediatr Res 45, 603–609 (1999). https://doi.org/10.1203/00006450-199904010-00022
Received:
Accepted:
Issue date:
DOI: https://doi.org/10.1203/00006450-199904010-00022
This article is cited by
-
Perinatal exposure to UDCA prevents neonatal cholestasis in Cyp2c70-/- mice with human-like bile acids
Pediatric Research (2023)
-
One-pot biosynthesis of 7β-hydroxyandrost-4-ene-3,17-dione from phytosterols by cofactor regeneration system in engineered mycolicibacterium neoaurum
Microbial Cell Factories (2022)
-
Bile Acid Synthesis Disorders in Japan: Long-Term Outcome and Chenodeoxycholic Acid Treatment
Digestive Diseases and Sciences (2021)
-
Urinary and serum oxysterols in children: developmental pattern and potential biomarker for pediatric liver disease
Scientific Reports (2020)
-
A combination of mutations in AKR1D1 and SKIV2L in a family with severe infantile liver disease
Orphanet Journal of Rare Diseases (2013)