Abstract
Background:
Perinatal exposure to infectious agents with associated maternal immune activation (MIA) leads to neuroanatomical and behavioral dysregulation reminiscent of autism spectrum disorders. Persistent microglial activation as well as increased choline acetyltransferase (ChAT) activity in the basal forebrain (BF) are characteristic of autistic subjects. Previous studies have shown that medium from activated microglia promotes cholinergic differentiation of precursors in the BF. We sought to determine whether MIA in vivo would lead to a similar effect on developing BF neurons.
Methods:
Pregnant mice were treated with the viral mimic polyinosinic–polycytidylic acid (poly(I:C)) or saline.
Results:
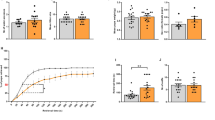
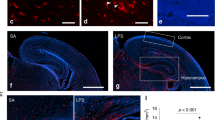
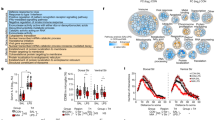
Poly(I:C) treatment resulted in increased production of cytokines and chemokines in fetal microglia and increased ChAT activity and cholinergic cell number in the perinatal BF. Whether microglial activation causes these changes is unclear. Examination of fetal brains from mice lacking interleukin-6 (IL-6 KOs) revealed an elevation in non–microglial-derived cytokines and chemokines over wild-type controls. Moreover, IL-6 KO offspring showed an elevation of ChAT activity even in the absence of poly(I:C) administration.
Conclusion:
These data suggest that elevations in cytokines and/or chemokines caused either by maternal poly(I:C) administration or by the absence of IL-6 are associated with alterations in cholinergic development in the BF.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Wang K, Zhang H, Ma D, et al. Common genetic variants on 5p14.1 associate with autism spectrum disorders. Nature 2009;459:528–33.
Glessner JT, Wang K, Cai G, et al. Autism genome-wide copy number variation reveals ubiquitin and neuronal genes. Nature 2009;459:569–73.
Lipkin WI, Hornig M . Microbiology and immunology of autism spectrum disorders. Novartis Found Symp 2003;251:129–43; discussion 144–8, 281–97.
Hagberg H, Mallard C . Effect of inflammation on central nervous system development and vulnerability. Curr Opin Neurol 2005;18:117–23.
Macêdo DS, Araújo DP, Sampaio LR, et al. Animal models of prenatal immune challenge and their contribution to the study of schizophrenia: a systematic review. Braz J Med Biol Res 2012;45:179–86.
Pardo CA, Eberhart CG . The neurobiology of autism. Brain Pathol 2007;17:434–47.
Croonenberghs J, Bosmans E, Deboutte D, Kenis G, Maes M . Activation of the inflammatory response system in autism. Neuropsychobiology 2002;45:1–6.
Patterson PH . Maternal infection: window on neuroimmune interactions in fetal brain development and mental illness. Curr Opin Neurobiol 2002;12:115–8.
Ratnayake U, Quinn TA, Castillo-Melendez M, Dickinson H, Walker DW . Behaviour and hippocampus-specific changes in spiny mouse neonates after treatment of the mother with the viral-mimetic Poly I:C at mid-pregnancy. Brain Behav Immun 2012;26:1288–99.
Meyer U, Murray PJ, Urwyler A, Yee BK, Schedlowski M, Feldon J . Adult behavioral and pharmacological dysfunctions following disruption of the fetal brain balance between pro-inflammatory and IL-10-mediated anti-inflammatory signaling. Mol Psychiatry 2008;13:208–21.
Fatemi SH, Earle J, Kanodia R, et al. Prenatal viral infection leads to pyramidal cell atrophy and macrocephaly in adulthood: implications for genesis of autism and schizophrenia. Cell Mol Neurobiol 2002;22:25–33.
Rousset CI, Chalon S, Cantagrel S, et al. Maternal exposure to LPS induces hypomyelination in the internal capsule and programmed cell death in the deep gray matter in newborn rats. Pediatr Res 2006;59:428–33.
Shi L, Fatemi SH, Sidwell RW, Patterson PH . Maternal influenza infection causes marked behavioral and pharmacological changes in the offspring. J Neurosci 2003;23:297–302.
Jonakait GM . The effects of maternal inflammation on neuronal development: possible mechanisms. Int J Dev Neurosci 2007;25:415–25.
Vargas DL, Nascimbene C, Krishnan C, Zimmerman AW, Pardo CA . Neuroglial activation and neuroinflammation in the brain of patients with autism. Ann Neurol 2005;57:67–81.
Zimmerman AW, Jyonouchi H, Comi AM, et al. Cerebrospinal fluid and serum markers of inflammation in autism. Pediatr Neurol 2005;33:195–201.
Morgan JT, Chana G, Pardo CA, et al. Microglial activation and increased microglial density observed in the dorsolateral prefrontal cortex in autism. Biol Psychiatry 2010;68:368–76.
Mazzoni IE, Kenigsberg RL . Microglia from the developing rat medial septal area can affect cholinergic and GABAergic neuronal differentiation in vitro. Neuroscience 1997;76:147–57.
Jonakait GM, Luskin MB, Wei R, Tian XF, Ni L . Conditioned medium from activated microglia promotes cholinergic differentiation in the basal forebrain in vitro. Dev Biol 1996;177:85–95.
Ni L, Acevedo G, Muralidharan B, Padala N, To J, Jonakait GM . Toll-like receptor ligands and CD154 stimulate microglia to produce a factor(s) that promotes excess cholinergic differentiation in the developing rat basal forebrain: implications for neurodevelopmental disorders. Pediatr Res 2007;61:15–20.
Perry EK, Lee ML, Martin-Ruiz CM, et al. Cholinergic activity in autism: abnormalities in the cerebral cortex and basal forebrain. Am J Psychiatry 2001;158:1058–66.
Bauman ML, Kemper TL . Neuroanatomic observations of the brain in autism: a review and future directions. Int J Dev Neurosci 2005;23:183–7.
Cai Z, Pan ZL, Pang Y, Evans OB, Rhodes PG . Cytokine induction in fetal rat brains and brain injury in neonatal rats after maternal lipopolysaccharide administration. Pediatr Res 2000;47:64–72.
Liverman CS, Kaftan HA, Cui L, et al. Altered expression of pro-inflammatory and developmental genes in the fetal brain in a mouse model of maternal infection. Neurosci Lett 2006;399:220–5.
Kannan S, Saadani-Makki F, Muzik O, et al. Microglial activation in perinatal rabbit brain induced by intrauterine inflammation: detection with 11C-(R)-PK11195 and small-animal PET. J Nucl Med 2007;48:946–54.
Hutton LC, Castillo-Melendez M, Smythe GA, Walker DW . Microglial activation, macrophage infiltration, and evidence of cell death in the fetal brain after uteroplacental administration of lipopolysaccharide in sheep in late gestation. Am J Obstet Gynecol 2008;198:117.e1–11.
Smith SE, Li J, Garbett K, Mirnics K, Patterson PH . Maternal immune activation alters fetal brain development through interleukin-6. J Neurosci 2007;27:10695–702.
Lasala N, Zhou H . Effects of maternal exposure to LPS on the inflammatory response in the offspring. J Neuroimmunol 2007;189:95–101.
Garay PA, Hsiao EY, Patterson PH, McAllister AK . Maternal immune activation causes age- and region-specific changes in brain cytokines in offspring throughout development. Brain Behav Immun 2013;31:54–68.
Jonakait GM, Pratt L, Acevedo G, Ni L . Microglial regulation of cholinergic differentiation in the basal forebrain. Dev Neurobiol 2012;72:857–64.
Di Cristo G, Pizzorusso T, Cancedda L, Sernagor E . GABAergic circuit development and its implication for CNS disorders. Neural Plast 2011;2011:623705.
Wei H, Zou H, Sheikh AM, et al. IL-6 is increased in the cerebellum of autistic brain and alters neural cell adhesion, migration and synaptic formation. J Neuroinflammation 2011;8:52.
Samuelsson AM, Jennische E, Hansson HA, Holmäng A . Prenatal exposure to interleukin-6 results in inflammatory neurodegeneration in hippocampus with NMDA/GABA(A) dysregulation and impaired spatial learning. Am J Physiol Regul Integr Comp Physiol 2006;290:R1345–56.
Ashdown H, Dumont Y, Ng M, Poole S, Boksa P, Luheshi GN . The role of cytokines in mediating effects of prenatal infection on the fetus: implications for schizophrenia. Mol Psychiatry 2006;11:47–55.
Zaretsky MV, Alexander JM, Byrd W, Bawdon RE . Transfer of inflammatory cytokines across the placenta. Obstet Gynecol 2004;103:546–50.
Urakubo A, Jarskog LF, Lieberman JA, Gilmore JH . Prenatal exposure to maternal infection alters cytokine expression in the placenta, amniotic fluid, and fetal brain. Schizophr Res 2001;47:27–36.
Jung S, Aliberti J, Graemmel P, et al. Analysis of fractalkine receptor CX(3)CR1 function by targeted deletion and green fluorescent protein reporter gene insertion. Mol Cell Biol 2000;20:4106–14.
Crawley JN . What’s Wrong with My Mouse? Hoboken, NJ: Wiley, 2007.
Fonnum F . A rapid radiochemical method for the determination of choline acetyltransferase. J Neurochem 1975;24:407–9.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pratt, L., Ni, L., Ponzio, N. et al. Maternal inflammation promotes fetal microglial activation and increased cholinergic expression in the fetal basal forebrain: role of interleukin-6. Pediatr Res 74, 393–401 (2013). https://doi.org/10.1038/pr.2013.126
Received:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/pr.2013.126
This article is cited by
-
Dysregulated gene expression associated with inflammatory and translation pathways in activated monocytes from children with autism spectrum disorder
Translational Psychiatry (2022)
-
Maternal Mid-Gestation Cytokine Dysregulation in Mothers of Children with Autism Spectrum Disorder
Journal of Autism and Developmental Disorders (2022)
-
Maternal immune activation induces sustained changes in fetal microglia motility
Scientific Reports (2020)
-
Maternal Immune Activation Causes Schizophrenia-like Behaviors in the Offspring through Activation of Immune-Inflammatory, Oxidative and Apoptotic Pathways, and Lowered Antioxidant Defenses and Neuroprotection
Molecular Neurobiology (2020)
-
In utero electroporation induces cell death and alters embryonic microglia morphology and expression signatures in the developing hypothalamus
Journal of Neuroinflammation (2018)