Abstract
Background
Obesity has become a worldwide epidemic affecting millions of people. Obesity and associated health consequences tend to be complicated by diverse causes and multi-systemic involvement. Previous studies have investigated obesity induced by a single factor, such as a high-fat diet (HF) of typical energy-dense food and infection by an adipogenic virus, such as a widely studied human adenovirus serotype 36 (Ad-36). In this study, we hypothesized and investigated the synergistic effect of two causal factors, HF and Ad-36, in obesity induction.
Methods
The 7-week-old Wistar rats (n = 1214/group) were randomly divided into weight-matched groups and induced for obesity with mock-control, HF, Ad-36, or HF + Ad-36 for 8–30 weeks, and compared for obesity phenotype. A global transcriptomic RNA-Seq analysis was used to profile signature gene response pathways in ileal tissues from 8-week control and obese animals during this early phase of obesity induction.
Results
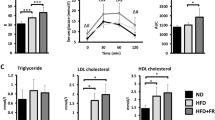
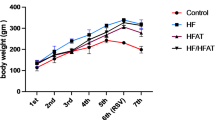
HF only and particularly co-administration of Ad-36 and HF (HF + Ad-36) induced significant obesity in rats (p < 0.05 or p < 0.005). Compared with either Ad-36 or HF alone, HF + Ad-36 treatment significantly aggravates obesity in rats regarding body weight (n = 12–14/group) and adiposity index (n = 6–7). Genome-wide transcriptomic analyses of intestinal tissues revealed signature genes on an inter-systemic scale, including many genes in the pathways of circadian rhythm and antiviral immunity focusing on IFN signaling.
Conclusions
Ad-36 exacerbated the induction of obesity in rats compared with those treated with HF alone. Gene-responsive pathways involved in circadian rhythm and antiviral immunity in ileal tissues were significantly (p < 0.05, and FDR < 0.01) regulated during the early phase of obesity induction. This study provided a co-factorial model for obesity induction and profiled molecular targets for further validation and molecular manipulation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Speakman JR. Evolutionary perspectives on the obesity epidemic: adaptive, maladaptive, and neutral viewpoints. Annu Rev Nutr. 2013;33:289–317.
Atkinson RL, Dhurandhar NV, Allison DB, Bowen RL, Israel BA, Albu JB, et al. Human adenovirus-36 is associated with increased body weight and paradoxical reduction of serum lipids. Int J Obes (Lond). 2005;29:281–6.
Ogden CL, Carroll MD, Kit BK, Flegal KM. Prevalence of childhood and adult obesity in the United States, 2011-2012. JAMA. 2014;311:806–14.
McAllister EJ, Dhurandhar NV, Keith SW, Aronne LJ, Barger J, Baskin M, et al. Ten putative contributors to the obesity epidemic. Crit Rev Food Sci Nutr. 2009;49:868–913.
Duca FA, Yue JT. Fatty acid sensing in the gut and the hypothalamus: in vivo and in vitro perspectives. Mol Cell Endocrinol. 2014;397:23–33.
Atkinson RL. Current status of the field of obesity. Trends Endocrinol Metab. 2014;25:283–4.
Genoni G, Prodam F, Marolda A, Giglione E, Demarchi I, Bellone S, et al. Obesity and infection: two sides of one coin. Eur J Pediatr. 2014;173:25–32.
Wigand R, Gelderblom H, Wadell G. New human adenovirus (candidate adenovirus 36), a novel member of subgroup D. Arch Virol. 1980;64:225–33.
Ponterio E, Gnessi L. Adenovirus 36 and obesity: an overview. Viruses. 2015;7:3719–40.
Lin WY, Dubuisson O, Rubicz R, Liu N, Allison DB, Curran JE, et al. Long-term changes in adiposity and glycemic control are associated with past adenovirus infection. Diabetes Care. 2013;36:701–7.
Dhurandhar NV, Israel BA, Kolesar JM, Mayhew GF, Cook ME, et al. Increased adiposity in animals due to a human virus. Int J Obes Relat Metab Disord. 2000;24:989–96.
Pasarica M, Loiler S, Dhurandhar NV. Acute effect of infection by adipogenic human adenovirus Ad36. Arch Virol. 2008;153:2097–102.
Dhurandhar NV, Whigham LD, Abbott DH, Schultz-Darken NJ, Israel BA, et al. Human adenovirus Ad-36 promotes weight gain in male rhesus and marmoset monkeys. J Nutr. 2002;132:3155–60.
Pasarica M, Shin AC, Yu M, Ou-Yang HM, Rathod M, Jen KL, et al. Human adenovirus 36 induces adiposity, increases insulin sensitivity, and alters hypothalamic monoamines in rats. Obesity (Silver Spring). 2006;14:1905–13.
Dhurandhar NV, Israel BA, Kolesar JM, Mayhew G, Cook ME, Atkinson RL. Transmissibility of adenovirus-induced adiposity in a chicken model. Int J Obes Relat Metab Disord. 2001;25:990–6.
Na HN, Hong YM, Kim J, Kim HK, Jo I, Nam JH. Association between human adenovirus-36 and lipid disorders in Korean schoolchildren. Int J Obes (Lond). 2010;34:89–93.
Tosh AK, Broy-Aschenbrenner A, El Khatib J, Ge B. Adenovirus-36 antibody status & BMI comparison among obese Missouri adolescents. Mol Med. 2012;109:402–3.
Yamada T, Hara K, Kadowaki T. Association of adenovirus 36 infection with obesity and metabolic markers in humans: a meta-analysis of observational studies. PLoS One. 2012;7:e42031.
Almgren M, Atkinson R, He J, Hilding A, Hagman E, Wolk A, et al. Adenovirus-36 is associated with obesity in children and adults in Sweden as determined by rapid ELISA. PLoS One. 2012;7:e41652.
Na HN, Kim J, Lee HS, Shim KW, Kimm H, Jee SH, et al. Association of human adenovirus-36 in overweight Korean adults. Int J Obes (Lond). 2012;36:281–5.
Parra-Rojas I, Del Moral-Hernández O, Salgado-Bernabé AB, Guzmán-Guzmán IP, Salgado-Goytia L, Muñoz-Valle JF. Adenovirus-36 seropositivity and its relation with obesity and metabolic profile in children. Int J Endocrinol. 2013;2013:463194.
Cakmakliogullari EK, Sanlidag T, Ersoy B, Akcali S, Var A, Cicek C. Are human adenovirus-5 and 36 associated with obesity in children? J Investig Med. 2014;62:821–4.
Aldhoon-Hainerová I, Zamrazilová H, Atkinson RL, Dušátková L, Sedláčková B, Hlavatý P, et al. Clinical and laboratory characteristics of 1179 Czech adolescents evaluated for antibodies to human adenovirus 36. Int J Obes (Lond). 2014;38:285–91.
Broderick MP, Hansen CJ, Irvine M, Metzgar D, Campbell K, Baker C, et al. Adenovirus 36 seropositivity is strongly associated with race and gender, but not obesity, among US military personnel. Int J Obes (Lond). 2010;34:302–8.
Voss JD, Burnett DG, Olsen CH, Haverkos HW, Atkinson RL. Adenovirus 36 antibodies associated with clinical diagnosis of overweight/obesity but not BMI gain: a military cohort study. J Clin Endocrinol Metab. 2014;99:E1708–1712.
Berger PK, Pollock NK, Laing EM, Warden SJ, Hill Gallant KM, et al. Association of adenovirus 36 infection with adiposity and inflammatory-related markers in children. J Clin Endocrinol Metab. 2014;99:3240–6.
Goossens VJ, deJager SA, Grauls GE, Gielen M, Vlietinck RF, Derom CA, et al. Lack of evidence for the role of human adenovirus-36 in obesity in a European cohort. Obesity (Silver Spring). 2011;19:220–1.
de La Serre CB, Ellis CL, Lee J, Hartman AL, Rutledge JC, Raybould HE. Propensity to high-fat diet-induced obesity in rats is associated with changes in the gut microbiota and gut inflammation. Am J Physiol Gastrointest Liver Physiol. 2010;299:G440–448.
Ding S, Chi MM, Scull BP, Rigby R, Schwerbrock NM, Magness S, et al. High-fat diet: bacteria interactions promote intestinal inflammation which precedes and correlates with obesity and insulin resistance in mouse. PLoS One. 2010;5:e12191.
Dhurandhar NV. A framework for identification of infections that contribute to human obesity. Lancet Infect Dis. 2011;11:963–9.
Greenway FL, Whitehouse MJ, Guttadauria M, Anderson JW, Atkinson RL, Fujioka K, et al. Rational design of a combination medication for the treatment of obesity. Obesity (Silver Spring). 2009;17:30–39.
Sang Y, Brichalli W, Rowland RR, Blecha F. Genome-wide analysis of antiviral signature genes in porcine macrophages at different activation statuses. PLoS One. 2014;9:e87613.
Sang Y, Schade A, Blecha F. Profiling ileal immune response genes in obese rats induced by high-fat diet and adenovirus infection (INM7P.432). J Immunol. 2014;192 Suppl 1:123.10–123.10.
Huh JY, Park YJ, Ham M, Kim JB. Crosstalk between adipocytes and immune cells in adipose tissue inflammation and metabolic dysregulation in obesity. Mol Cells. 2014;37:365–71.
Salzman NH, Hung K, Haribhai D, Chu H, Karlsson-Sjöberg J, Amir E, et al. Enteric defensins are essential regulators of intestinal microbial ecology. Nat Immunol. 2010;11:76–83.
Chahal JS, Gallagher C, DeHart CJ, Flint SJ. The repression domain of the E1B 55-kilodalton protein participates in countering interferon-induced inhibition of adenovirus replication. J Virol. 2013;87:4432–44.
Windheim M, Southcombe JH, Kremmer E, Chaplin L, Urlaub D, Falk CS, et al. A unique secreted adenovirus E3 protein binds to the leukocyte common antigen CD45 and modulates leukocyte functions. Proc Natl Acad Sci USA. 2013;110:E4884–4893.
Acknowledgements
We thank Dr. Barbara Lutjemeier, Mr. Andrew Schade, Ms. Lyndie Holmes, and Ms. Jessica Pearson for their excellent technical support.
Funding
This work was primarily supported by NIH P20-RR017686 (KSU), and in part by the USDA NIFA AFRI 2013-67015-21236 and USDA NIFA AFRI 2015-665 67015-23216 to YS.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Sang, Y., Shields, L.E., Sang, E.R. et al. Ileal transcriptome analysis in obese rats induced by high-fat diets and an adenoviral infection. Int J Obes 43, 2134–2142 (2019). https://doi.org/10.1038/s41366-019-0323-2
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41366-019-0323-2
This article is cited by
-
What we know and what we need to know about adenovirus 36-induced obesity
International Journal of Obesity (2020)
-
Obesity and Diabetes in an Arab population: Role of Adenovirus 36 Infection
Scientific Reports (2020)