Abstract
Background:
Clinical findings in children with premature adrenarche (PA) correlate only partly with circulating levels of adrenal androgens. It is not known whether the prepubertal low circulating concentrations of testosterone (T) and dihydrotestosterone, together with those of adrenal androgens, are capable of activating the androgen receptor.
Methods:
This cross-sectional study was performed at a university hospital. Circulating androgen bioactivity was measured in 67 prepubertal children with clinical signs of PA and 94 control children using a novel androgen bioassay.
Results:
Circulating androgen bioactivity was low in the PA and control children. In the subgroup of children (n = 28) with serum T concentration over the assay sensitivity (0.35 nmol/l) and a signal in the androgen bioassay, we found a positive correlation between androgen bioactivity and serum T (r = 0.50; P < 0.01) and the free androgen index (r = 0.61; P < 0.01) and a negative correlation with serum sex hormone–binding globulin concentration (r = −0.41; P < 0.05).
Conclusion:
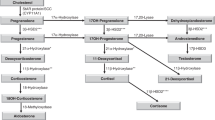
Peripheral metabolism of adrenal androgen precursors may be required for any androgenic effects in PA. However, the limitations in the sensitivity of the bioassay developed herein may hide some differences between the PA and control children.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Dhom G . The prepuberal and puberal growth of the adrenal (adrenarche). Beitr Pathol 1973;150:357–77.
Paterson WF, Ahmed SF, Bath L, et al. Exaggerated adrenarche in a cohort of Scottish children: clinical features and biochemistry. Clin Endocrinol (Oxf) 2010;72:496–501.
Utriainen P, Voutilainen R, Jääskeläinen J . Continuum of phenotypes and sympathoadrenal function in premature adrenarche. Eur J Endocrinol 2009;160:657–65.
Reed MJ, Purohit A, Woo LW, Newman SP, Potter BV . Steroid sulfatase: molecular biology, regulation, and inhibition. Endocr Rev 2005;26:171–202.
Hammer F, Subtil S, Lux P, et al. No evidence for hepatic conversion of dehydroepiandrosterone (DHEA) sulfate to DHEA: in vivo and in vitro studies. J Clin Endocrinol Metab 2005;90:3600–5.
Chen F, Knecht K, Birzin E, et al. Direct agonist/antagonist functions of dehydroepiandrosterone. Endocrinology 2005;146:4568–76.
Brinkmann AO . Molecular mechanisms of androgen action–a historical perspective. Methods Mol Biol 2011;776:3–24.
Chen F, Knecht K, Leu C, et al. Partial agonist/antagonist properties of androstenedione and 4-androsten-3beta,17beta-diol. J Steroid Biochem Mol Biol 2004;91:247–57.
Labrie F, Luu-The V, Labrie C, Simard J . DHEA and its transformation into androgens and estrogens in peripheral target tissues: intracrinology. Front Neuroendocrinol 2001;22:185–212.
Toda K, Simpson ER, Mendelson CR, Shizuta Y, Kilgore MW . Expression of the gene encoding aromatase cytochrome P450 (CYP19) in fetal tissues. Mol Endocrinol 1994;8:210–7.
Labrie F, Bélanger A, Simard J, Van Luu-The, Labrie C . DHEA and peripheral androgen and estrogen formation: intracinology. Ann N Y Acad Sci 1995;774:16–28.
Lappalainen S, Utriainen P, Kuulasmaa T, Voutilainen R, Jääskeläinen J . Androgen receptor gene CAG repeat polymorphism and X-chromosome inactivation in children with premature adrenarche. J Clin Endocrinol Metab 2008;93:1304–9.
Ibáñez L, Ong KK, Mongan N, et al. Androgen receptor gene CAG repeat polymorphism in the development of ovarian hyperandrogenism. J Clin Endocrinol Metab 2003;88:3333–8.
Petry CJ, Ong KK, Michelmore KF, et al. Associations between common variation in the aromatase gene promoter region and testosterone concentrations in two young female populations. J Steroid Biochem Mol Biol 2006;98:199–206.
Petry CJ, Ong KK, Wingate DL, de Zegher F, Ibáñez L, Dunger DB . Lack of association between common polymorphisms in the 17beta-hydroxysteroid dehydrogenase type V gene (HSD17B5) and precocious pubarche. J Steroid Biochem Mol Biol 2007;105:176–80.
Wang L, Salavaggione E, Pelleymounter L, Eckloff B, Wieben E, Weinshilboum R . Human 3beta-hydroxysteroid dehydrogenase types 1 and 2: Gene sequence variation and functional genomics. J Steroid Biochem Mol Biol 2007;107:88–99.
Hui XG, Akahira J, Suzuki T, et al. Development of the human adrenal zona reticularis: morphometric and immunohistochemical studies from birth to adolescence. J Endocrinol 2009;203:241–52.
Rege J, Rainey WE . The steroid metabolome of adrenarche. J Endocrinol 2012;214:133–43.
Raivio T, Palvimo JJ, Dunkel L, Wickman S, Jänne OA . Novel assay for determination of androgen bioactivity in human serum. J Clin Endocrinol Metab 2001;86:1539–44.
Paris F, Servant N, Térouanne B, Sultan C . Evaluation of androgenic bioactivity in human serum by recombinant cell line: preliminary results. Mol Cell Endocrinol 2002;198:123–9.
Rodriguez PC, Yee DJ, Sames D . Expanding the use of fluorogenic enzyme reporter substrates to imaging metabolic flux changes: the activity measurement of 5a-steroid reductase in intact mammalian cells. ACS Chem Biol 2010;5:1045–52.
Vermeulen A, Verdonck L, Kaufman JM . A critical evaluation of simple methods for the estimation of free testosterone in serum. J Clin Endocrinol Metab 1999;84:3666–72.
Rosenfield RL . Normal and almost normal precocious variations in pubertal development premature pubarche and premature thelarche revisited. Horm Res 1994;41:Suppl 2:7–13.
Vottero A, Capelletti M, Giuliodori S, et al. Decreased androgen receptor gene methylation in premature pubarche: a novel pathogenetic mechanism? J Clin Endocrinol Metab 2006;91:968–72.
Labrie F, Luu-The V, Lin SX, et al. The key role of 17 beta-hydroxysteroid dehydrogenases in sex steroid biology. Steroids 1997;62:148–58.
Nakamura Y, Hornsby PJ, Casson P, et al. Type 5 17beta-hydroxysteroid dehydrogenase (AKR1C3) contributes to testosterone production in the adrenal reticularis. J Clin Endocrinol Metab 2009;94:2192–8.
Rijk JC, Bovee TF, Peijnenburg AA, Groot MJ, Rietjens IM, Nielen MW . Bovine liver slices: A multifunctional in vitro model to study the prohormone dehydroepiandrosterone (DHEA). Toxicol In Vitro 2012;26:1014–21.
Hero M, Jänne OA, Näntö-Salonen K, Dunkel L, Raivio T . Circulating antiandrogenic activity in children with congenital adrenal hyperplasia during peroral flutamide treatment. J Clin Endocrinol Metab 2005;90:5141–5.
Kamrath C, Hochberg Z, Hartmann MF, Remer T, Wudy SA . Increased activation of the alternative “backdoor” pathway in patients with 21-hydroxylase deficiency: evidence from urinary steroid hormone analysis. J Clin Endocrinol Metab 2012;97:E367–75.
Heinlein CA, Chang C . Androgen receptor (AR) coregulators: an overview. Endocr Rev 2002;23:175–200.
Siiteri PK, Murai JT, Hammond GL, Nisker JA, Raymoure WJ, Kuhn RW . The serum transport of steroid hormones. Recent Prog Horm Res 1982;38:457–510.
Todorova B, Salonen M, Jääskeläinen J, et al. Adrenocortical hormonal activity in 20-year-old subjects born small or appropriate for gestational age. Horm Res Paediatr 2012;77:298–304.
Palvimo JJ, Reinikainen P, Ikonen T, Kallio PJ, Moilanen A, Jänne OA . Mutual transcriptional interference between RelA and androgen receptor. J Biol Chem 1996;271:24151–6.
Adeyemo O, Kallio PJ, Palvimo JJ, Kontula K, Jänne OA . A single-base substitution in exon 6 of the androgen receptor gene causing complete androgen insensitivity: the mutated receptor fails to transactivate but binds to DNA in vitro. Hum Mol Genet 1993;2:1809–12.
Makkonen H, Jääskeläinen T, Rytinki MM, Palvimo JJ . Analysis of androgen receptor activity by reporter gene assays. Methods Mol Biol 2011;776:71–80.
Saari A, Sankilampi U, Hannila ML, Kiviniemi V, Kesseli K, Dunkel L . New Finnish growth references for children and adolescents aged 0 to 20 years: Length/height-for-age, weight-for-length/height, and body mass index-for-age. Ann Med 2011;43:235–48.
Acknowledgements
Alise Tapio is thanked for her careful assistance in the laboratory studies.
Author information
Authors and Affiliations
Corresponding author
PowerPoint slides
Rights and permissions
About this article
Cite this article
Liimatta, J., Laakso, S., Utriainen, P. et al. Serum androgen bioactivity is low in children with premature adrenarche. Pediatr Res 75, 645–650 (2014). https://doi.org/10.1038/pr.2014.21
Received:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/pr.2014.21
This article is cited by
-
Association of dehydroepiandrosterone sulfate, birth size, adiposity and cardiometabolic risk factors in 7-year-old children
Pediatric Research (2022)
-
11-Oxygenated androgens in health and disease
Nature Reviews Endocrinology (2020)