Abstract
Background:
Congenital disorders of glycosylation (CDG) patients share a basic feature of protein hypoglycosylation. Activity of growth factors and their receptors, glycoproteins playing a pivotal role during child development, remains unexplored in CDG patients.
Methods:
Peripheral blood lymphocytes (PBL) isolated from 9 CDG patients and 12 healthy controls were cultured in the presence of fetal bovine serum (FBS), platelet-derived growth factor (PDGF), basic fibroblast growth factor (bFGF), and insulin-like growth factor-1 (IGF-1), and BrdU incorporation was measured. Levels of plasma IGF-1 and PBL IGF-1 receptor (IGF-1R) and its glycosylation were detected using immunoassay and western blot.
Results:
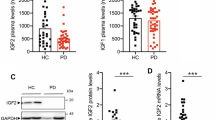
CDG patients showed significantly less (P < 0.01) serum-induced 5′-Bromo-2′-deoxyuridine (BrdU) incorporation in PBL than in controls. PDGF-/FGF-stimulated BrdU incorporation showed no difference in patients and controls, whereas IGF-1-induced DNA synthesis was significantly (P < 0.01) less in patients. Plasma IGF-1 levels and PBL IGF-1 receptor protein were significantly (P < 0.01) reduced in patients as compared to controls. IGF-1 receptor in PBL of all CDG patients had significantly (P < 0.01) reduced carbohydrate content when compared with control.
Conclusions:
These results show selective impairment of IGF-1-induced DNA synthesis in lymphocytes of CDG patients through decreased gene expression and hypoglycosylation of the IGF-1 receptor.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Jaeken J, Matthijs G. Congenital disorders of glycosylation: a rapidly expanding disease family. Annu Rev Genomics Hum Genet 2007;8:261–78.
Jaeken J. Congenital disorders of glycosylation. Ann N Y Acad Sci 2010;1214:190–8.
Woods AG, Woods CW, Snow TM. Congenital disorders of glycosylation. Adv Neonatal Care 2012;12:90–5.
Jaeken J. Congenital disorders of glycosylation (CDG): it’s (nearly) all in it! J Inherit Metab Dis 2011;34:853–8.
Makhseed N, Dhaunsi G, Jaeken J. Distinct features of congenital disorder of glycosylation type IIx in Kuwait: a case report. J Child Neurol 2012;27:222–4.
Seta N, Barnier A, Hochedez F, Besnard MA, Durand G. Diagnostic value of Western blotting in carbohydrate-deficient glycoprotein syndrome. Clin Chim Acta 1996;254:131–40.
Carchon H, Van Schaftingen E, Matthijs G, Jaeken J. Carbohydrate-deficient glycoprotein syndrome type IA (phosphomannomutase-deficiency). Biochim Biophys Acta 1999;1455:155–65.
Baker J, Liu JP, Robertson EJ, Efstratiadis A. Role of insulin-like growth factors in embryonic and postnatal growth. Cell 1993;75:73–82.
Savage MO. Insulin-like growth factors, nutrition and growth. World Rev Nutr Diet 2013;106:52–9.
Yakar S, Rosen CJ, Beamer WG, et al. Circulating levels of IGF-1 directly regulate bone growth and density. J Clin Invest 2002;110:771–81.
Kolarich D, Lepenies B, Seeberger PH. Glycomics, glycoproteomics and the immune system. Curr Opin Chem Biol 2012;16:214–20.
Perdue JF. The role of somatomedin/insulin-like growth factors and their receptors in skeletal growth and fetal development: a mini-review. Prog Clin Biol Res 1985; 132B: 405–413.
Werner H, LeRoith D. Insulin and insulin-like growth factor receptors in the brain: physiological and pathological aspects. Eur Neuropsychopharmacol 2014;24:1947–53.
LeRoith D, Yakar S. Mechanisms of disease: metabolic effects of growth hormone and insulin-like growth factor 1. Nat Clin Pract Endocrinol Metab 2007;3:302–10.
Aberg ND, Brywe KG, Isgaard J. Aspects of growth hormone and insulin-like growth factor-I related to neuroprotection, regeneration, and functional plasticity in the adult brain. ScientificWorldJournal 2006;6:53–80.
Mohan S, Kesavan C. Role of insulin-like growth factor-1 in the regulation of skeletal growth. Curr Osteoporos Rep 2012;10:178–86.
Lee C, Zhang F, Tang Z, Liu Y, Li X. PDGF-C: a new performer in the neurovascular interplay. Trends Mol Med 2013;19:474–86.
Shah P, Keppler L, Rutkowski J. A review of platelet derived growth factor playing pivotal role in bone regeneration. J Oral Implantol 2014;40:330–40.
Itoh N, Ornitz DM. Fibroblast growth factors: from molecular evolution to roles in development, metabolism and disease. J Biochem 2011;149:121–30.
Setia S, Sridhar MG. Changes in GH/IGF-1 axis in intrauterine growth retardation: consequences of fetal programming? Horm Metab Res 2009;41:791–8.
Gasperi M, Castellano AE. Growth hormone/insulin-like growth factor I axis in neurodegenerative diseases. J Endocrinol Invest 2010;33:587–91.
Siddle K. The insulin receptor and type I IGF receptor: comparison of structure and function. Prog Growth Factor Res 1992;4:301–20.
Mohanraj L, Oh Y. Targeting IGF-I, IGFBPs and IGF-I receptor system in cancer: the current and future in breast cancer therapy. Recent Pat Anticancer Drug Discov 2011;6:166–77.
Haller D, Blum S, Bode C, Hammes WP, Schiffrin EJ. Activation of human peripheral blood mononuclear cells by nonpathogenic bacteria in vitro: evidence of NK cells as primary targets. Infect Immun 2000;68:752–9.
Acknowledgements
Technical assistance of Nini Mathew is greatly acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dhaunsi, G. Receptor-mediated selective impairment of insulin-like growth factor-1 activity in congenital disorders of glycosylation patients. Pediatr Res 81, 526–530 (2017). https://doi.org/10.1038/pr.2016.96
Received:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/pr.2016.96
This article is cited by
-
Defective IGF-1 prohormone N-glycosylation and reduced IGF-1 receptor signaling activation in congenital disorders of glycosylation
Cellular and Molecular Life Sciences (2022)
-
Mosaicism of the UDP-Galactose transporter SLC35A2 in a female causing a congenital disorder of glycosylation: a case report
BMC Medical Genetics (2018)