Abstract
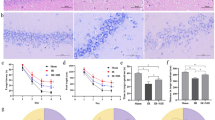
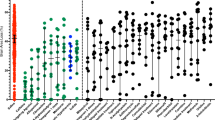
Dexamethasone (DEX) pretreatment has been shown to be neuroprotective in a neonatal rat model of hypoxia ischemia (HI). The exact mechanism of this neuroprotection is still unknown. This study used 31P nuclear magnetic resonance spectroscopy to monitor energy metabolism during a 3-h episode of HI in 7-d-old rat pups in one of two groups. The first group was pretreated with 0.1 mL saline (i.p.) and the second group was treated with 0.1 mL of 0.1mg/kg DEX (i.p.) 22 h before HI. Animals pretreated with DEX had elevated nucleoside triphosphate and phosphocreatine levels during HI when compared with controls. Saline-treated animals had significant decreases in nucleoside triphosphate and phosphocreatine and increases in inorganic phosphate over this same period. 31P nuclear magnetic resonance data unequivocally demonstrate preservation of energy metabolism during HI in neonatal rats pretreated with DEX. Animals pretreated with DEX had little or no brain damage following 3 h of HI when compared with matched controls, which experienced severe neuronal loss and cortical infarction. These same pretreated animals had an increase in blood beta-hydroxybutyrate levels before ischemia, suggesting an increase in ketone bodies, which is the neonate's primary energy source. Elevation of ketone bodies appears to be one of the mechanisms by which DEX pretreatment provides neuroprotection during HI in the neonatal rat.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Abbreviations
- DEX:
-
dexamethasone
- HI:
-
hypoxia ischemia
- NTP:
-
nucleoside triphosphate
- NMR:
-
nuclear magnetic resonance
- NMRS:
-
nuclear magnetic resonance spectroscopy
- BHB:
-
beta-hydroxybutyrate
- D-β-HBA:
-
D-β-hydroxybutyrate dehydrogenase
References
Effect of corticosteroids for fetal maturation on perinatal outcomes.. 1995 NIH Consensus Development Panel on the Effect of Corticosteroids for Fetal Maturation on Perinatal Outcomes see comments. JAMA 273: 413–418.
Altman DI, Young RS, Yagel SK 1984 Effects of dexamethasone in hypoxic-ischemic brain injury in the neonatal rat. Biol Neonate 46: 149–156.
Barks JD, Post M, Tuor UI 1991 Dexamethasone prevents hypoxic-ischemic brain damage in the neonatal rat. Pediatr Res 29: 558–563.
Tuor UI, Simone CS, Arellano R, Tanswell K, Post M 1993 Glucocorticoid prevention of neonatal hypoxic-ischemic damage: role of hyperglycemia and antioxidant enzymes. Brain Res 604: 165–172.
Tuor UI, Simone CS, Barks JD, Post M 1993 Dexamethasone prevents cerebral infarction without affecting cerebral blood flow in neonatal rats. Stroke 24: 452–457.
Tuor UI 1995 Dexamethasone and the prevention of neonatal hypoxic-ischemic brain damage. Ann N Y Acad Sci 765: 179–95; discussion 196–197.
Williams GD, Palmer C, Roberts RL, Heitjan DF, Smith MB 1992 31P NMR spectroscopy of perinatal hypoxic-ischemic brain damage: a model to evaluate neuroprotective drugs in immature rats. NMR Biomed 5: 145–153.
Cataltepe OVR, Heitjan DF, Towfighi J 1995 Effect of status epilepticus on hypoxic-ischemic brain damage in the immature rat. Pediatr Res 38: 251–257.
Williams GD PC, Heitjan DF, Smith MB 1992 Allopurinol preserves cerebral energy metabolism during perinatal hypoxia-ischemia: a 31P NMR study in unanesthetized immature rats. Neurosci Lett 144: 103–106.
Williams GD, Dardzinski BJ, Buckalew AR, Smith MB 1997 Modest hypothermia preserves cerebral energy metabolism during hypoxia-ischemia and correlates with brain damage: a 31P nuclear magnetic resonance study in unanesthetized neonatal rats. Pediatr Res 42: 700–708.
Vannucci RC, Yager JY 1992 Glucose, lactic acid, and perinatal hypoxic-ischemic brain damage. Pediatr Neurol 8: 3–12.
Vannucci RC 1992 Cerebral carbohydrate and energy metabolism in perinatal hypoxic- ischemic brain damage. Brain Pathol 2: 229–234.
Hall R, Murdoch J 1990 Brain protection: physiological and pharmacological considerations. Can J Anaesth 37: 762–777.
Vannucci RC 1990 Experimental biology of cerebral hypoxia-ischemia: relation to perinatal brain damage. Pediatr Res 27: 317–326.
Rehncrona SRI, Siesjo BK 1981 Brain lactic acidosis and ischemic cell damage: 1. J Cereb Blood Flow Metab 1: 297–311.
Palmer C, Brucklacher RM, Christensen MA, Vannucci RC 1990 Carbohydrate and energy metabolism during the evolution of hypoxic-ischemic brain damage in the immature rat. J Cereb Blood Flow Metab 10: 227–235.
Vannucci RC, Vasta F, Vannucci SJ 1987 Cerebral metabolic responses of hyperglycemic immature rats to hypoxia-ischemia. Pediatr Res 21: 524–529.
Welsh FA, Vannucci RC, Brierley JB 1982 Columnar alterations of NADH fluorescence during hypoxia-ischemia in immature rat brain. J Cereb Blood Flow Metab 2: 221–228.
Rice JEd, Vannucci RC, Brierley JB 1981 The influence of immaturity on hypoxic-ischemic brain damage in the rat. Ann Neurol 9: 131–141.
Vannucci RC, Lyons DT, Vasta F 1988 Regional cerebral blood flow during hypoxia-ischemia in immature rats. Stroke 19: 245–250.
Murdoch J, Hall R 1990 Brain protection: physiological and pharmacological considerations. Can J Anaesth 37: 663–671.
Yager JY, Brucklacher RM, Vannucci RC 1991 Cerebral oxidative metabolism and redox state during hypoxia-ischemia and early recovery in immature rats. Am J Physiol 261:H1102–1108.
Voorhies TM, Rawlinson D, Vannucci RC 1986 Glucose and perinatal hypoxic-ischemic brain damage in the rat. Neurology 36: 1115–1118.
Yager JY, Brucklacher RM, Vannucci RC 1992 Cerebral energy metabolism during hypoxia-ischemia and early recovery in immature rats. Am J Physiol 262:H672–677.
Yager JY, Heitjan DF, Towfighi J, Vannucci RC 1992 Effect of insulin-induced and fasting hypoglycemia on perinatal hypoxic-ischemic brain damage. Pediatr Res 31: 138–142.
Moore TJ LA, Regen DM, Tarpley HL, Raines PL 1971 Brain glucose metabolism in the newborn rat. Am J Physiol 221: 1746–1753.
Krebs HA WD, Bates MW, Page MA, Hawkins RA 1971 The role of ketone bodies in caloric homeostasis. Adv Enzyme Regul 9: 387–409.
Hawkins RA WD, Krebs HA 1971 Ketone-body utilization by adult and suckling rat brain in vivo. Biochem J 122: 13–18.
Collins B 1995 The Effect of Ketonemia on Cerebral Energy Metabolism and Cerebral Damage in Immature Rats Exposed to Hypoxia-Ischemia: An In Vivo 31P NMRS Study. [thesis] Pennsylvania State University, Hershey, PA
Go KG, Prenen G, Korf J 1988 Protective effect of fasting upon cerebral hypoxic-ischemic injury. Metab Brain Dis 3: 257–263.
Marie CBA, Gueldry S, Bralet J 1990 Fasting prior to transient cerebral ischemia reduces delayed neuronal necrosis. Metab Brain Dis 5: 65–75.
Myers RE, Yamaguchi S 1977 Nervous system effects of cardiac arrest in monkeys. Arch Neurol 34: 65–74.
Holowach-Thurston JHR 1989 Ketonemia increases survival of young mice in anoxic as well as hypoxic environments. Pediatr Res 25: 326A
Eiger SM KJ, D'Alecy LG 1980 Hypoxic tolerance enhanced by B-hydroxybutyrate-glucagon in the mouse. Stroke 11: 513–517.
Gueldry S, Marie C, Rochette L, Bralet J 1990 Beneficial effect of 1,3-butanediol on cerebral energy metabolism and edema following brain embolization in rats. Stroke 21: 1458–1463.
Gueldry S, Bralet J 1994 Effect of 1,3-butanediol on cerebral energy metabolism. Metab Brain Dis 9: 171–181.
Lundy EF DJ, Luyck B, Zelenock GB, D'Alecy LG 1985 Reduction of neurologic deficit by 1,3-butanediol induced ketosis in Levine rats. Stroke 16: 855–860.
Author information
Authors and Affiliations
Additional information
Funding for this research was provided by National Institutes of Health program project grant 1P01HD30704–01A1.
Michael B. Smith, Ph.D, Center for NMR Research, Radiology MC H066, Penn State College of Medicine, Hershey Medical Center, 500 University Drive, Hershey, PA 17033, U.S.A.
Rights and permissions
About this article
Cite this article
Dardzinski, B., Smith, S., Towfighi, J. et al. Increased Plasma Beta-Hydroxybutyrate, Preserved Cerebral Energy Metabolism, and Amelioration of Brain Damage During Neonatal Hypoxia Ischemia with Dexamethasone Pretreatment. Pediatr Res 48, 248–255 (2000). https://doi.org/10.1203/00006450-200008000-00021
Received:
Accepted:
Issue date:
DOI: https://doi.org/10.1203/00006450-200008000-00021
This article is cited by
-
The effect of piglet vitality, birth order, and blood lactate on the piglet growth performances and preweaning survival
Porcine Health Management (2022)
-
Emerging Therapeutic Promise of Ketogenic Diet to Attenuate Neuropathological Alterations in Alzheimer’s Disease
Molecular Neurobiology (2020)
-
Augmentation of Normal and Glutamate-Impaired Neuronal Respiratory Capacity by Exogenous Alternative Biofuels
Translational Stroke Research (2013)
-
Neuroprotection by acetoacetate and β-hydroxybutyrate against NMDA-induced RGC damage in rat—possible involvement of kynurenic acid
Graefe's Archive for Clinical and Experimental Ophthalmology (2010)
-
Metabonomic phenotype and identification of “heart blood stasis obstruction pattern” and “qi and yin deficiency pattern” of myocardial ischemia rat models
Science in China Series C: Life Sciences (2009)