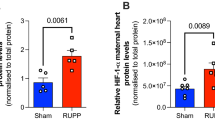
Abstract
In humans, infants who are born small have been reported to have higher blood pressure in adulthood than do larger infants. This suggests that factors in the intrauterine environment that affect fetal growth can program the individual for hypertension later in life. The present study determined whether there is a similar, naturally occurring relationship between birth weight and adult blood pressure in rats. Female Sprague-Dawley rats bred in our colony were fed a normal diet during pregnancy. On the day of delivery, any pups that weighed <90% of the mean pup weight for the litter were identified as runts. For each runt, a sex-matched littermate of normal weight was also identified and assigned to this study. These pairs were chronically instrumented at ∼20 wk of age. Mean arterial pressure was significantly higher in runt male and female offspring compared with their normal birth weight littermates (males: 149 ± 7, runts versus 129 ± 4 mm Hg, controls; females: 128 ± 1, runts versus 119 ± 2 mm Hg, controls). Although the runts had smaller body weights at study than did their littermate controls, the kidney-to-body weight ratio and renal function normalized to kidney or body weight were not different. These studies indicate that adult blood pressure is related to birth weight in rats, as it is in humans. The relative hypertension in runt animals is not due to gross differences in renal function but may be related to more subtle renal structural and/or functional differences.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Abbreviations
- ERPF:
-
effective renal plasma flow
- PAH:
-
para-aminohippurate
References
Barker DJ, Bull AR, Osmond C, Simmonds SJ 1990 Fetal and placental size and risk of hypertension in adult life. BMJ 301: 259–262
Curhan GC, Chertow GM, Willett WC, Spiegelman D, Colditz GA, Manson JE, Speizer FE, Stampfer MJ 1996 Birth weight and adult hypertension and obesity in women. Circulation 94: 1310–1315
Curhan GC, Willett WC, Rimm EB, Spiegelman D, Ascherio AL, Stampfer MJ 1996 Birth weight and adult hypertension, diabetes mellitus, and obesity in US men. Circulation 94: 3246–3250
Law CM, de Swiet M, Osmond C, Fayers PM, Barker DJ, Cruddas AM, Fall CH 1993 Initiation of hypertension in utero and its amplification throughout life. BMJ 306: 24–27
Thame M, Osmond C, Wilks RJ, Bennett FI, McFarlane-Anderson N, Forrester TE 2000 Blood pressure is related to placental volume and birth weight. Hypertension 35: 662–667
Langley SC, Jackson AA 1994 Increased systolic blood pressure in adult rats induced by fetal exposure to maternal low protein diets. Clin Sci (Lond) 86: 217–222
Woods LL, Ingelfinger JR, Nyengaard JR, Rasch R 2001 Maternal protein restriction suppresses the newborn renin-angiotensin system and programs adult hypertension in rats. Pediatr Res 49: 460–467
Woodall SM, Johnston BM, Breier BH, Gluckman PD 1996 Chronic maternal undernutrition in the rat leads to delayed postnatal growth and elevated blood pressure of offspring. Pediatr Res 40: 438–443
Benediktsson R, Lindsay RS, Noble J, Seckl JR, Edwards CR 1993 Glucocorticoid exposure in utero: new model for adult hypertension. Lancet 341: 339–341
Levitt NS, Lindsay RS, Holmes MC, Seckl JR 1996 Dexamethasone in the last week of pregnancy attenuates hippocampal glucocorticoid receptor gene expression and elevates blood pressure in the adult offspring in the rat. Neuroendocrinology 64: 412–418
Woods LL, Weeks DA, Rasch R 2004 Programming of adult blood pressure by maternal protein restriction: role of nephrogenesis. Kidney Int 65: 1339–1348
Woods LL, Rasch R 1998 Perinatal ANGII programs adult blood pressure, glomerular number, and renal function in rats. Am J Physiol 275: R1593–R1599
Woods LL 1999 Neonatal uninephrectomy causes hypertension in adult rats. Am J Physiol 276: R974–R978
Woods LL, Weeks DA, Rasch R 2001 Hypertension after neonatal uninephrectomy in rats precedes glomerular damage. Hypertension 38: 337–342
Poore KR, Forhead AJ, Gardner DS, Giussani DA, Fowden AL 2002 The effects of birth weight on basal cardiovascular function in pigs at 3 months of age. J Physiol 539: 969–978
Waugh WH 1977 Photometry of inulin and polyfructosan by use of a cysteine/tryptophan reaction. Clin Chem 23: 639–645
Brun C 1951 A rapid method for the determination of para-aminohippuric acid in kidney function tests. J Lab Clin Med 37: 955–958
Huxley R, Neil A, Collins R 2002 Unravelling the fetal origins hypothesis: is there really an inverse association between birthweight and subsequent blood pressure?. Lancet 360: 659–665
Hanson MA, Hawkins P, Ozaki T, Steyn C, Matthews SG, Noakes D, Poston L 1999 Effects of experimental dietary manipulation during early pregnancy on cardiovascular and endocrine function in fetal sheep and young lambs. In: O'Brien PMS, Wheeler T, Barker DJP (eds) Fetal Programming: Influences on Development and Disease in Later Life. RCOG Press, London, pp 365–373
Persson E, Jansson T 1992 Low birth weight is associated with elevated adult blood pressure in the chronically catheterized guinea-pig. Acta Physiol Scand 145: 195–196
Robinson JS, McMillen C, Edwards L, Erwich JJHM, Roberts C, Kind KL, Owens JA The effect of maternal nutrition on growth and development before and after birth. In: O'Brien PMS, Wheeler T, Barker DJP (eds) Fetal Programming: Influences on Development and Disease in Later Life. RCOG Press, London, pp 217–230
Louey S, Cock ML, Stevenson KM, Harding R 2000 Placental insufficiency and fetal growth restriction lead to postnatal hypotension and altered postnatal growth in sheep. Pediatr Res 48: 808–814
Even MD, Laughlin MH, Krause GF, vom Saal FS 1994 Differences in blood flow to uterine segments and placentae in relation to sex, intrauterine location and side in pregnant rats. J Reprod Fertil 102: 245–252
Jones SE, Nyengaard JR, Flyvbjerg A, Bilous RW, Marshall SM 2001 Birth weight has no influence on glomerular number and volume. Pediatr Nephrol 16: 340–345
Tonkiss J, Trzcinska M, Galler JR, Ruiz-Opazo N, Herrera VL 1998 Prenatal malnutrition-induced changes in blood pressure: dissociation of stress and nonstress responses using radiotelemetry. Hypertension 32: 108–114
Phillips DI 2001 Fetal growth and programming of the hypothalamic-pituitary-adrenal axis. Clin Exp Pharmacol Physiol 28: 967–970
Ozaki T, Nishina H, Hanson MA, Poston L 2001 Dietary restriction in pregnant rats causes gender-related hypertension and vascular dysfunction in offspring. J Physiol 530: 141–152
Acknowledgements
We thank Kathryn Parent and Melinda Davison for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
These studies were supported by grants R01HD041074 and P01HD34430 from the National Institute of Child Health and Human Development, HL070132 from the National Heart, Lung, and Blood Institute, and a Grant-in-Aid from the American Heart Association (all LLW).
Rights and permissions
About this article
Cite this article
Woods, L., Weeks, D. Naturally Occurring Intrauterine Growth Retardation and Adult Blood Pressure in Rats. Pediatr Res 56, 763–767 (2004). https://doi.org/10.1203/01.PDR.0000142589.08246.77
Received:
Accepted:
Issue date:
DOI: https://doi.org/10.1203/01.PDR.0000142589.08246.77
This article is cited by
-
Autonomic dysfunction in programmed hypertension
Journal of Human Hypertension (2019)
-
Genetic and Environmental Contributions to Variation in the Posterior Communicating Collaterals of the Circle of Willis
Translational Stroke Research (2019)
-
Ambulatory blood pressure monitoring in healthy children with parental hypertension
Pediatric Nephrology (2009)
-
Growth restriction before or after birth reduces nephron number and increases blood pressure in male rats
Kidney International (2008)