Abstract
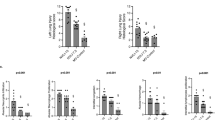
Nitric oxide (NO) production may depend on the uptake of l-arginine (l-arg), the substrate for NO synthase in inflammatory lung diseases. The cellular transport of l-arg occurs via the cationic amino acid transporters (CAT), and l-lysine (l-lys) competitively inhibits CAT. Neonatal pigs were treated with lipopolysaccharide (LPS) or vehicle for 4 h. LPS increased exhaled NO (exNO; 0.026 ± 0.003 to 0.046 ± 0.003 nmol · kg−1 · min−1; p < 0.005) and decreased mean systemic arterial blood pressure (89 ± 4 to 67 ± 4 mm Hg; p < 0.05), whereas vehicle did not affect exNO or mean systemic arterial blood pressure. The lungs were then isolated and perfused; exNO was greater in lungs from LPS-treated animals (0.08 ± 0.01 nmol/kg/min) than in lungs from vehicle-treated animals (0.05 ± 0.01 nmol · kg−1· min−1; p < 0.05). The addition of l-arg (0.3 mM) significantly (p < 0.05) increased exNO production in both groups of lungs (mean increase 0.04 ± 0.01 nmol · kg−1 · min−1 LPS-treated lungs, p < 0.05; mean increase 0.02 ± 0.01 nmol · kg−1 · min−1vehicle-treated lungs); however, l-arg decreased pulmonary vascular resistance (PVR) only in LPS-treated lungs (mean decrease 0.03 ± 0.01 mm Hg · ml−1 · kg−1 · min−1, p < 0.05). l-lys caused a dose-dependent decrease in exNO production and a dose-dependent increase in PVR in LPS-treated lungs. l-lys decreased exNO only at 30 mM and had no effect on PVR in vehicle-treated lungs. In four lungs each from vehicle-and LPS-treated animals, reverse transcriptase–PCR demonstrated CAT-2 mRNA only in LPS-treated animals. These results suggest that the increased NO production in the lungs from LPS-treated animals depends on the uptake of vascular l-arg.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Abbreviations
- bPAEC:
-
bovine pulmonary arterial endothelial cells
- CAT:
-
cationic amino acid transporter
- eNOS:
-
endothelial nitric oxide synthase
- exNO:
-
exhaled nitric oxide
- iNOS:
-
inducible nitric oxide synthase
- l-arg:
-
l-arginine
- l-lys:
-
l-lysine
- LPS:
-
lipopolysaccharide
- MAP:
-
mean systemic arterial blood pressure
- nNOS:
-
neuronal nitric oxide synthase
- NO:
-
nitric oxide
- NOS:
-
nitric oxide synthase
- Pa:
-
pulmonary arterial pressure
- PVR:
-
pulmonary vascular resistance
- RR:
-
respiratory rate
- TNF-α:
-
tumor necrosis factor-α
- Vt:
-
tidal volume
References
Aaron SD, Valenza F, Volgyesi G, Mullen JB, Slutsky AS, Stewart TE 1998 Inhibition of exhaled nitric oxide production during sepsis does not prevent lung inflammation. Crit Care Med 26: 309–314.
Kirkeboen KA, Strand OA 1999 The role of nitric oxide in sepsis. Acta Anesthesiol Scand 43: 275–288.
Stoclet JC, Martinez MC, Ohlmann P, Chasserot S, Schott C, Kleschyov AL, Schneider F, Andriantsitohaina R 1999 Induction of nitric oxide synthase and dual effects of nitric oxide and cyclooxygenase products in regulation of arterial contraction in human septic shock. Circulation 100: 107–112.
Vincent JL, Zhang H, Szabo C, Preiser JC 2000 Effects of nitric oxide in septic shock. Am J Respir Crit Care Med 161: 1781–1785.
Bogle RG, Baydoun AR, Pearson JD, Moncada S, Mann GE 1992 L-arginine transport is increased in macrophages generating nitric oxide. Biochem J 284: 15–18.
Gill DJ, Low BC, Grigor MR 1996 Interleukin-1 and tumor necrosis factor stimulate the CAT-2 gene of the L-arginine transporter in cultured vascular smooth muscle cells. J Biol Chem 271: 11280–11283.
Nelin LD, Nash HE, Chicoine LG 2001 Cytokine treatment increases arginine metabolism and uptake in bovine pulmonary arterial endothelial cells. Am J Physiol Lung Cell Mol Physiol 281:L1232–L1239.
Nelin LD, Krenz GS, Chicoine LG, Dawson CA, Schapira RM 2002 L-arginine uptake and metabolism following in vivo silica exposure in rat lungs. Am J Respir Cell Mol Biol 26: 348–355.
Simmons WW, Closs EI, Cunningham JM, Smith TW, Kelly RA 1996 Cytokines and insulin induce cationic amino acid transporter CAT expression in cardiac myocytes. J Biol Chem 271: 11694–11702.
Stathopulos PB, Lu X, Shen J, Scott JA, Hammond JR, McCormack DG, Arnold JM, Feng Q 2001 Increased L-arginine uptake and inducible nitric oxide synthase activity in aortas of rats with heart failure. Am J Physiol Heart Circ Physiol 280:H895–H867.
Wang H, Kavanaugh MP, North RA, Kavat D 1991 Cell surface receptor for ecotropic murine retroviruses is a basic amino acid transporter. Nature 352: 729–731.
Devés R, Boyd CAR 1998 Transporters of cationic amino acids in animal cells: discovery, structure and function. Physiol Rev 78: 487–545.
Block ER, Herrera H, Couch M 1995 Hypoxia inhibits L-arginine uptake by pulmonary artery endothelial cells. Am J Physiol 269:L574–L580.
Hattori Y, Kasai K, Gross SS 1999 Cationic amino acid transporter gene expression in cultured vascular smooth muscle cells and in rats. Am J Physiol 276:H2020–H2028.
Fike CD, Kaplowitz MR, Rehorst-Paea LA, Nelin LD 2000 L-arginine increases nitric oxide production in isolated lungs of chronically hypoxic newborn pigs. J Appl Physiol 88: 1797–1803.
Nelin LD, Thomas CJ, Dawson CA 1996 The effect of hypoxia on nitric oxide production in the neonatal pig lung. Am J Physiol 271:H8–H14.
Nelin LD, Dawson CA 1993 The effect of Nω−nitro-L-arginine methylester on hypoxic vasoconstriction in the neonatal pig lung. Pediatr Res 34: 349–353.
Matejovic M, Radermacher P, Tugtekin I, Stehr A, Theisen M, Vogt J, Wachter U, Ploner F, Georgieff M, Trager K 2001 Effects of selective iNOS inhibition on gut and liver O2-exchange and energy metabolism during hyperdynamic porcine endotoxemia. Shock 16: 203–210.
Fujii Y, Goldberg P, Hussain SN 1998 Intrathoracic and extrathoracic sources of exhaled nitric oxide in porcine endotoxemic shock. Chest 114: 569–576.
Kaftan HA, Clark PL, Norberg M, Garg U, Thibeault DW, Truog WE 2003 Endogenous production of nitric oxide in endotoxemic piglets. Biol Neonate 83: 42–48.
Neumann P, Berglung JE, Mondéjar EF, Magnusson A, Hedenstierna G 1998 Dynamics of lung collapse and recruitment during prolonged breathing in porcine lung injury. J Appl Physiol 85: 1533–1543.
Müller G, Steinbach G, Berndt A, Köhler 2002 Effects of various applications of lipopolysaccharides on blood parameters of pigs. J Vet Med B Infect Dis Vet Public Health 49: 429–437.
Allman KG, Stoddart AP, Kennedy MM, Young JD 1996 L-arginine augments nitric oxide production and mesenteric blood flow in ovine endotoxemia. Am J Physiol 271:H1296–H1301.
Gundersen Y, Saetre T, Scholz T, Hovig T, Lilleaasen P, Aasen AO 1999 Selective inhibition of inducible nitric oxide synthase maintains haemodynamic stability without untoward consequences for hepatic function or morphology. Eur J Surg 165: 1167–1174.
Fischer LG, Horstman DJ, Hahnenkamp K, Kechner NE, Rich GF 1999 Selective iNOS inhibition attenuates acetylcholine-and bradykinin-induced vasoconstriction in lipopolysaccharide-exposed rat lungs. Anesthesiology 91: 1724–1732.
Lee RP, Wang D, Kao SJ, Chen HI 2001 The lung is the major site that produces nitric oxide to induce pulmonary oedema in endotoxin shock. Clin Exp Pharmacol Physiol 28: 315–320.
Resta TC, O'Donaughy TL, Earley S, Chicoine LG, Walker BR 1999 Unaltered vasoconstrictor responsiveness after iNOS inhibition in lungs from chronically hypoxic rats. Am J Physiol Lung Cell Mol Physiol 276:L122–L130.
Fineman JR, Chang R, Soifer SJ 1991 L-arginine, a precursor of EDRF in vitro, produces pulmonary vasodilation in lambs. Am J Physiol 261:H1563–H1569.
Pearson DL, Dawling S, Walsh WF, Haines JL, Christman BW, Bazyk A, Scott N, Summar ML 2001 Neonatal pulmonary hypertension-urea cycle intermediates, nitric oxide production, and carbamoyl-phosphate synthetase function. N Engl J Med 344: 1832–1838.
McCaffrey MJ, Bose CL, Reiter PD, Stiles AD 1995 Effect of L-arginine infusion on infants with persistent pulmonary hypertension of the newborn. Biol Neonate 67: 240–243.
Allman KG, Stoddart AP, Young JD 1998 Effect of L-lysine on nitric oxide production in ovine endotoxaemia. Br J Anaesth 81: 188–192.
Liaudet L, Gnaegi A, Rosselet A, Mankert M, Boulat O, Perret C, Feihl F 1997 Effect of L-lysine on nitric oxide overproduction in endotoxic shock. Br J Pharmacol 122: 742–748.
Fike CD, Kaplowitz MR, Thomas CJ, Nelin LD 1998 Chronic hypoxia decreases nitric oxide production and endothelial nitric oxide synthase in newborn pig lungs. Am J Physiol 274:L517–L526.
Turley JE, Nelin LD, Kaplowitz MR, Zhang Y, Fike CD 2003 Exhaled NO is reduced at an early stage of hypoxia-induced pulmonary hypertension in newborn piglets. Am J Physiol Lung Cell Mol Physiol 284:L489–L500.
Carlin RE, Ferrario L, Boyd JT, Camporesi EM, McGraw DJ, Hakim TS 1997 Determinants of nitric oxide in exhaled gas in the isolated rabbit lung. Am J Respir Crit Care Med 155: 922–927.
Ogasa T, Nakano H, Ide H, Yamamato Y, Sasaki N, Osanai S, Akiba Y, Kikuchi K, Iwamoto J 2001 Flow-mediated release of nitric oxide in isolated, perfused rabbit lungs. J Appl Physiol 91: 363–370.
Vaughn DJ, Brogan TV, Kerr ME, Deem S, Luchtel DL, Swenson ER 2003 Contributions of nitric oxide synthase isozymes to exhaled nitric oxide and hypoxic pulmonary vasoconstriction in rabbit lungs. Am J Physiol Lung Cell Mol Physiol 284:L834–L843.
Nicholson B, Manner CK, Kleeman J, MacLeod CL 2001 Sustained nitric oxide production in macrophages requires the arginine transporter CAT2. J Biol Chem 276: 15881–15885.
Acknowledgements
We thank Kelly Billings for excellent technical assistance and Prof. Benjimen R. Walker for assistance with study design.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported by a Grant-in-Aid from the American Heart Association, Desert Mountain Affiliate (L.D.N.) and National Heart, Lung and Blood Institute Grant HL-004050 (L.G.C.).
Rights and permissions
About this article
Cite this article
Carter, B., Chicoine, L. & Nelin, L. l-Lysine Decreases Nitric Oxide Production and Increases Vascular Resistance in Lungs Isolated from Lipopolysaccharide-Treated Neonatal Pigs. Pediatr Res 55, 979–987 (2004). https://doi.org/10.1203/01.pdr.0000127722.55965.b3
Received:
Accepted:
Issue date:
DOI: https://doi.org/10.1203/01.pdr.0000127722.55965.b3
This article is cited by
-
Shuttle between arginine and lysine: influence on cancer immunonutrition
Amino Acids (2023)
-
Lysine nutrition in swine and the related monogastric animals: muscle protein biosynthesis and beyond
SpringerPlus (2015)
-
L-lysine as adjunctive treatment in patients with schizophrenia: a single-blinded, randomized, cross-over pilot study
BMC Medicine (2011)
-
The amino acid l-lysine blocks the disruptive effect of phencyclidine on prepulse inhibition in mice
Psychopharmacology (2007)
-
Plasma arginine correlations in trauma and sepsis
Amino Acids (2006)