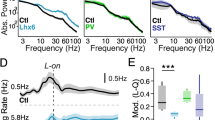
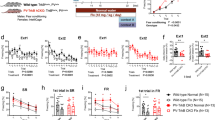
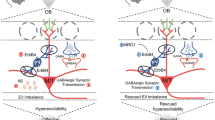
Abstract
Consolidated memories influence later learning and cognitive processes when new information is overlapped with previous events. To reveal which cellular and molecular factors are associated with this proactive interference, we challenged mice with odor–reward associative learning followed by a reversal-learning task. The results showed that genetical ablation of ErbB4 in parvalbumin (PV)-positive interneurons improved performance in reversal-learning phase, with no alteration in learning phase, supporting that PV interneuron ErbB4 is required for proactive interference. Mechanistically, olfactory learning promoted PV interneuron excitatory synaptic plasticity and direct binding of ErbB4 with presynaptic Neurexin1β (NRXN1β) and postsynaptic scaffold PSD-95 in the prefrontal cortex. Interrupting ErbB4–NRXN1β interaction impaired network activity-driven excitatory inputs and excitatory synaptic transmission onto PV interneurons. Neuronal activity-induced ErbB4-PSD-95 association facilitated transsynaptic binding of ErbB4–NRXN1β and excitatory synapse formation in ErbB4-positive interneurons. Furthermore, ErbB4–NRXN1β binding was responsible for the activity-regulated activation of ErbB4 and extracellular signal-regulated kinase (ERK) 1/2 in PV interneurons, as well as synaptic plasticity-related expression of brain-derived neurotrophic factor (BDNF). Correlatedly, blocking ErbB4–NRXN1β coupling in the medial prefrontal cortex of adult mice facilitated reversal learning of an olfactory associative task. These findings provide novel insight into the physiological role of PV interneuron ErbB4 signaling in cognitive processes and reveal an associative learning-related transsynaptic NRXN1β-ErbB4-PSD-95 complex that affects the ERK1/2-BDNF pathway and underlies local inhibitory circuit plasticity and proactive interference.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Crossley M, Lorenzetti FD, Naskar S, O’Shea M, Kemenes G, Benjamin PR, et al. Proactive and retroactive interference with associative memory consolidation in the snail Lymnaea is time and circuit dependent. Commun Biol. 2019;2:242.
Devkar DT, Wright AA. Event-based proactive interference in rhesus monkeys. Psychon Bull Rev. 2016;23:1474–82.
Wright AA, Katz JS, Ma WJ. How to be proactive about interference: lessons from animal memory. Psychol Sci. 2012;23:453–58.
Whissell PD, Cajanding JD, Fogel N, Kim JC. Comparative density of CCK- and PV-GABA cells within the cortex and hippocampus. Front Neuroanat. 2015;9:124.
Rudy B, Fishell G, Lee S, Hjerling-Leffler J. Three groups of interneurons account for nearly 100% of neocortical GABAergic neurons. Dev Neurobiol. 2011;71:45–61.
Hansen MG, Ledri LN, Kirik D, Kokaia M, Ledri M. Preserved function of afferent parvalbumin-positive perisomatic inhibitory synapses of dentate granule cells in rapidly kindled mice. Front Cell Neurosci. 2018;11:433.
Volman V, Behrens MM, Sejnowski TJ. Downregulation of parvalbumin at cortical GABA synapses reduces network gamma oscillatory activity. J Neurosci. 2011;31:18137–48.
Groisman AI, Yang SM, Schinder AF. Differential coupling of adult-born granule cells to parvalbumin and somatostatin interneurons. Cell Rep. 2020;30:202–14.
Tripodi M, Bhandari K, Chowdhury A, Mukherjee A, Caroni P. Parvalbumin interneuron plasticity for consolidation of reinforced learning. Cold Spring Harb Symp Quant Biol. 2018;83:25–35.
Favuzzi E, Marques-Smith A, Deogracias R, Winterflood CM, Sánchez-Aguilera A, Mantoan L, et al. Activity-dependent gating of parvalbumin interneuron function by the perineuronal net protein Brevican. Neuron 2017;95:639–55.
Ognjanovski N, Schaeffer S, Wu J, Mofakham S, Maruyama D, Zochowski M, et al. Parvalbumin-expressing interneurons coordinate hippocampal network dynamics required for memory consolidation. Nat Commun. 2017;8:15039.
Xia F, Richards BA, Tran MM, Josselyn SA, Takehara-Nishiuchi K, Frankland PW. Parvalbumin-positive interneurons mediate neocortical-hippocampal interactions that are necessary for memory consolidation. Elife 2017;6:e27868.
Alvarez DD, Giacomini D, Yang SM, Trinchero MF, Temprana SG, Büttner KA, et al. A disynaptic feedback network activated by experience promotes the integration of new granule cells. Science 2016;354:459–65.
Donato F, Rompani SB, Caroni P. Parvalbumin-expressing basket-cell network plasticity induced by experience regulates adult learning. Nature 2013;504:272–6.
Yin DM, Sun XD, Bean JC, Lin TW, Sathyamurthy A, Xiong WC, et al. Regulation of spine formation by ErbB4 in PV-positive interneurons. J Neurosci. 2013;33:19295–303.
Ting AK, Chen Y, Wen L, Yin DM, Shen C, Tao Y, et al. Neuregulin 1 promotes excitatory synapse development and function in GABAergic interneurons. J Neurosci. 2011;31:15–25.
Fazzari P, Paternain AV, Valiente M, Pla R, Luján R, Lloyd K, et al. Control of cortical GABA circuitry development by Nrg1 and ErbB4 signalling. Nature 2010;464:1376–80.
Vullhorst D, Neddens J, Karavanova I, Tricoire L, Petralia RS, McBain CJ, et al. Selective expression of ErbB4 in interneurons, but not pyramidal cells, of the rodent hippocampus. J Neurosci. 2009;29:12255–64.
Yau HJ, Wang HF, Lai C, Liu FC. Neural development of the neuregulin receptor ErbB4 in the cerebral cortex and the hippocampus: preferential expression by interneurons tangentially migrating from the ganglionic eminences. Cereb Cortex. 2003;13:252–64.
Huang YZ, Won S, Ali DW, Wang Q, Tanowitz M, Du QS, et al. Regulation of neuregulin signaling by PSD-95 interacting with ErbB4 at CNS synapses. Neuron 2000;26:443–55.
Deng W, Luo F, Li BM, Mei L. NRG1-ErbB4 signaling promotes functional recovery in a murine model of traumatic brain injury via regulation of GABA release. Exp Brain Res. 2019;237:3351–62.
Mei L, Nave KA. Neuregulin-ERBB signaling in the nervous system and neuropsychiatric diseases. Neuron 2014;83:27–49.
Mei L, Xiong WC. Neuregulin 1 in neural development, synaptic plasticity and schizophrenia. Nat Rev Neurosci. 2008;9:437–52.
Grieco SF, Wang G, Mahapatra A, Lai C, Holmes TC, Xu X. Neuregulin and ErbB expression is regulated by development and sensory experience in mouse visual cortex. J Comp Neurol. 2020;528:419–32.
Domínguez S, Rey CC, Therreau L, Fanton A, Massotte D, Verret L, et al. Maturation of PNN and ErbB4 signaling in area CA2 during adolescence underlies the emergence of PV interneuron plasticity and social memory. Cell Rep. 2019;29:1099–112.
Sun Y, Ikrar T, Davis MF, Gong N, Zheng X, Luo ZD, et al. Neuregulin-1/ErbB4 signaling regulates visual cortical plasticity. Neuron 2016;92:160–73.
Yang JM, Zhang J, Chen XJ, Geng HY, Ye M, Spitzer NC, et al. Development of GABA circuitry of fast-spiking basket interneurons in the medial prefrontal cortex of erbb4-mutant mice. J Neurosci. 2013;33:19724–33.
Chen YJ, Zhang M, Yin DM, Wen L, Ting A, Wang P, et al. ErbB4 in parvalbumin-positive interneurons is critical for neuregulin 1 regulation of long-term potentiation. Proc Natl Acad Sci USA. 2010;107:21818–23.
Corfas G, Roy K, Buxbaum JD. Neuregulin 1-erbB signaling and the molecular/cellular basis of schizophrenia. Nat Neurosci. 2004;7:575–80.
Barros CS, Calabrese B, Chamero P, Roberts AJ, Korzus E, Lloyd K, et al. Impaired maturation of dendritic spines without disorganization of cortical cell layers in mice lacking NRG1/ErbB signaling in the central nervous system. Proc Natl Acad Sci USA. 2009;106:4507–12.
Xu Y, Hou XY, Liu Y, Zong YY. Different protection of K252a and N-acetyl-L-cysteine against amyloid-beta peptide-induced cortical neuron apoptosis involving inhibition of MLK3-MKK7-JNK3 signal cascades. J Neurosci Res. 2009;87:918–27.
Janssen MJ, Leiva-Salcedo E, Buonanno A. Neuregulin directly decreases voltage-gated sodium current in hippocampal ErbB4-expressing interneurons. J Neurosci. 2012;32:13889–95.
Du CP, Wang M, Geng C, Hu B, Meng L, Xu Y, et al. Activity-induced SUMOylation of neuronal nitric oxide synthase is associated with plasticity of synaptic transmission and extracellular signal-regulated kinase 1/2 signaling. Antioxid Redox Signal. 2020;32:18–34.
Zhu QJ, Kong FS, Xu H, Wang Y, Du CP, Sun CC, et al. Tyrosine phosphorylation of GluK2 up-regulates kainate receptor-mediated responses and downstream signaling after brain ischemia. Proc Natl Acad Sci USA. 2014;111:13990–95.
Foggetti A, Baccini G, Arnold P, Schiffelholz T, Wulff P. Spiny and non-spiny parvalbumin-positive hippocampal interneurons show different plastic properties. Cell Rep. 2019;27:3725–32.
Kehagia AA, Murray GK, Robbins TW. Learning and cognitive flexibility: frontostriatal function and monoaminergic modulation. Curr Opin Neurobiol. 2010;20:199–204.
Takehara-Nishiuchi K, McNaughton BL. Spontaneous changes of neocortical code for associative memory during consolidation. Science 2008;322:960–3.
Anderson GR, Aoto J, Tabuchi K, Földy C, Covy J, Yee AX, et al. β-Neurexins control neural circuits by regulating synaptic endocannabinoid signaling. Cell 2015;162:593–606.
Futai K, Doty CD, Baek B, Ryu J, Sheng M. Specific trans-synaptic interaction with inhibitory interneuronal neurexin underlies differential ability of neuroligins to induce functional inhibitory synapses. J Neurosci. 2013;33:3612–23.
Shipman SL, Nicoll RA. Dimerization of postsynaptic neuroligin drives synaptic assembly via transsynaptic clustering of neurexin. Proc Natl Acad Sci USA. 2012;109:19432–7.
Donato F, Chowdhury A, Lahr M, Caroni P. Early- and late-born parvalbumin basket cell subpopulations exhibiting distinct regulation and roles in learning. Neuron 2015;85:770–86.
Südhof TC. Neuroligins and neurexins link synaptic function to cognitive disease. Nature 2008;455:903–11.
Ma L, Huang YZ, Pitcher GM, Valtschanoff JG, Ma YH, Feng LY, et al. Ligand-dependent recruitment of the ErbB4 signaling complex into neuronal lipid rafts. J Neurosci. 2003;23:3164–75.
Meng L, Du CP, Lu CY, Zhang K, Li L, Yan JZ, et al. Neuronal activity-induced SUMOylation of Akt1 by PIAS3 is required for long-term potentiation of synaptic transmission. FASEB J. 2021;35:e21769.
Yang JM, Shen CJ, Chen XJ, Kong Y, Liu YS, Li XW, et al. erbb4 deficits in chandelier cells of the medial prefrontal cortex confer cognitive dysfunctions: implications for schizophrenia. Cereb Cortex. 2019;29:4334–46.
Epp JR, Silva Mera R, Köhler S, Josselyn SA, Frankland PW. Neurogenesis-mediated forgetting minimizes proactive interference. Nat Commun. 2016;7:10838.
Madan CR, Fujiwara E, Gerson BC, Caplan JB. High reward makes items easier to remember, but harder to bind to a new temporal context. Front Integr Neurosci. 2012;6:61.
Jung MW, Baeg EH, Kim MJ, Kim YB, Kim JJ. Plasticity and memory in the prefrontal cortex. Rev Neurosci. 2008;19:29–46.
Jeong H, Kim D, Song M, Paik SB, Jung MW. Distinct roles of parvalbumin- and somatostatin-expressing neurons in flexible representation of task variables in the prefrontal cortex. Prog Neurobiol. 2020;187:101773.
Guise KG, Shapiro ML. Medial prefrontal cortex reduces memory interference by modifying hippocampal encoding. Neuron 2017;94:183–92.
Whitmire CJ, Stanley GB. Rapid sensory adaptation redux: a circuit perspective. Neuron 2016;92:298–315.
Winstanley CA, Floresco SB. Deciphering decision making: variation in animal models of effort- and uncertainty-based choice reveals distinct neural circuitries underlying core cognitive processes. J Neurosci. 2016;36:12069–79.
Chen LY, Jiang M, Zhang B, Gokce O, Südhof TC. Conditional deletion of all neurexins defines diversity of essential synaptic organizer functions for neurexins. Neuron 2017;94:611–25.
Li MY, Miao WY, Wu QZ, He SJ, Yan G, Yang Y, et al. A critical role of presynaptic Cadherin/Catenin/p140Cap complexes in stabilizing spines and functional synapses in the neocortex. Neuron 2017;94:1155–72.
Sytnyk V, Leshchyns’ka I, Schachner M. Neural cell adhesion molecules of the immunoglobulin superfamily regulate synapse formation, maintenance, and function. Trends Neurosci. 2017;40:295–308.
Dean C, Scholl FG, Choih J, DeMaria S, Berger J, Isacoff E, et al. Neurexin mediates the assembly of presynaptic terminals. Nat Neurosci. 2003;6:708–16.
Sugita S, Saito F, Tang J, Satz J, Campbell K, Südhof TC. A stoichiometric complex of neurexins and dystroglycan in brain. J Cell Biol. 2001;154:435–45.
Südhof TC. Synaptic neurexin complexes: a molucular code for the logic of neural circuits. Cell 2017;171:745–69.
Ibata K, Kono M, Narumi S, Motohashi J, Kakegawa W, Kohda K, et al. Activity-dependent secretion of synaptic organizer Cbln1 from lysosomes in granule cell axons. Neuron 2019;102:1184–98.
Peixoto RT, Kunz PA, Kwon H, Mabb AM, Sabatini BL, Philpot BD, et al. Transsynaptic signaling by activity-dependent cleavage of neuroligin-1. Neuron 2012;76:396–409.
Meis S, Endres T, Munsch T, Lessmann V. Impact of chronic BDNF depletion on GABAergic synaptic transmission in the lateral amygdala. Int J Mol Sci. 2019;20:4310.
Duman RS, Deyama S, Fogaça MV. Role of BDNF in the pathophysiology and treatment of depression: activity-dependent effects distinguish rapid-acting antidepressants. Eur J Neurosci. 2021;53:126–39.
Czéh B, Vardya I, Varga Z, Febbraro F, Csabai D, Martis LS, et al. Long-term stress disrupts the structural and functional integrity of GABAergic neuronal networks in the medial prefrontal cortex of rats. Front Cell Neurosci. 2018;12:148.
Samrani G, Bäckman L, Persson J. Age-differences in the temporal properties of proactive interference in working memory. Psychol Aging. 2017;32:722–31.
Smets J, Wessel I, Raes F. Reduced autobiographical memory specificity relates to weak resistance to proactive interference. J Behav Ther Exp Psychiatry. 2014;45:234–41.
Daneshvar S, Taghavi MR, Goodarzi MA, Jobson L. Emotionally valenced and modality-specific dual tasks: effects on voluntary reminding and proactive interference in trauma-exposed individuals suffering from PTSD. Psychol Trauma. 2021;13:586–95.
Acknowledgements
We would like to thank Prof. Lin Mei at Case Western Reserve University and Prof. Yanmei Tao at Hangzhou Normal University for helpful discussions on experimental design. We also thank Prof. Lin Mei for kindly providing PV-Cre mice, floxed Erbb4 mice, and NRXN1βNTF plasmids.
Funding
This work was supported by grants from the National Natural Science Foundation of China (81673418 and 81473185) and a project founded by the Jiangsu 333 program (BRA2018059).
Author information
Authors and Affiliations
Contributions
Design of experiments: YX and XYH. Investigation and data acquisition: YX, MLW, HT, CG, FG, BH, RW. Data analysis: YX, MLW, CG, and BH. Writing-original draft: YX. Conception, revision of manuscript, and funding: XYH. All authors approved the final manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Xu, Y., Wang, ML., Tao, H. et al. ErbB4 in parvalbumin-positive interneurons mediates proactive interference in olfactory associative reversal learning. Neuropsychopharmacol. 47, 1292–1303 (2022). https://doi.org/10.1038/s41386-021-01205-0
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41386-021-01205-0
This article is cited by
-
Targeted activation of ErbB4 receptor ameliorates neuronal deficits and neuroinflammation in a food-borne polystyrene microplastic exposed mouse model
Journal of Neuroinflammation (2025)
-
Neuronal LAMP2A-mediated reduction of adenylyl cyclases induces acute neurodegenerative responses and neuroinflammation after ischemic stroke
Cell Death & Differentiation (2025)
-
Second-order threat conditioning in the amygdala-posterior piriform cortex network
Communications Biology (2025)
-
Prefrontal parvalbumin neurons as a target for enhancing cognition in non-pathological and 22q11.2 microdeletion syndrome mice
Molecular Psychiatry (2025)
-
Functional diversities within neurons and astrocytes in the adult rat auditory cortex revealed by single-nucleus RNA sequencing
Scientific Reports (2024)