Abstract
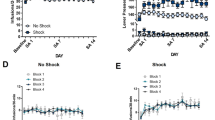
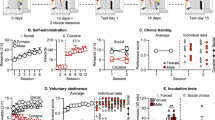
One behavioral feature of drug addiction is continued drug use despite awareness that this causes negative consequences. Attempts to model this feature in animals typically involve punishing drug self-administration with electrical footshock to identify individuals whose drug use is differently suppressed by punishment. Here we sought to further study individual responsiveness of drug use to punishment in rats self-administering intravenous cocaine. Rats were first trained during several weeks to self-administer cocaine under a fixed-ratio 3 schedule of reinforcement. Then, their self-administration behavior was punished with increasing intensity of footshock (i.e., from 0.1 mA to 0.9 mA, every 30 min). With increasing intensity of punishment, rats first continued to self-administer cocaine before eventually stopping near completely. When retested, however, drug use became more responsive to punishment and was suppressed by a low and initially ineffective footshock intensity (i.e., 0.1 mA). This increase in responsiveness to punishment was seen in all individuals tested, albeit with varying degrees, and was acquired after one single experience with an intensity of punishment that near completely suppressed drug self-administration. Mere passive, non-contingent exposure to the same intensity, however, had no such effect. Once acquired, increased responsiveness to punishment persisted during at least one month when rats were tested every week, but not every day. Finally, increased responsiveness to punishment was not observed after exposure to a non-painful form of punishment (i.e., histamine). Overall, this study reveals that initial responsiveness of drug use to punishment can change rapidly and persistently with experience. We discuss several possible mechanisms that may account for this change in punishment responsiveness and also draw some of the implications and future perspectives for research on animal models of compulsion-like behavior.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Association AP. Diagnostic and statistical manual of mental disorders (DSM-5). American Psychiatric Association: Washington, DC; 2013.
Martin CS, Langenbucher JW, Chung T, Sher KJ. Truth or consequences in the diagnosis of substance use disorders. Addiction. 2014;109:1773–8.
Hasin DS, O’Brien CP, Auriacombe M, Borges G, Bucholz K, Budney A, et al. DSM-5 criteria for substance use disorders: recommendations and rationale. Am J Psychiatry. 2013;170:834–51.
Pickard H, Ahmed SH. How do you know you have a drug problem? The role of knowledge of negative consequences in explaining drug choice in humans and rats. In: Heather N, Segal G, editors. Addiction and choice. Oxford: Oxford University Press; 2017. p. 29–48.
Klingemann H, Sobell MB, Sobell LC. Continuities and changes in self-change research. Addiction. 2010;105:1510–8.
Heather N. Is the concept of compulsion useful in the explanation or description of addictive behaviour and experience? Addictive Behav Rep. 2017;6:15–38.
Hogarth L. Addiction is driven by excessive goal-directed drug choice under negative affect: translational critique of habit and compulsion theory. Neuropsychopharmacology. 2020;45:720–35.
Heyman GM. Quitting drugs: quantitative and qualitative features. Annu Rev Clin Psychol. 2013;9:29–59.
Smith GS, Davis M. Punishment of amphetamine and morphine self-administration behavior. Psychological Rec. 1974;24:477–80.
Grove RN, Schuster CR. Suppression of cocaine self-administration by extinction and punishment. Pharm Biochem Behav. 1974;2:199–208.
Bergman J, Johanson CE. The effects of electric shock on responding maintained by cocaine in rhesus monkeys. Pharm Biochem Behav. 1981;14:423–6.
Wolffgramm J, Heyne A. From controlled drug intake to loss of control: the irreversible development of drug addiction in the rat. Behav Brain Res. 1995;70:77–94.
Vanderschuren LJMJ, Minnaard AM, Smeets JAS, Lesscher HM. Punishment models of addictive behavior. Curr Opin Behav Sci. 2017;13:77–84.
Vanderschuren L, Ahmed SH. Animal models of the behavioral symptoms of substance use disorders. New York: Cold Spring Harbor Perspectives in Medicine; 2020.
Jean-Richard-Dit-Bressel P, Killcross S, McNally GP. Behavioral and neurobiological mechanisms of punishment: implications for psychiatric disorders. Neuropsychopharmacology. 2018;43:1639–50.
Ahmed SH. “A walk on the wild side” of addiction: the history and significance of animal models. In: Pickard H, Ahmed SH, editors. The Routledge Handbook of Philosophy and Science of Addiction. New York: Routledge; 2019. p. 192–204.
Ahmed SH. In search of addiction in the brains of laboratory animals. Evaluating the Brain Disease Model of Addiction. Routledge;2021, in press.
Deroche-Gamonet V, Belin D, Piazza PV. Evidence for addiction-like behavior in the rat. Science. 2004;305:1014–7.
Panlilio LV, Thorndike EB, Schindler CW. Reinstatement of punishment-suppressed opioid self-administration in rats: an alternative model of relapse to drug abuse. Psychopharmacology. 2003;168:229–35.
Chen BT, Yau HJ, Hatch C, Kusumoto-Yoshida I, Cho SL, Hopf FW, et al. Rescuing cocaine-induced prefrontal cortex hypoactivity prevents compulsive cocaine seeking. Nature. 2013;496:359–62.
Pascoli V, Terrier J, Hiver A, Luscher C. Sufficiency of mesolimbic dopamine neuron stimulation for the progression to addiction. Neuron. 2015;88:1054–66.
Ahmed SH. Validation crisis in animal models of drug addiction: beyond non-disordered drug use toward drug addiction. Neurosci Biobehav Rev. 2010;35:172–84.
Ahmed SH. The science of making drug-addicted animals. Neuroscience. 2012;211:107–25.
Kasanetz F, Deroche-Gamonet V, Berson N, Balado E, Lafourcade M, Manzoni O, et al. Transition to addiction is associated with a persistent impairment in synaptic plasticity. Science. 2010;328:1709–12.
Kasanetz F, Lafourcade M, Deroche-Gamonet V, Revest JM, Berson N, Balado E, et al. Prefrontal synaptic markers of cocaine addiction-like behavior in rats. Mol Psychiatry. 2013;18:729–37.
Farrell MR, Ruiz CM, Castillo E, Faget L, Khanbijian C, Liu S, et al. Ventral pallidum is essential for cocaine relapse after voluntary abstinence in rats. Neuropsychopharmacology. 2019;44:2174–85.
Giuliano C, Belin D, Everitt BJ. Compulsive alcohol seeking results from a failure to disengage dorsolateral striatal control over behavior. J Neurosci. 2019;39:1744–54.
Giuliano C, Pena-Oliver Y, Goodlett CR, Cardinal RN, Robbins TW, Bullmore ET, et al. Evidence for a long-lasting compulsive alcohol seeking phenotype in rats. Neuropsychopharmacology. 2018;43:728–38.
Krasnova IN, Marchant NJ, Ladenheim B, McCoy MT, Panlilio LV, Bossert JM, et al. Incubation of methamphetamine and palatable food craving after punishment-induced abstinence. Neuropsychopharmacology. 2014;39:2008–16.
Torres OV, Jayanthi S, Ladenheim B, McCoy MT, Krasnova IN, Cadet JL. Compulsive methamphetamine taking under punishment is associated with greater cue-induced drug seeking in rats. Behav Brain Res. 2017;326:265–71.
Marchant NJ, Campbell EJ, Whitaker LR, Harvey BK, Kaganovsky K, Adhikary S, et al. Role of ventral subiculum in context-induced relapse to alcohol seeking after punishment-imposed abstinence. J Neurosci. 2016;36:3281–94.
Marchant NJ, Kaganovsky K, Shaham Y, Bossert JM. Role of corticostriatal circuits in context-induced reinstatement of drug seeking. Brain Res. 2015;1628:219–32.
Marchant NJ, Khuc TN, Pickens CL, Bonci A, Shaham Y. Context-induced relapse to alcohol seeking after punishment in a rat model. Biol Psychiatry. 2013;73:256–62.
Marchant NJ, Rabei R, Kaganovsky K, Caprioli D, Bossert JM, Bonci A, et al. A critical role of lateral hypothalamus in context-induced relapse to alcohol seeking after punishment-imposed abstinence. J Neurosci. 2014;34:7447–57.
Pelloux Y, Hoots JK, Cifani C, Adhikary S, Martin J, Minier-Toribio A, et al. Context-induced relapse to cocaine seeking after punishment-imposed abstinence is associated with activation of cortical and subcortical brain regions. Addict Biol. 2018;23:699–712.
Pelloux Y, Minier-Toribio A, Hoots JK, Bossert JM, Shaham Y. Opposite effects of basolateral amygdala inactivation on context-induced relapse to cocaine seeking after extinction versus punishment. J Neurosci. 2018;38:51–9.
Datta U, Martini M, Fan M, Sun W. Compulsive sucrose- and cocaine-seeking behaviors in male and female Wistar rats. Psychopharmacology. 2018;235:2395–405.
Datta U, Martini M, Sun W. Different functional domains measured by cocaine self-administration under the progressive-ratio and punishment schedules in male Wistar rats. Psychopharmacology. 2018;235:897–907.
Ahmed SH, Koob GF. Transition to drug addiction: a negative reinforcement model based on an allostatic decrease in reward function. Psychopharmacology. 2005;180:473–90.
Pickens R, Meisch RA, Thompson T. Drug self-administration: an analysis of the reinforcing effects of drugs. In: Iversen LL, Iversen, SD, Solomon, SH, editors. Handbook of Psychopharmacology: Drugs of Abuse. New York: Plenum Press; 1978. p. 1–37.
Yokel RA. Intravenous self-administration: response rates, the effects of pharmacological challenges, and drug preference. In: Bozarth MA, editor. Methods of Assessing the Reinforcing Properties of Abused Drugs. New York: Springer-Verlag; 1987. p. 1–33.
Fontes RM, Shahan TA. Punishment and its putative fallout: a reappraisal. J Exp Anal Behav. 2021;115:185–203.
Holtz NA, Carroll ME. Cocaine self-administration punished by intravenous histamine in adolescent and adult rats. Behav Pharm. 2015;26:393–7.
Negus SS. Effects of punishment on choice between cocaine and food in rhesus monkeys. Psychopharmacology. 2005;181:244–52.
Gancarz-Kausch AM, Adank DN, Dietz DM. Prolonged withdrawal following cocaine self-administration increases resistance to punishment in a cocaine binge. Sci Rep. 2014;4:6876.
Goldberg SR. Histamine as a punisher in squirrel monkeys: effects of pentobarbital, chlordiazepoxide and H1- and H2-receptor antagonists on behavior and cardiovascular responses. J Pharm Exp Ther. 1980;214:726–36.
Holtz NA, Anker JJ, Regier PS, Claxton A, Carroll ME. Cocaine self-administration punished by i.v. histamine in rat models of high and low drug abuse vulnerability: effects of saccharin preference, impulsivity, and sex. Physiol Behav. 2013;122:32–8.
Podlesnik CA, Jimenez-Gomez C, Woods JH. A choice procedure to assess the aversive effects of drugs in rodents. J Exp Anal Behav. 2010;93:203–23.
Woolverton WL. A novel choice method for studying drugs as punishers. Pharm Biochem Behav. 2003;76:125–31.
Woolverton WL, Freeman KB, Myerson J, Green L. Suppression of cocaine self-administration in monkeys: effects of delayed punishment. Psychopharmacology. 2012;220:509–17.
Bolles RC, Holtz R, Dunn T, Hill W. Comparisons of stimulus learning and response learning in a punishment situation. Learn Motiv. 1980;11:78–96.
Dickinson A, Pearce JM. Inhibitory interactions between appetitive and aversive stimuli. Psychological Bull. 1977;84:690–711.
Miller NE. Learning resistance to pain and fear: effects of overlearning, exposure, and rewarded expsure in context. J Exp Psychol. 1960;60:137–45.
Terris W, Barnes M. Learned resistance to punishment and subsequent responsiveness to the same and novel punishments. Psychon Sci. 1969;15:49–50.
Kober H, Kross EF, Mischel W, Hart CL, Ochsner KN. Regulation of craving by cognitive strategies in cigarette smokers. Drug Alcohol Depend. 2010;106:52–5.
Suzuki S, Mell MM, O’Malley SS, Krystal JH, Anticevic A, Kober H. Regulation of craving and negative emotion in alcohol use disorder. Biol Psychiatry Cogn Neurosci Neuroimaging. 2020;5:239–50.
Volkow ND, Fowler JS, Wang GJ, Telang F, Logan J, Jayne M, et al. Cognitive control of drug craving inhibits brain reward regions in cocaine abusers. Neuroimage. 2010;49:2536–43.
Parvaz MA, Malaker P, Zilverstand A, Moeller SJ, Alia-Klein N, Goldstein RZ. Attention bias modification in drug addiction: enhancing control of subsequent habits. Proc Natl Acad Sci USA. 2021;118:e2012941118.
Acknowledgements
We thank Christophe Bernard, Mathieu Louvet and Eric Wattelet for administrative assistance. We also thank Drs. Karine Guillem and Magalie Lenoir for their helpful comments on a previous version of the manuscript. We also thank Pr. Gavan McNally for bringing our attention to the old literature on punishment. We also thank the reviewers for their insightful and constructive criticisms.
Funding
This work was supported by the French Research Council (CNRS), the Université de Bordeaux, the Conseil Régional d’Aquitaine (CRA20101301022; CRA11004375/11004699) and the French National Agency (ANR2010-BLAN-1404-01, ANR-10-EQX-008-1, LabEx BRAIN). LF was supported by the CAPES–Brazilian Federal Agency for Support and Evaluation of Graduate Education within the Ministry of Education of Brazil.
Author information
Authors and Affiliations
Contributions
SHA conceived the project. AD, SHA designed the experiments. AD carried out the experiments with the participation of PG and LF. AD collected the experimental data. AD, SHA analyzed the data. AD, SHA wrote the paper. All authors reviewed content and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Durand, A., Girardeau, P., Freese, L. et al. Increased responsiveness to punishment of cocaine self-administration after experience with high punishment. Neuropsychopharmacol. 47, 444–453 (2022). https://doi.org/10.1038/s41386-021-01159-3
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41386-021-01159-3
This article is cited by
-
Sex and dose-dependent effects of cannabidiol on cocaine consumption in mice
Translational Psychiatry (2026)
-
Role of serotonin neurons in the dorsal raphe nucleus in heroin self-administration and punishment
Neuropsychopharmacology (2025)
-
Remission from addiction: erasing the wrong circuits or making new ones?
Nature Reviews Neuroscience (2025)
-
Delay of punishment highlights differential vulnerability to developing addiction-like behavior toward sweet food
Translational Psychiatry (2024)
-
Effects of pharmacological and environmental manipulations on choice between fentanyl and shock avoidance/escape in male and female rats under mutually exclusive and non-exclusive choice conditions
Neuropsychopharmacology (2024)