Abstract
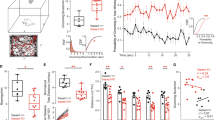
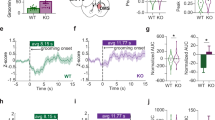
Obsessive-compulsive disorder (OCD) is a circuit disorder involving corticostriatal projections, which play a role in motor control. The Sapap3-knockout (KO) mouse is a mouse model to study OCD and recapitulates OCD-like compulsion through excessive grooming behavior, with skin lesions appearing at advanced age. Deficits in corticostriatal control provide a link to the pathophysiology of OCD. However, there remain significant gaps in the characterization of the Sapap3-KO mouse, with respect to age, specificity of synaptic dysfunction, and locomotor phenotype. We therefore investigated the corticostriatal synaptic phenotype of Sapap3-KO mice using patch–clamp slice electrophysiology, in adult mice and with projection specificity. We also analyzed grooming across age and locomotor phenotype with a novel, unsupervised machine learning technique (MoSeq). Increased grooming in Sapap3-KO mice without skin lesions was age independent. Synaptic deficits persisted in adulthood and involved the projections from the motor cortices and cingulate cortex to the dorsolateral and dorsomedial striatum. Decreased synaptic strength was evident at the input from the primary motor cortex by reduction in AMPA receptor function. Hypolocomotion, i.e., slowness of movement, was consistently observed in Sapap3-KO mice. Our findings emphasize the utility of young adult Sapap3-KO mice to investigate corticostriatal synaptic dysfunction in motor control.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Gillan CM, Robbins TW. Goal-directed learning and obsessive-compulsive disorder. Philos Trans R Soc Lond B Biol Sci. 2014;369:1655.
Graybiel AM, Rauch SL. Toward a neurobiology of obsessive-compulsive disorder. Neuron. 2000;28:343–7.
Park H, Popescu A, Poo MM. Essential role of presynaptic NMDA receptors in activity-dependent BDNF secretion and corticostriatal LTP. Neuron. 2014;84:1009–22.
Hunnicutt BJ, Jongbloets BC, Birdsong WT, Gertz KJ, Zhong H, Mao T. A comprehensive excitatory input map of the striatum reveals novel functional organization. Elife. 2016;5:e19103.
Graybiel AM, Aosaki T, Flaherty AW, Kimura M. The basal ganglia and adaptive motor control. Science. 1994;265:1826–31.
Rasmussen AH, Rasmussen HB, Silahtaroglu A. The DLGAP family: neuronal expression, function and role in brain disorders. Mol Brain. 2017;10:43.
Zuchner S, Wendland JR, Ashley-Koch AE, Collins AL, Tran-Viet KN, Quinn K, et al. Multiple rare SAPAP3 missense variants in trichotillomania and OCD. Mol Psychiatry. 2009;14:6–9.
Bienvenu OJ, Wang Y, Shugart YY, Welch JM, Grados MA, Fyer AJ, et al. Sapap3 and pathological grooming in humans: results from the OCD collaborative genetics study. Am J Med Genet B Neuropsychiatr Genet. 2009;150B:710–20.
Welch JM, Lu J, Rodriguiz RM, Trotta NC, Peca J, Ding JD, et al. Cortico-striatal synaptic defects and OCD-like behaviours in Sapap3-mutant mice. Nature.2007;448:894–900.
van den Boom BJG, Pavlidi P, Wolf CJH, Mooij AH, Willuhn I. Automated classification of self-grooming in mice using open-source software. J Neurosci Methods. 2017;289:48–56.
Burguiere E, Monteiro P, Feng G, Graybiel AM. Optogenetic stimulation of lateral orbitofronto-striatal pathway suppresses compulsive behaviors. Science 2013;340:1243–6.
Ade KK, Wan Y, Hamann HC, O’Hare JK, Guo W, Quian A, et al. Increased metabotropic glutamate receptor 5 signaling underlies obsessive-compulsive disorder-like behavioral and striatal circuit abnormalities in mice. Biol Psychiatry. 2016;80:522–33.
Manning EE, Dombrovski AY, Torregrossa MM, Ahmari SE. Impaired instrumental reversal learning is associated with increased medial prefrontal cortex activity in Sapap3 knockout mouse model of compulsive behavior. Neuropsychopharmacology. 2019;44:1494–504.
van den Boom BJG, Mooij AH, Miseviciute I, Denys D, Willuhn I. Behavioral flexibility in a mouse model for obsessive-compulsive disorder: Impaired Pavlovian reversal learning in SAPAP3 mutants. Genes Brain Behav. 2019;18:e12557.
Hadjas LC, Luscher C, Simmler LD. Aberrant habit formation in the Sapap3-knockout mouse model of obsessive-compulsive disorder. Sci Rep. 2019;9:12061.
Ehmer I, Feenstra M, Willuhn I, Denys D. Instrumental learning in a mouse model for obsessive-compulsive disorder: impaired habit formation in Sapap3 mutants. Neurobiol Learn Mem. 2020;168:107162.
Pinhal CM, van den Boom BJG, Santana-Kragelund F, Fellinger L, Bech P, Hamelink R, et al. Differential effects of deep brain stimulation of the internal capsule and the striatum on excessive grooming in Sapap3 mutant mice. Biol Psychiatry. 2018;84:917–25.
Todorov G, Mayilvahanan K, Ashurov D, Cunha C. Amelioration of obsessive-compulsive disorder in three mouse models treated with one epigenetic drug: unraveling the underlying mechanism. Sci Rep. 2019;9:8741.
Chen M, Wan Y, Ade K, Ting J, Feng G, Calakos N. Sapap3 deletion anomalously activates short-term endocannabinoid-mediated synaptic plasticity. J Neurosci. 2011;31:9563–73.
Wan Y, Feng G, Calakos N. Sapap3 deletion causes mGluR5-dependent silencing of AMPAR synapses. J Neurosci. 2011;31:16685–91.
Wan Y, Ade KK, Caffall Z, Ilcim Ozlu M, Eroglu C, Feng G, et al. Circuit-selective striatal synaptic dysfunction in the Sapap3 knockout mouse model of obsessive-compulsive disorder. Biol Psychiatry. 2014;75:623–30.
Corbit VL, Manning EE, Gittis AH, Ahmari SE. Strengthened inputs from secondary motor cortex to striatum in a mouse model of compulsive behavior. J Neurosci. 2019;39:2965–75.
Luscher C, Malenka RC. NMDA receptor-dependent long-term potentiation and long-term depression (LTP/LTD). Cold Spring Harb Perspect Biol. 2012;4:a005710.
Kindler S, Rehbein M, Classen B, Richter D, Bockers TM. Distinct spatiotemporal expression of SAPAP transcripts in the developing rat brain: a novel dendritically localized mRNA. Brain Res Mol Brain Res. 2004;126:14–21.
Wiltschko AB, Johnson MJ, Iurilli G, Peterson RE, Katon JM, Pashkovski SL, et al. Mapping sub-second structure in mouse behavior. Neuron. 2015;88:1121–35.
Mathis A, Mamidanna P, Cury KM, Abe T, Murthy VN, Mathis MW, et al. DeepLabCut: markerless pose estimation of user-defined body parts with deep learning. Nat Neurosci. 2018;21:1281–89.
Abbas W, Masip Rodo D. Computer methods for automatic locomotion and gesture tracking in mice and small animals for neuroscience applications: a survey. Sensors. 2019;19:3274.
Pascoli V, Hiver A, Van Zessen R, Loureiro M, Achargui R, Harada M, et al. Stochastic synaptic plasticity underlying compulsion in a model of addiction. Nature.2018;564:366–71.
Paxinos G, Franklin KBJ. The mouse brain in stereotaxic coordinates. Compact 2nd ed. Amsterdam; Boston: Elsevier Academic Press; 2004.
Yuan T, Bellone C. Glutamatergic receptors at developing synapses: the role of GluN3A-containing NMDA receptors and GluA2-lacking AMPA receptors. Eur J Pharmaol. 2013;719:107–11.
Ruscio AM, Stein DJ, Chiu WT, Kessler RC. The epidemiology of obsessive-compulsive disorder in the National Comorbidity Survey Replication. Mol Psychiatry. 2010;15:53–63.
Kalueff AV, Stewart AM, Song C, Berridge KC, Graybiel AM, Fentress JC. Neurobiology of rodent self-grooming and its value for translational neuroscience. Nat Rev Neurosci. 2016;17:45–59.
Ehmer I, Crown L, van Leeuwen W, Feenstra M, Willuhn I, Denys D. Evidence for distinct forms of compulsivity in the SAPAP3 mutant-mouse model for obsessive-compulsive disorder. eNeuro. 2020;7:ENEURO.0245-19.2020.
Albin RL, Young AB, Penney JB. The functional anatomy of basal ganglia disorders. Trends Neurosci. 1989;12:366–75.
Wood J, LaPalombara Z, Ahmari SE. Monoamine abnormalities in the SAPAP3 knockout model of obsessive-compulsive disorder-related behaviour. Philos Trans R Soc Lond B Biol Sci. 2018;373:20170023.
Mahgoub M, Adachi M, Suzuki K, Liu X, Kavalali ET, Chahrour MH, et al. MeCP2 and histone deacetylases 1 and 2 in dorsal striatum collectively suppress repetitive behaviors. Nat Neurosci. 2016;19:1506–12.
Hirjak D, Meyer-Lindenberg A, Fritze S, Sambataro F, Kubera KM, Wolf RC. Motor dysfunction as research domain across bipolar, obsessive-compulsive and neurodevelopmental disorders. Neurosci Biobehav Rev. 2018;95:315–35.
Bihari K, Pato MT, Hill JL, Murphy DL. Neurologic soft signs in obsessive-compulsive disorder. Arch Gen Psychiatry. 1991;48:278–9.
Hollander E, Schiffman E, Cohen B, Rivera-Stein MA, Rosen W, Gorman JM, et al. Signs of central nervous system dysfunction in obsessive-compulsive disorder. Arch Gen Psychiatry. 1990;47:27–32.
Mergl R, Mavrogiorgou P, Juckel G, Zaudig M, Hegerl U. Effects of sertraline on kinematic aspects of hand movements in patients with obsessive-compulsive disorder. Psychopharmacol. 2004;171:179–85.
Kuelz AK, Hohagen F, Voderholzer U. Neuropsychological performance in obsessive-compulsive disorder: a critical review. Biol Psychol. 2004;65:185–236.
Snyder HR, Kaiser RH, Warren SL, Heller W. Obsessive-compulsive disorder is associated with broad impairments in executive function: a meta-analysis. Clin Psychol Sci. 2015;3:301–30.
Tukel R, Gurvit H, Ertekin BA, Oflaz S, Ertekin E, Baran B, et al. Neuropsychological function in obsessive-compulsive disorder. Compr Psychiatry. 2012;53:167–75.
Hymas N, Lees A, Bolton D, Epps K, Head D. The neurology of obsessional slowness. Brain. 1991;114:2203–33.
Benzina N, Mallet L, Burguiere E, N’Diaye K, Pelissolo A. Cognitive dysfunction in obsessive-compulsive disorder. Curr Psychiatry Rep. 2016;18:80.
Gremel CM, Costa RM. Premotor cortex is critical for goal-directed actions. Front Comput Neurosci. 2013;7:110.
Gremel CM, Chancey JH, Atwood BK, Luo G, Neve R, Ramakrishnan C, et al. Endocannabinoid modulation of orbitostriatal circuits gates habit formation. Neuron. 2016;90:1312–24.
Simmler LD, Ozawa T. Neural circuits in goal-directed and habitual behavior: Implications for circuit dysfunction in obsessive-compulsive disorder. Neurochem Int. 2019;129:104464.
Ahmari SE, Spellman T, Douglass NL, Kheirbek MA, Simpson HB, Deisseroth K, et al. Repeated cortico-striatal stimulation generates persistent OCD-like behavior. Science.2013;340:1234–9.
Acknowledgements
The authors would like to thank Agnès Hiver and Sébastien Pellat for excellent laboratory assistance and technical support.
Author information
Authors and Affiliations
Contributions
LDS, LCH, JC, MCC, and VP performed experiments. MMS implemented and performed MoSeq analysis. LDS and CL conceptualized and supervised the study. LDS wrote the paper. All authors contributed to and approved the final paper.
Corresponding author
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hadjas, L.C., Schartner, M.M., Cand, J. et al. Projection-specific deficits in synaptic transmission in adult Sapap3-knockout mice. Neuropsychopharmacol. 45, 2020–2029 (2020). https://doi.org/10.1038/s41386-020-0747-3
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/s41386-020-0747-3
This article is cited by
-
Late development of OCD-like phenotypes in Dlgap1 knockout mice
Psychopharmacology (2025)
-
Closed-loop recruitment of striatal interneurons prevents compulsive-like grooming behaviors
Nature Neuroscience (2024)
-
Conditional deletion of neurexin-2 impaired behavioral flexibility to alterations in action–outcome contingency
Scientific Reports (2024)
-
Hyperactivity of indirect pathway-projecting spiny projection neurons promotes compulsive behavior
Nature Communications (2024)
-
Characterizing the structure of mouse behavior using Motion Sequencing
Nature Protocols (2024)