Abstract
Background
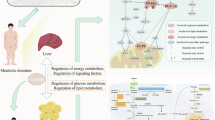
Metabolic dysregulation, a defining feature of obesity, disrupts essential signalling pathways involved in nutrient sensing and mitochondria homeostasis. The nuclear factor erythroid 2-related factor 2 (NRF-2) serves as a pivotal regulator of the cellular stress response, and recent studies have implicated it in the pathogenesis of obesity, diabetes, and metabolic syndrome. Curcumin, a polyphenolic compound derived from turmeric, has been identified as a potent activator of NRF-2. Evidence suggests curcumin impacts obesity and metabolic disorders by modulating gut microbiota composition, increasing energy expenditure, and regulating lipid metabolism. Orlistat, an anti-obesity drug, inhibits fat absorption in the gastrointestinal tract, but its side effects limits its broader use.
Objectives
The present study aims to investigate the potential synergetic effect of a hybrid combination between orlistat and curcumin. Additionally, we provide a detailed understanding of the molecular mechanisms through which this combination mitigates glucose-induced lipid accumulation in Caenorhabditis elegans, with a focus on the role of the skinhead 1 (SKN-1) transcription factor, an orthologue of NRF2.
Methods
We assessed the lipid accumulation and the changes in skn-1 transcriptional activity in C. elegans using confocal GFP-based detection, alongside mRNA expression analysis of genes from lipid metabolism and oxidative stress response in wild-type, QV225 and LD1 strains. Furthermore, we evaluated locomotion, chemotaxis and mitochondrial dynamics to enhance our understanding of the proposed molecular-based model.
Results
Our findings reveal that the orlistat/curcumin combination exerts an anti-obesogenic effect through SKN-1/NRF2-dependent regulation of conserved genes involved in carbohydrate and lipid metabolism in C. elegans. Moreover, the combination stimulates mitochondrial potential, further contributing to the observed synergistic effects.
Conclusion
The hybrid combination of orlistat and curcumin demonstrates significant anti-obesity activity by regulating nutrient-sensing pathways through SKN-1/NRF-2 modulation. This approach may allow for the reduction of orlistat dosage, thereby minimizing its adverse effects while maintaining its therapeutic efficacy.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All relevant data are within the manuscript. The data set generated and analysed during the current study also available from the corresponding author upon request.
References
Wen X, Zhang B, Wu B, Haitao X, Zehua L, Ruoyu L, et al. Signalling pathways in obesity: mechanisms and therapeutic interventions. Sign Transduct Target Ther. 2022;7:298.
Phelps NH, Singleton RK, Zhou B, Heap RA, Mishra A, Bennett JE, et al. Worldwide trends in underweight and obesity from 1990 to 2022: a pooled analysis of 3663 population-representative studies with 222 million children, adolescents, and adults. Lancet. 2024;403:1027–50.
Savova MS, Mihaylova LV, Tews D, Wabitsch M, Georgiev MI. Targeting PI3K/AKT signaling pathway in obesity. Biomed Pharmacother. 2023;159:114244.
Muller TD, Blüher M, Tschöp MH, DiMarchi RD. Anti-obesity drug discovery: advances and challenges. Nat Rev Drug Discov. 2022;21:201–23.
Zhao XY, Wang JQ, Neely GG, Shi YC, Wang QP. Natural compounds as obesity pharmacotherapies. Phytother Res. 2024;38:797–838.
Holmback U, Grudén S, Litorp H, Willhems D, Kuusk S, Alderborn G, et al. Effects of a novel weight-loss combination product containing orlistat and acarbose on obesity: A randomized, placebo-controlled trial. Obesity. 2022;30:2222–32.
Longo VD, Di Tano M, Mattson MP, Guidi N. Intermittent and periodic fasting, longevity and disease. Nat Aging. 2021;1:47–59.
Weir HJ, Yao P, Huynh FK, Escoubas CC, Goncalves RL, Burkewitz K, et al. Dietary restriction and AMPK increase lifespan via mitochondrial network and peroxisome remodelling. Cell Metab. 2017;26:884–96.
Madeo F, Carmona-Gutierrez D, Hofer SJ, Kroemer G. Caloric restriction mimetics against age-associated disease: targets, mechanisms, and therapeutic potential. Cell Metab. 2019;29:592–610.
Wei M, Brandhorlistat S, Shelehchi M, Mirzaei H, Cheng CW, Budniak J, et al. Fasting-mimicking diet and markers/risk factors for aging, diabetes, cancer, and cardiovascular disease. Sci Transl Med. 2017;15:eaai8700.
Atanasov AG, Zotchev SB, Dirsch VM, Orhan IE, Banach M, Rollinger JM, et al. Natural products in drug discovery: advances and opportunities. Nat Rev Drug Discov. 2021;20:200–16.
Thomas C, Erni R, Wu JY, Fischer F, Lamers G, Grigolon G, et al. A naturally occurring polyacetylene isolated from carrots promotes health and delays signatures of aging. Nat Commun. 2023;14:8142.
Bolger GT, Pucaj K, Minta YO, Sordillo P. Relationship between the in vitro efficacy, pharmacokinetics and in vivo efficacy of curcumin. Biochem Pharmacol. 2022;205:115251. https://doi.org/10.1016/j.bcp.2022.115251.
Vasileva LV, Saracheva KE, Ivanovska MV, Petrova AP, Marchev AS, Georgiev MI, et al. Antidepressant-like effect of salidroside and curcumin on the immunoreactivity of rats subjected to a chronic mild stress model. Food Chem Toxicol. 2018;121:604–11.
Shin JW, Chun KS, Kim DH, Kim SJ, Kim SH, Cho NC, et al. Curcumin induces stabilization of Nrf2 protein through Keap1 cysteine modification. Biochem Pharmacol. 2020;173:113820.
Ren L, Zhan P, Wang Q, Wang C, Liu Y, Yu Z, et al. Curcumin upregulates the Nrf2 system by repressing inflammatory signalling-mediated Keap1 expression in insulin-resistant conditions. Biochem Biophys Res Commun. 2019;514:691–8.
Li N, Yan X, Huang W, Chu M, Dong Y, Song H, et al. Curcumin protects against the age-related hearing loss by attenuating apoptosis and senescence via activating Nrf2 signaling in cochlear hair cells. Biochem Pharmacol. 2023;212:115575.
Wu X, Zhou X, Lai S, Liu J, Qi J. Curcumin activates Nrf2/HO-1 signaling to relieve diabetic cardiomyopathy injury by reducing ROS in vitro and in vivo. FASEB J. 2022;36:e22505.
Sampath C, Wilus D, Tabatabai M, Freeman ML, Gangula PR. Mechanistic role of antioxidants in rescuing delayed gastric emptying in high fat diet induced diabetic female mice. Biomed Pharmacoth. 2021;137:111370.
Vasileva LV, Savova MS, Amirova KM, Dinkova-Kostova AT, Georgiev MI. Obesity and NRF2-mediated cytoprotection: Where is the missing link? Pharmacol Res. 2020;156:104760.
Paek J, Jacqueline YL, Naracimhan SD, Nguyen TN, Glover-Cutter K, Robida-Stubbs S, et al. Mitochondrial SKN-1/Nrf mediates a conserved starvation response. Cell Metab. 2012;16:526–3.
Frankino PA, Siddiqi TF, Bolas T, Bar-Ziv R, Gildea HK, Zhang H, et al. SKN-1 regulates stress resistance downstream of amino catabolism pathways. iScience. 2022;25:104571.
Pang S, Lynn D, Lo J, Paek J, Curran SP. SKN-1 and Nrf2 couples proline catabolism with lipid metabolism during nutrient deprivation. Nat Commun. 2014;5:5048.
Blackwell TK, Steinbaugh MJ, Hourihan JM, Ewald CY, Isik M. SKN-1/Nrf, stress responses, and aging in Caenorhabditis elegans. Free Radic Biol Med. 2015;88:290–301.
Costa RM, Dias MC, Alves JV, Silva JLM, Rodrigues D, Silva JF, et al. Pharmacological activation of nuclear factor erythroid 2-related factor-2 prevents hyperglycemia-induced renal oxidative damage: Possible involvement of O-GlcNAcylation. Biochem Pharmacol. 2024;220:115982.
Steinbaugh MJ, Narasimhan SD, Robida-Stubbs S, Moronetti Mazzeo LE, Dreyfuss JM, Hourihan JM, et al. Lipid-mediated regulation of SKN-1/Nrf in response to germ cell absence. eLife. 2015;4:e07836.
Dou Y, Huang R, Li Q, Liu Y, Li Y, Chen H, et al. Oxyberberine, an absorbed metabolite of berberine, possess superior hypoglycemic effect via regulating the PI3K/Akt and Nrf2 signalling pathways. Biomed Pharmacother. 2021;137:111312.
Gao W, Wang W, Zhang J, Deng P, Hu J, Yang J, et al. Allicin ameliorates obesity comorbid depressive-like behaviors: involvement of the oxidative stress, mitochondrial function, autophagy, insulin resistance and NOX/Nrf2 imbalance in mice. Metab Brain Dis. 2019;34:1267–80.
Islam MT, Tuday E, Allen S, Kim J, Trott DW, Holland WL, et al. Senolytic drugs, dasatinib and quercetin, attenuate adipose tissue inflammation, and ameliorate metabolic function in old age. Aging Cell. 2023;22:e13767.
Novais EJ, Tran VA, Johnston SN, Darris KR, Roupas AJ, Sessions GA, et al. Long-term treatment with senolytic drugs dasatinib and quercetin ameliorates age-dependent intervertebral disc degeneration in mice. Nat Commun. 2021;12:5213.
Huang SL, Chang TC, Sun NK. Curcumin reduces paclitaxel resistance in ovarian carcinoma cells by upregulating SNIP1 and inhibiting NFκB activity. Biochem Pharmacol. 2023;212:115581.
Chen B, He Q, Chen C, Lin Y, Xiao J, Pan Z, et al. Combination of curcumin and catalase protects against chondrocyte injury and knee osteoarthritis progression by suppressing oxidative stress. Biomed Pharmacoth. 2023;168:115751.
Li WJ, Wang CW, Tao L, Yan YH, Zhang MJ, Liu ZX, et al. Insulin signalling regulates longevity through protein phosphorylation in Caenorhabditis elegans. Nat Commun. 2021;12:4568.
Shen P, Yue Y, Park Y. A living model for obesity and aging research: Caenorhabditis elegans. Crit Rev Food Sci Nutr. 2018;58:741–54.
Mora I, Puiggròs F, Serras F, Gil-Cardoso K, Escoté X. Emerging models for studying adipose tissue metabolism. Biochem Pharmacol. 20214;223:116123.
Yue Y, Hao G, Cho J, Park Y. Curcumin reduced fat accumulation in Caenorhabditis elegans. Curr Res Food Sci. 2021;4:551–6.
Bai J, Farias‐Pereira R, Zhang Y, Jang M, Park Y, Kim HK. C. elegans ACAT regulates lipolysis and its related lifespan in fasting through modulation of the genes in lipolysis and insulin/IGF-1 signalling. BioFactors. 2020;46:754–65.
Michaeli L, Spector E, Haeussler S, Carvalho CA, Grobe H, Abu-Shach UB, et al. ULP-2 SUMO protease regulates UPRmt and mitochondrial homeostasis in Caenorhabditis elegans. Free Radic Biol Med. 2024;214:19–27.
Navarro-Hortal MD, Romero-Márquez JM, Esteban-Muñoz A, Sánchez-González C, Rivas-García L, Llopis L, et al. Strawberry (Fragaria×ananassa cv. Romina) methanolic extract attenuates Alzheimer’s beta amyloid production and oxidative stress by SKN-1/NRF and DAF-16/FOXO mediated mechanisms in C. elegans. Food Chem. 2022;372:131272.
An JH, Blackwell TK. SKN-1 links C. elegans mesendodermal specification to a conserved oxidative stress response. Genes Dev. 2003;17:1882–93.
Aranaz P, Navarro-Herrera D, Romo-Hualde A, Miguel L, Vizmanos L, Milagro I. Phenolic compounds reduce the fat content in Caenorhabditis elegans by affecting lipogenesis, lipolysis, and different stress responses. Pharmaceuticals. 2020;13:355.
Savova MS, Todorova MN, Apostolov AG, Yahubyan GT, Georgiev MI. Betulinic acid counteracts the lipid accumulation in Caenorhabditis elegans by modulation of nhr-49 expression. Biomed Pharmacother. 2022;156:113862.
Mladenova SG, Todorova MN, Savova MS, Georgiev MI, Mihaylova LV. Maackiain mimics caloric restriction through aak-2-mediated lipid reduction in Caenorhabditis elegans. Int J Mol Sci. 2023;24:17442.
Todorova MN, Savova MS, Mihaylova LV, Georgiev MI. Icariin improves stress resistance and extends lifespan in Caenorhabditis elegans through hsf-1 and daf-2-driven hormesis. Int J Mol Sci. 2024;25:352.
Queiros L, Marques C, Pereira JL, Gonçalves FJM, Aschner M, Pereira P. Overview of chemotaxis behavior assays in Caenorhabditis elegans. Curr Protoc. 2021;1:e120.
Stuhr N, Nhan J, Hammerquist A, Van Camp B, Reoyo D, Curran S. Rapid lipid quantification in Caenorhabditis elegans by oil red O and Nile red staining. Bio Protoc. 2022;12:e4340.
Berry BJ, Vodičková A, Müller-Eigner A, Meng C, Ludwig C, Kaeberlein M, et al. Optogenetic rejuvenation of mitochondrial membrane potential extends C. elegans lifespan. Nat Aging. 2023;3:157–61.
Pai SA, Munshi RP, Panchal FH, Gaur IS, Mestry SN, Gursahani MS, et al. Plumbagin reduces obesity and nonalcoholic fatty liver disease induced by fructose in rats through regulation of lipid metabolism, inflammation and oxidative stress. Biomed Pharmacother. 2019;111:686–94.
Nunez S, Moliner C, Valero MS, Mustafa AM, Maggi F, Gómez-Rincón C, et al. Antidiabetic and anti-obesity properties of a polyphenol-rich flower extract from Tagetes erecta L. and its effects on Caenorhabditis elegans fat storages. J Physiol Biochem. 2023;79:427–40.
Lin C, Lin Y, Chen Y, Xu J, Li J, Cao Y, et al. Effects of: Momordica saponin extract on alleviating fat accumulation in Caenorhabditis elegans. Food Funct. 2019;10:3237–51.
Chen Y, Qin Q, Luo J, Dong Y, Lin C, Chen H, et al. Litchi flower essential oil balanced lipid metabolism through the regulation of DAF-2/IIS, MDT-15/SBP-1, and MDT-15/NHR-49 pathway. Front Nutr. 2022;9:934518.
Goh GYS, Winter JJ, Bhanshali F, Doering KRS, Lai R, Lee K, et al. NHR-49/HNF4 integrates regulation of fatty acid metabolism with a protective transcriptional response to oxidative stress and fasting. Aging Cell. 2018;17:e12743.
Li Y, Ding W, Li CY, Ying L. HLH-11 modulates lipid metabolism in response to nutrient availability. Nat Commun. 2020;11:5959.
Inoue H, Hisamoto N, AnJH, Oliveira RP, Nishida E, Blackwell TK, et al. The C. elegans p38 MAPK pathway regulates nuclear localization of the transcription factor SKN-1 in oxidative stress response. Genes Dev. 2005;19:2278–83.
Lee D, Jeong DE, Son HG, Yamaoka Y, Kim H, Seo K, et al. SREBP and MDT-15 protect C. elegans from glucose-induced accelerated aging by preventing accumulation of saturated fat. Genes Dev. 2015;29:2490–503.
Zhang Y, Lanjuin A, Chowdhury SR, Mistry M, Silva-García CG, Weir HJ, et al. Neuronal TORC1 modulates longevity via AMPK and cell nonautonomous regulation of mitochondrial dynamics in C. elegans. eLife. 2019;8:e49158.
Bakar MHA, Shariff KA, Tan JS, Lee LK. Celastrol attenuates inflammatory responses in adipose tissues and improves skeletal muscle mitochondrial functions in high fat diet-induced obese rats via upregulation of AMPK/SIRT1 signalling pathways. Eur J Pharmacol. 202;883:173371.
Guo X, García LR. SIR-2.1 integrates metabolic homeostasis with the reproductive neuromuscular excitability in early aging male Caenorhabditis elegans. eLife. 2014;3:e01730.
Statzer C, Meng J, Venz R, Bland M, Robida-Stubbs R, Patel K, et al. ATF-4 and hydrogen sulfide signalling mediate longevity in response to inhibition of translation or mTORC1. Nat Commun. 2022;13:967.
Farias-Pereira R, Savarese J, Yue Y, Lee SH, Park Y. Fat-lowering effects of isorhamnetin are via NHR-49-dependent pathway in Caenorhabditis elegans. Curr Res Food Sci. 2019;2:70–76.
Acknowledgements
This research received funding from the Bulgarian National Science Fund under BG-175467353-2023-03 programme for project Natural NRF2 activators modulating obesity mechanisms: molecular pharmacology-based in vivo study (contract number КП-06-КОСТ/4), the European Union’s Horizon 2020 research and innovation programme, project PlantaSYST (SGA No 739582 under FPA No. 664620) and by the European Regional Development Fund through the Bulgarian “Science and Education for Smart Growth” Operational Programme (project BG05M2OP001-1.003-0001-C01) and Programme Research Innovation and Digitalisation for Smart Transformation. This article is based upon work from COST Action CA20121 BenBedPhar, supported by COST (European Cooperation in Science and Technology).
Author information
Authors and Affiliations
Contributions
Martina S. Savova: Conceptualization, Methodology, Formal analysis, Data curation, Writing – original draft, review and editing. Monika N. Todorova: Conceptualization, Methodology, Data curation, Writing – original draft, Visualization, Investigation. Biser K. Binev: Methodology. Milen I. Georgiev: Conceptualization, Methodology, Supervision, Funding acquisition, Writing – review and editing. Liliya V. Mihaylova: Conceptualization, Methodology, Formal analysis, Writing – review and editing, Funding acquisition, Supervision.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
This study does not involve human participants, human material, or live vertebrates. The research was conducted using C. elegans as a model organism, which aligns with the Replacement principle of the 3 R’s (Replacement, Reduction and Refinement) philosophy. Therefore, ethics approval or consent to participate were not required.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Savova, M.S., Todorova, M.N., Binev, B.K. et al. Curcumin enhances the anti-obesogenic activity of orlistat through SKN-1/NRF2-dependent regulation of nutrient metabolism in Caenorhabditis elegans. Int J Obes 49, 516–526 (2025). https://doi.org/10.1038/s41366-025-01724-6
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41366-025-01724-6