Abstract
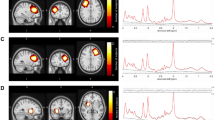
Research findings implicate cerebral glutamate in the pathophysiology of schizophrenia, including genetic studies reporting associations with glutamatergic neurotransmission. The extent to which aberrant glutamate levels can be explained by genetic factors is unknown, and if glutamate can serve as a marker of genetic susceptibility for schizophrenia remains to be established. We investigated the heritability of cerebral glutamate levels and whether a potential association with schizophrenia spectrum disorders could be explained by genetic factors. Twenty-three monozygotic (MZ) and 20 dizygotic (DZ) proband pairs con- or discordant for schizophrenia spectrum disorders, along with healthy control pairs (MZ = 28, DZ = 18) were recruited via the National Danish Twin Register and the Psychiatric Central Register (17 additional twins were scanned without their siblings). Glutamate levels in the left thalamus and the anterior cingulate cortex (ACC) were measured using 1[H]-magnetic resonance spectroscopy at 3 Tesla and analyzed by structural equation modeling. Glutamate levels in the left thalamus were heritable and positively correlated with liability for schizophrenia spectrum disorders (phenotypic correlation, 0.16, [0.02–0.29]; p = 0.010). The correlation was explained by common genes influencing both the levels of glutamate and liability for schizophrenia spectrum disorders. In the ACC, glutamate and glx levels were heritable, but not correlated to disease liability. Increases in thalamic glutamate levels found in schizophrenia spectrum disorders are explained by genetic influences related to the disease, and as such the measure could be a potential marker of genetic susceptibility, useful in early detection and stratification of patients with psychosis.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Marsman A, van den Heuvel MP, Klomp DWJ, Kahn RS, Luijten PR, Hulshoff Pol HE. Glutamate in schizophrenia: a focused review and meta-analysis of 1H-MRS studies. Schizophr Bull. 2013;39:120–9.
Merritt K, Egerton A, Kempton MJ, Taylor MJ, Mcguire PK. Nature of glutamate alterations in schizophrenia a meta-analysis of proton magnetic resonance spectroscopy studies. JAMA Psychiatry. 2016;73:1–10.
Ripke S, Neale BM, Corvin A, Walters JTR, Farh K-H, Holmans PA, et al. Biological insights from 108 schizophrenia-associated genetic loci. Nature. 2014. https://doi.org/10.1038/nature13595.
Schobel SA, Chaudhury NH, Khan UA, Paniagua B, Styner MA, Asllani I, et al. Imaging patients with psychosis and a mouse model establishes a spreading pattern of hippocampal dysfunction and implicates glutamate as a driver. Neuron. 2013;78:81–93.
Bustillo JR, Patel V, Jones T, Jung R, Payaknait N, Qualls C, et al. Risk-conferring glutamatergic genes and brain glutamate plus glutamine in schizophrenia. Front Psychiatry. 2017;8:1–9.
Javitt DC, Balla A, Burch S, Suckow R, Xie S, Sershen H. Reversal of phencyclidine-induced dopaminergic dysregulation by N-methyl-D-aspartate receptor/glycine-site agonists. Neuropsychopharmacology. 2004;29:300–7.
Egerton A, Brugger S, Raffin M, Barker GJ, Lythgoe DJ, McGuire PK, et al. Anterior cingulate glutamate levels related to clinical status following treatment in first-episode schizophrenia. Neuropsychopharmacology. 2012;37:2515–21.
Poels EMP, Kegeles LS, Kantrowitz JT, Javitt DC, Lieberman JA, Abi-Dargham A, et al. Glutamatergic abnormalities in schizophrenia: a review of proton MRS findings. Schizophr Res. 2014;152:325–32.
Egerton A, Bhachu A, Merritt K, McQueen G, Szulc A, McGuire P. Effects of antipsychotic administration on brain glutamate in schizophrenia: a systematic review of longitudinal 1H-MRS studies. Front Psychiatry. 2017;8:1–7.
Bojesen K, Jessen K, Sigvard A, Jensen MB, Rostrup E, Broberg BV, et al. 87. Glutamate and GABA in antipsychotic-naive schizophrenia and association with treatment outcome. Schizophr Bull. 2017;43:S48–S48.
Théberge J, Bartha R, Drost DJ, Menon RS, Malla A, Takhar J, et al. Glutamate and glutamine measured with 4.0 T proton MRS in never-treated patients with schizophrenia and healthy volunteers. Am J Psychiatry. 2002;159:1944–6.
Théberge J, Williamson KE, Aoyama N, Drost DJ, Manchanda R, Malla AK, et al. Longitudinal grey-matter and glutamatergic losses in first-episode schizophrenia. Br J Psychiatry. 2007;191:325–34.
Théberge J, Al-Semaan Y, Williamson PC, Menon RS, Neufeld RWJ, Rajakumar N, et al. Glutamate and glutamine in the anterior cingulate and thalamus of medicated patients with chronic schizophrenia and healthy comparison subjects measured with 4.0-T proton MRS. Am J Psychiatry. 2003;160:2231–3.
Galińska B, Szulc A, Tarasów E, Kubas B, Dzienis W, Czernikiewicz A, et al. Duration of untreated psychosis and proton magnetic resonance spectroscopy (1H-MRS) findings in first-episode schizophrenia. Med Sci Monit. 2009;15:CR82–88.
Szulc A, Galinska B, Tarasow E, Dzienis W, Kubas B, Konarzewska B, et al. The effect of risperidone on metabolite measures in the frontal lobe, temporal lobe, and thalamus in schizophrenic patients. A proton magnetic resonance spectroscopy (1 H MRS) study. Pharmacopsychiatry. 2005;38:214–9.
Szulc A, Galinska B, Tarasow E, Waszkiewicz N, Konarzewska B, Poplawska R, et al. Proton magnetic resonance spectroscopy study of brain metabolite changes after antipsychotic treatment. Pharmacopsychiatry. 2011;44:148–57.
Bustillo JR, Rowland LM, Mullins P, Jung R, Chen H, Qualls C, et al. 1H-MRS at 4 Tesla in minimally treated early schizophrenia. Mol Psychiatry. 2010;15:629–36.
Bartha R, Williamson PC, Drost DJ, Malla A, Carr TJ, Cortese L, et al. Measurement of glutamate and glutamine in the medial prefrontal cortex of never-treated schizophrenic patients and healthy controls by proton magnetic resonance spectroscopy. Arch Gen Psychiatry. 1997;54:959–65.
Egerton A, Broberg BV., Haren N van, Merritt K, Barker GJ, Lythgoe DJ, et al. Response to initial antipsychotic treatment in first episode psychosis is related to anterior cingulate glutamate levels: a multicentre1H-MRS study (OPTiMiSE). Mol Psychiatry 2018;1–11. https://doi.org/10.1038/s41380-018-0082-9
Tibbo PG, Bernier D, Hanstock CC, Seres P, Lakusta B, Purdon SE. 3-T proton magnetic spectroscopy in unmedicated first episode psychosis: a focus on creatine. Magn Reson Med. 2013;69:613–20.
Kegeles LS, Mao X, Stanford AD, Girgis R, Ojeil N, Xu X, et al. Elevated prefrontal cortex γ-aminobutyric acid and glutamate-glutamine levels in schizophrenia measured in vivo with proton magnetic resonance spectroscopy. Arch Gen Psychiatry. 2012;69:449–59.
la Fuente-Sandoval C de, Reyes-Madrigal F, Mao X, León-Ortiz P, Rodríguez-Mayoral O, Jung-Cook H, et al. Prefrontal and sStriatal gamma-aminobutyric acid levels and the effect of antipsychotic treatment in first-episode psychosis patients. Biol Psychiatry. 2018;83:475–483.
Yang Z, Zhu Y, Song Z, Mei L, Zhang J, Chen T, et al. Comparison of the density of gamma-aminobutyric acid in the ventromedial prefrontal cortex of patients with first-episode psychosis and healthy controls. Shanghai Arch Psychiatry. 2015;27:341–8.
Wang J, Wang J, Tang Y, Zhang T, Cui H, Xu L, et al. Reduced γ-aminobutyric acid and glutamate+glutamine levels in drug-naïve patients with first-episode schizophrenia but not in those at ultrahigh risk. Neural Plast. 2016;2016. https://doi.org/10.1155/2016/3915703.
Natsubori T, Inoue H, Abe O, Takano Y, Iwashiro N, Aoki Y, et al. Reduced frontal glutamate+glutamine and N-acetylaspartate levels in patients with chronic schizophrenia but not in those at clinical high risk for psychosis or with first-episode schizophrenia. Schizophr Bull. 2014;40:1128–39.
Tayoshi S, Sumitani S, Taniguchi K, Shibuya-Tayoshi S, Numata S, ichi Iga J, et al. Metabolite changes and gender differences in schizophrenia using 3-Tesla proton magnetic resonance spectroscopy (1H-MRS). Schizophr Res. 2009;108:69–77.
Marsman A, Mandl RCW, Klomp DWJ, Bohlken MM, Boer VO, Andreychenko A, et al. GABA and glutamate in schizophrenia: a 7 T 1H-MRS study. NeuroImage Clin. 2014;6:398–407.
Öngür D, Prescot AP, McCarthy J, Cohen BM, Renshaw PF. Elevated gamma-aminobutyric acid levels in chronic schizophrenia. Biol Psychiatry. 2010;68:667–70.
Lutkenhoff ES, van Erp TG, Thomas MA, Therman S, Manninen M, Huttunen MO, et al. Proton MRS in twin pairs discordant for schizophrenia. Mol Psychiatry. 2010;15:308–18.
Byun MS, Choi JS, Yoo SY, Kang DH, Choi CH, Jang DP, et al. Depressive symptoms and brain metabolite alterations in subjects at ultra-high risk for psychosis: a preliminary study. Psychiatry Investig. 2009;6:264–71.
Egerton A, Stone JM, Chaddock CA, Barker GJ, Bonoldi I, Howard RM, et al. Relationship between brain glutamate levels and clinical outcome in individuals at ultra high risk of psychosis. Neuropsychopharmacology. 2014;39:2891–9.
Stone JM, Day F, Tsagaraki H, Valli I, McLean MA, Lythgoe DJ, et al. Glutamate dysfunction in people with prodromal symptoms of ppsychosis: relationship to gray matter volume. Biol Psychiatry. 2009;66:533–9.
Tandon N, Bolo NR, Sanghavi K, Mathew IT, Francis AN, Stanley JA, et al. Brain metabolite alterations in young adults at familial high risk for schizophrenia using proton magnetic resonance spectroscopy. Schizophr Res. 2013;148:59–66.
Hegarty JP, Gu M, Spielman DM, Cleveland SC, Hallmayer JF, Lazzeroni LC, et al. A proton MR spectroscopy study of the thalamus in twins with autism spectrum disorder. Prog Neuro-Psychopharmacol Biol Psychiatry. 2017;81:153–60.
Hilker R, Helenius D, Fagerlund B, Skytthe A, Christensen K, Werge TM, et al. Heritability of schizophrenia and schizophrenia spectrum based on the nationwide Danish Twin Register. Biol Psychiatry. 2018;83:492–8.
Falconer D, Mackay T. Introduction to quantitative genetics. Essex, UK: Longman Group Ltd. 1996.
Boomsma D, Busjahn A, Peltonen L. Classical twin studies and beyond. Nat Rev Genet. 2002;3:872–82.
Batouli SAH, Sachdev PS, Wen W, Wright MJ, Suo C, Ames D, et al. The heritability of brain metabolites on proton magnetic resonance spectroscopy in older individuals. Neuroimage. 2012;62:281–9.
Kraguljac NV, Reid M, White D, Jones R, den Hollander J, Lowman D, et al. Neurometabolites in schizophrenia and bipolar disorder - a systematic review and meta-analysis. Psychiatry Res. 2012;203:111–25.
Schwerk A, Alves FDS, Pouwels PJW, Van Amelsvoort T. Metabolic alterations associated with schizophrenia: a critical evaluation of proton magnetic resonance spectroscopy studies. J Neurochem. 2014;128:1–87.
Steen RG, Hamer RM, Lieberman JA. Measurement of brain metabolites by 1H magnetic resonance spectroscopy in patients with schizophrenia: a systematic review and meta-analysis. Neuropsychopharmacology. 2005;30:1949–62.
Pedersen CB, Gøtzsche H, Møller JØ, Mortensen PB. The Danish civil registration system A cohort of eight million persons. Dan Med Bull. 2006;53:441–9.
Skytthe A, Ohm Kyvik K, Vilstrup Holm N, Christensen K. The Danish twin registry. Scand J Public Health. 2011;39:75–78.
Mors O, Perto GP, Mortensen PB. The Danish psychiatric central research register. Scand J Public Health. 2011;39:54–57.
Wing J, Babor T, T B, Burke J, Cooper J, Giel R, et al. SCAN schedules for clinical assessment in neuropsychiatry. Arch Gen Psychiatry. 1990;47:589–93.
Yung AR, Yuen HP, McGorry PD, Phillips LJ, Kelly D, Dell’Olio M, et al. Mapping the onset of psychosis: the comprehensive assessment of at-risk mental states. Aust N Z J Psychiatry. 2005;39:964–71.
Toulopoulou T, Picchioni M, Rijsdijk F, Hua-Hall M, Ettinger U, Sham P, et al. Substantial genetic overlap between neurocognition and schizophrenia. Arch Gen Psychiatry. 2007;64:1348.
Dominicus A, Skrondal A, Gjessing HK, Pedersen NL, Palmgren J. Likelihood ratio tests in behavioral genetics: problems and solutions. Behav Genet. 2006;36:331–40.
Boker S, Neale M, Maes H, Wilde M, Spiegel M, Brick T, et al. OpenMx: an open source extended structural equation modeling framework. Psychometrika. 2011;76:306–17.
van Haren NEM, Rijsdijk F, Schnack HG, Picchioni MM, Toulopoulou T, Weisbrod M, et al. The genetic and environmental determinants of the association between brain abnormalities and schizophrenia: the schizophrenia twins and relatives consortium. Biol Psychiatry. 2012;71:915–21.
Carlsson A. The neurochemical circuitry of schizophrenia. Pharmacopsychiatry. 2006;39:10–14.
Carlsson M, Carlsson A. Schizophrenia: a subcortical neurotransmitter imbalance syndrome? Schizophr Bull. 1990;16:425–32.
Bartha R. Measurement of glutamate and glutamine in the medial prefrontal cortex of never-treated schizophrenic patients and healthy controls by proton magnetic resonance spectroscopy. Arch Gen Psychiatry. 1997;54:959.
Durazzo TC, Meyerhoff DJ, Mon A, Abé C, Gazdzinski S, Murray DE. Chronic cigarette smoking in healthy middle-aged individuals is associated with decreased regional brain n-acetylaspartate and glutamate levels. Biol Psychiatry. 2016;79:481–8.
Gallinat J, Lang UE, Jacobsen LK, Bajbouj M, Kalus P, Von Haebler D, et al. Abnormal hippocampal neurochemistry in smokers: evidence from proton magnetic resonance spectroscopy at 3 T. J Clin Psychopharmacol. 2007;27:80–84.
Goto N, Yoshimura, ReijiKakeda S, Nishimura J, Moriya J, Hayashi K, et al. Six-month treatment with atypical antipsychotic drugs decreased frontal-lobe levels of glutamate plus glutamine in early-stage first-episode schizophrenia. Neuropsychiatr Dis Treat. 2012;8:119–22.
De La Fuente-Sandoval C, León-Ortiz P, Azcárraga M, Stephano S, Favila R, Díaz-Galvis L, et al. Glutamate levels in the associative striatum before and after 4 weeks of antipsychotic treatment in first-episode psychosis: a longitudinal proton magnetic resonance spectroscopy study. JAMA Psychiatry. 2013;70:1057–66.
Van Veenendaal TM, Backes WH, Van Bussel FCG, Edden RAE. Glutamate quantification by PRESS or MEGA-PRESS: validation, repeatability, and concordance. Magn Reson Imaging. 2018;48:107–14.
Moffett JR, Ross B, Arun P, Madhavarao CN, Namboodiri AMA. N-acetylaspartate in the CNS: from neurodiagnostics to neurobiology. Prog Neurobiol. 2007;81:89–131.
Béard E, Braissant O. Synthesis and transport of creatine in the CNS: importance for cerebral functions. J Neurochem. 2010;115:297–313.
Taylor R, Schaefer B, Densmore M, Neufeld RWJ, Rajakumar N, Williamson PC, et al. Increased glutamate levels observed upon functional activation in the anterior cingulate cortex using the Stroop Task and functional spectroscopy. Neuroreport. 2015;26:107–12.
Acknowledgements
The authors would like to acknowledge the valuable contribution of Mikkel Erlang Sørensen and Helle Schæbel in the recruitment and data collection process, and Lisbeth Jensen for keeping everything running smoothly.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interestst.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Legind, C.S., Broberg, B.V., Mandl, R.C.W. et al. Heritability of cerebral glutamate levels and their association with schizophrenia spectrum disorders: a 1[H]-spectroscopy twin study. Neuropsychopharmacol 44, 581–589 (2019). https://doi.org/10.1038/s41386-018-0236-0
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41386-018-0236-0
This article is cited by
-
Variability and magnitude of brain glutamate levels in schizophrenia: a meta and mega-analysis
Molecular Psychiatry (2023)
-
Thalamocortical dysrhythmia in patients with schizophrenia spectrum disorder and individuals at clinical high risk for psychosis
Neuropsychopharmacology (2022)
-
Glutamatergic and GABAergic metabolite levels in schizophrenia-spectrum disorders: a meta-analysis of 1H-magnetic resonance spectroscopy studies
Molecular Psychiatry (2022)
-
Evaluating endophenotypes for bipolar disorder
International Journal of Bipolar Disorders (2021)
-
Paternal exposure to excessive methionine altered behavior and neurochemical activities in zebrafish offspring
Amino Acids (2021)