Abstract
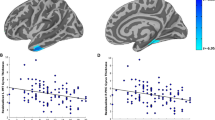
Female individuals are more likely to be diagnosed with PTSD following trauma exposure than males, potentially due, in part, to underlying neurobiological factors. Several brain regions underlying fear learning and expression have previously been associated with PTSD, with the hippocampus, amygdala, dorsal anterior cingulate cortex (dACC), and rostral ACC (rACC) showing altered volume and function in those with PTSD. However, few studies have examined how sex impacts the predictive value of subcortical volumes and cortical thickness in longitudinal PTSD studies. As part of an emergency department study completed at the Grady Trauma Project in Atlanta, GA, N = 93 (40 Female) participants were enrolled within 24 h following a traumatic event. Multi-echo T1-weighted MRI images were collected one-month post-trauma exposure. Bilateral amygdala and hippocampal volumes and rACC and dACC cortical thickness were segmented. To assess the longitudinal course of PTSD, the PTSD Symptom Scale (PSS) was collected 6 months post-trauma. We investigated whether regional volume/thickness interacted with sex to predict later PTSD symptom severity, controlling for PSS score at time of scan, age, race, and trauma type, as well as intracranial volume (ICV) for subcortical volumes. There was a significant interaction between sex and rACC for 6-month PSS, such that right rACC thickness was positively correlated with 6-month PSS scores in females, but not in males. In examining PTSD symptom subtypes and depression symptoms, greater rACC thickness in females predicted greater avoidance symptoms, while smaller rACC thickness in males predicted greater depression symptoms. Amygdala and hippocampus volume and dACC thickness showed no main effect or interaction with sex. The current findings provide evidence for sex-based differences in how brain volume predicts future PTSD severity and symptoms and supports the rACC as being a vital region regarding PTSD. Gender differences should be assessed in future longitudinal PTSD MRI studies for more accurate identification of future PTSD risk following trauma.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Tolin DF, Foa EB. Sex differences in trauma and posttraumatic stress disorder: a quantitative review of 25 years of research. Psychol Trauma. 2008;S:37–85.
Olff M, Langeland W, Draijer N, Gersons BPR. Gender differences in posttraumatic stress disorder. Psychological Bull. 2007;133:183–204.
Dell’Osso L, Carmassi C, Massimetti G, Stratta P, Riccardi I, Capanna C, et al. Age, gender and epicenter proximity effects on post-traumatic stress symptoms in L’Aquila 2009 earthquake survivors. J Affect Disord. 2013;146:174–80.
Kline A, Ciccone DS, Weiner M, Interian A, St Hill L, Falca-Dodson M, et al. Gender differences in the risk and protective factors associated with PTSD: a prospective study of National Guard troops deployed to Iraq. Psychiatry. 2013;76:256–72.
Kessler RC, Sonnega A, Bromet E, Hughes M, Nelson CB. Posttraumatic stress disorder in the National Comorbidity Survey. Arch Gen Psychiatry. 1995;52:1048–60.
Breslau N, Chilcoat HD, Kessler RC, Peterson EL, Lucia VC. Vulnerability to assaultive violence: further specification of the sex difference in post-traumatic stress disorder. Psychol Med. 1999;29:813–21.
Phelps EA, LeDoux JE. Contributions of the amygdala to emotion processing: from animal models to human behavior. Neuron 2005;48:175–87.
Maren S, Phan KL, Liberzon I. The contextual brain: implications for fear conditioning, extinction and psychopathology. Nat Rev Neurosci. 2013;14:417–28.
Garfinkel SN, Abelson JL, King AP, Sripada RK, Wang X, Gaines LM, et al. Impaired contextual modulation of memories in PTSD: an fMRI and psychophysiological study of extinction retention and fear renewal. J Neurosci. 2014;34:13435–43.
Stevens JS, Jovanovic T, Fani N, Ely TD, Glover EM, Bradley B, et al. Disrupted amygdala-prefrontal functional connectivity in civilian women with posttraumatic stress disorder. J Psychiatr Res. 2013;47:1469–78.
Milad MR, Pitman RK, Ellis CB, Gold AL, Shin LM, Lasko NB, et al. Neurobiological basis of failure to recall extinction memory in posttraumatic stress disorder. Biol Psychiatry. 2009;66:1075–82.
Shin LM, Whalen PJ, Pitman RK, Bush G, Macklin ML, Lasko NB, et al. An fMRI study of anterior cingulate function in posttraumatic stress disorder. Biol Psychiatry. 2001;50:932–42.
Shin LM, Wright CI, Cannistraro PA, Wedig MM, McMullin K, Martis B, et al. A functional magnetic resonance imaging study of amygdala and medial prefrontal cortex responses to overtly presented fearful faces in posttraumatic stress disorder. Arch Gen Psychiatry. 2005;62:273–81.
Williams LM, Kemp AH, Felmingham K, Barton M, Olivieri G, Peduto A, et al. Trauma modulates amygdala and medial prefrontal responses to consciously attended fear. Neuroimage. 2006;29:347–57.
Bush G, Luu P, Posner MI. Cognitive and emotional influences in anterior cingulate cortex. Trends Cogn Sci. 2000;4:215–22.
Rauch SL, Whalen PJ, Shin LM, McInerney SC, Macklin ML, Lasko NB, et al. Exaggerated amygdala response to masked facial stimuli in posttraumatic stress disorder: a functional MRI study. Biol Psychiatry. 2000;47:769–76.
Stevens JS, Kim YJ, Galatzer-Levy IR, Reddy R, Ely TD, Nemeroff CB, et al. Amygdala reactivity and anterior cingulate habituation predict posttraumatic stress disorder symptom maintenance after acute civilian trauma. Biol Psychiatry. 2017;81:1023–9.
Etkin A, Wager TD. Functional neuroimaging of anxiety: a meta-analysis of emotional processing in PTSD, social anxiety disorder, and specific phobia. Am J Psychiatry. 2007;164:1476–88.
Logue MW, van Rooij SJH, Dennis EL, Davis SL, Hayes JP, Stevens JS, et al. Smaller Hippocampal volume in posttraumatic stress disorder: a multisite ENIGMA-PGC study: subcortical volumetry results from Posttraumatic Stress Disorder Consortia. Biol Psychiatry. 2018;83:244–53.
Morey RA, Gold AL, LaBar KS, Beall SK, Brown VM, Haswell CC, et al. Amygdala volume changes in posttraumatic stress disorder in a large case-controlled veterans group. Arch Gen Psychiatry. 2012;69:1169–78.
Papagni SA, Benetti S, Arulanantham S, McCrory E, McGuire P, Mechelli A. Effects of stressful life events on human brain structure: a longitudinal voxel-based morphometry study. Stress. 2011;14:227–32.
Xie H, Claycomb Erwin M, Elhai JD, Wall JT, Tamburrino MB, Brickman KR, et al. Relationship of Hippocampal volumes and posttraumatic stress disorder symptoms over early posttrauma periods. Biol Psychiatry Cogn Neurosci Neuroimaging. 2018;3:968–75.
Ben-Zion Z, Artzi M, Niry D, Keynan NJ, Zeevi Y, Admon R, et al. Neuroanatomical risk factors for posttraumatic stress disorder in recent trauma survivors. Biol Psychiatry Cogn Neurosci Neuroimaging. 2020;5:311–9.
Quidé Y, Andersson F, Dufour-Rainfray D, Descriaud C, Brizard B, Gissot V, et al. Smaller hippocampal volume following sexual assault in women is associated with post-traumatic stress disorder. Acta Psychiatr Scand. 2018;138:312–24.
Koch SBJ, van Ast VA, Kaldewaij R, Hashemi MM, Zhang W, Klumpers F, et al. Larger dentate gyrus volume as predisposing resilience factor for the development of trauma-related symptoms. Neuropsychopharmacology. 2021;46:1283–92.
Stein MB, Yuh E, Jain S, Okonkwo DO, Mac Donald CL, Levin H, et al. Smaller Regional Brain Volumes Predict Posttraumatic Stress Disorder at 3 Months After Mild Traumatic Brain Injury. Biol Psychiatry Cogn Neurosci Neuroimaging. 2021;6:352–9.
Hu H, Sun Y, Su S, Wang Y, Qiu Y, Yang X, et al. Cortical surface area reduction in identification of subjects at high risk for post-traumatic stress disorder: A pilot study. Aust N Z J Psychiatry. 2018;52:1084–91.
Starcevic A, Postic S, Radojicic Z, Starcevic B, Milovanovic S, Ilankovic A, et al. Volumetric analysis of amygdala, hippocampus, and prefrontal cortex in therapy-naive PTSD participants. Biomed Res Int. 2014;2014:968495.
Morey RA, Clarke EK, Haswell CC, Phillips RD, Clausen AN, Mufford MS, et al. Amygdala nuclei volume and shape in military veterans with posttraumatic stress disorder. Biol Psychiatry Cogn Neurosci Neuroimaging. 2020;5:281–90.
Shors TJ, Chua C, Falduto J. Sex differences and opposite effects of stress on dendritic spine density in the male versus female hippocampus. J Neurosci. 2001;21:6292–7.
Garrett JE, Wellman CL. Chronic stress effects on dendritic morphology in medial prefrontal cortex: sex differences and estrogen dependence. Neuroscience 2009;162:195–207.
Shansky RM, Morrison JH. Stress-induced dendritic remodeling in the medial prefrontal cortex: effects of circuit, hormones and rest. Brain Res. 2009;1293:108–13.
Shansky RM, Hamo C, Hof PR, Lou W, McEwen BS, Morrison JH. Estrogen promotes stress sensitivity in a prefrontal cortex-amygdala pathway. Cereb Cortex. 2010;20:2560–7.
De Bellis MD, Hooper SR, Chen SD, Provenzale JM, Boyd BD, Glessner CE, et al. Posterior structural brain volumes differ in maltreated youth with and without chronic posttraumatic stress disorder. Dev Psychopathol. 2015;27:1555–76.
Lawson GM, Camins JS, Wisse L, Wu J, Duda JT, Cook PA, et al. Childhood socioeconomic status and childhood maltreatment: Distinct associations with brain structure. PLoS One. 2017;12:e0175690.
Woon F, Hedges DW. Gender does not moderate hippocampal volume deficits in adults with posttraumatic stress disorder: a meta-analysis. Hippocampus. 2011;21:243–52.
Hinojosa CA. Does Hippocampal volume in patients with posttraumatic stress disorder vary by trauma type? Harv Rev Psychiatry. 2022;30:118–34.
Stevens JS, Harnett NG, Lebois LAM. Brain-based biotypes of psychiatric vulnerability in the acute aftermath of trauma. Am J Psychiatry. 2021;79:1037–49.
Hinrichs R, Michopoulos V, Winters S, Rothbaum AO, Rothbaum BO, Ressler KJ, et al. Mobile assessment of heightened skin conductance in posttraumatic stress disorder. Depress Anxiety. 2017;34:502–7.
Michopoulos V, Beurel E, Gould F, Dhabhar FS, Schultebraucks K, Galatzer-Levy I, et al. Association of prospective risk for chronic PTSD symptoms with low TNFα and IFNγ concentrations in the immediate aftermath of trauma exposure. Am J Psychiatry. 2020;177:58–65.
Lalonde CS, Mekawi Y, Ethun KF, Beurel E, Gould F, Dhabhar FS, et al. Sex differences in peritraumatic inflammatory cytokines and steroid hormones contribute to prospective risk for nonremitting posttraumatic stress disorder. Chronic Stress. 2021;5:24705470211032208.
Foa EB, Rothbaum BO. Treating the trauma of rape: cognitive-behavioral therapy for PTSD. New York, NY: Guilford Press; 2001.
Foa EB, Tolin DF. Comparison of the PTSD Symptom Scale-Interview Version and the Clinician-administered PTSD scale. J Trauma Stress. 2000;13:181–91.
American Psychiatric Association. Diagnostic and statistical manual of mental disorders, 4th ed., Text Revision (DSM-IV-TR). Washington, DC: American Psychiatric Association; 2000.
Galatzer-Levy IR, Karstoft K-I, Statnikov A, Shalev AY. Quantitative forecasting of PTSD from early trauma responses: a Machine Learning application. J Psychiatr Res. 2014;59:68–76.
van Rooij SJH, Stevens JS, Ely TD, Hinrichs R, Michopoulos V, Winters SJ, et al. The role of the hippocampus in predicting future posttraumatic stress disorder symptoms in recently traumatized civilians. Biol Psychiatry. 2018;84:106–15.
Webb EK, Huggins AA, Belleau EL, Taubitz LE, Hanson JL, deRoon-Cassini TA, et al. Acute posttrauma resting-state functional connectivity of periaqueductal gray prospectively predicts posttraumatic stress disorder symptoms. Biol Psychiatry Cogn Neurosci Neuroimaging. 2020;5:891–900.
Fischl B, Salat DH, Busa E, Albert M, Dieterich M, Haselgrove C, et al. Whole brain segmentation: automated labeling of neuroanatomical structures in the human brain. Neuron. 2002;33:341–55.
Fischl B, Salat DH, van der Kouwe AJW, Makris N, Ségonne F, Quinn BT, et al. Sequence-independent segmentation of magnetic resonance images. Neuroimage. 2004;23:S69–S84.
Desikan RS, Ségonne F, Fischl B, Quinn BT, Dickerson BC, Blacker D, et al. An automated labeling system for subdividing the human cerebral cortex on MRI scans into gyral based regions of interest. Neuroimage. 2006;31:968–80.
Van Erp TG, Walton E, Hibar DP, Schmaal L, Jiang W, Glahn DC, et al. Cortical brain abnormalities in 4474 individuals with schizophrenia and 5098 control subjects via the enhancing neuro imaging genetics through meta analysis (ENIGMA) consortium. Biological psychiatry. 2018;84:644–54.
Dickerson BC, Wolk DA. Alzheimer’s Disease Neuroimaging Initiative. MRI cortical thickness biomarker predicts AD-like CSF and cognitive decline in normal adults. Neurology. 2012;78:84–90.
Durazzo TC, Mon A, Gazdzinski S, Meyerhoff DJ. Chronic cigarette smoking in alcohol dependence: associations with cortical thickness and N-acetylaspartate levels in the extended brain reward system. Addiction Biol. 2013;18:379–91.
Almeida Montes LG, Prado Alcántara H, Martínez García RB, De La Torre LB, Avila Acosta D, Duarte MG. Brain cortical thickness in ADHD: age, sex, and clinical correlations. J Atten Disord. 2013;17:641–54.
Burzynska AZ, Nagel IE, Preuschhof C, Gluth S, Bäckman L, Li S-C, et al. Cortical thickness is linked to executive functioning in adulthood and aging. Hum Brain Mapp. 2012;33:1607–20.
Wrocklage KM, Averill LA, Cobb Scott J, Averill CL, Schweinsburg B, Trejo M, et al. Cortical thickness reduction in combat exposed U.S. veterans with and without PTSD. Eur Neuropsychopharmacol. 2017;27:515–25.
Sadeh N, Spielberg JM, Logue MW, Wolf EJ, Smith AK, Lusk J, et al. SKA2 methylation is associated with decreased prefrontal cortical thickness and greater PTSD severity among trauma-exposed veterans. Mol Psychiatry. 2016;21:357–63.
Lindemer ER, Salat DH, Leritz EC, McGlinchey RE, Milberg WP. Reduced cortical thickness with increased lifetime burden of PTSD in OEF/OIF Veterans and the impact of comorbid TBI. Neuroimage Clin. 2013;2:601–11.
Wickham H, François R, Henry L, Müller K dplyr: a grammar of data manipulation. 2022. https://dplyr.tidyverse.org, https://github.com/tidyverse/dplyr.
Harrell Jr FE. Hmisc—Harrell miscellaneous—R package version 4.7-1: The Comprehensive R Archive Network web page. 2021. https://CRAN.R-project.org/package=Hmisc.
Kobayashi I, Sledjeski EM, Delahanty DL. Gender and age interact to predict the development of posttraumatic stress disorder symptoms following a motor vehicle accident. Psychol Trauma. 2019;11:328–36.
Koenen KC, Stellman JM, Stellman SD, Sommer JF Jr. Risk factors for course of posttraumatic stress disorder among Vietnam veterans: a 14-year follow-up of American Legionnaires. J Consult Clin Psychol. 2003;71:980–6.
Sibrava NJ, Bjornsson AS, Pérez Benítez ACI, Moitra E, Weisberg RB, Keller MB. Posttraumatic stress disorder in African American and Latinx adults: Clinical course and the role of racial and ethnic discrimination. Am Psychol. 2019;74:101–16.
King DW, Leskin GA, King LA, Weathers FW. Confirmatory factor analysis of the clinician-administered PTSD Scale: evidence for the dimensionality of posttraumatic stress disorder. Psychol Assess. 1998;10:90–96.
Wickham H. Data analysis. In: Wickham H, editor. ggplot2: elegant graphics for data analysis. Cham: Springer International Publishing; 2016;189–201.
Yehuda R, Hoge CW, McFarlane AC, Vermetten E, Lanius RA, Nievergelt CM, et al. Post-traumatic stress disorder. Nat Rev Dis Prim. 2015;1:15057.
Lanius RA, Vermetten E, Loewenstein RJ, Brand B, Schmahl C, Bremner JD, et al. Emotion Modulation in PTSD: clinical and Neurobiological Evidence for a Dissociative Subtype. AJP. 2010;167:640–7.
Frewen PA, Lanius RA. Toward a psychobiology of posttraumatic self-dysregulation: reexperiencing, hyperarousal, dissociation, and emotional numbing. Ann N Y Acad Sci. 2006;1071:110–24.
Lebois LAM, Li M, Baker JT, Wolff JD, Wang D, Lambros AM, et al. Large-scale functional brain network architecture changes associated with trauma-related dissociation. Am J Psychiatry. 2021;178:165–73.
Seligowski AV, Lebois LAM, Hill SB, Kahhale I, Wolff JD, Jovanovic T, et al. Autonomic responses to fear conditioning among women with PTSD and dissociation. Depress Anxiety. 2019;36:625–34.
Lanius RA, Brand B, Vermetten E, Frewen PA, Spiegel D. The dissociative subtype of posttraumatic stress disorder: rationale, clinical and neurobiological evidence, and implications. Depress Anxiety. 2012;29:701–8.
Crombie KM, Ross MC, Letkiewicz AM, Sartin-Tarm A, Cisler JM. Differential relationships of PTSD symptom clusters with cortical thickness and grey matter volumes among women with PTSD. Sci Rep. 2021;11:1825.
Schmaal L, for the ENIGMA-Major Depressive Disorder Working Group, Hibar DP, Sämann PG, Hall GB, Baune BT, et al. Cortical abnormalities in adults and adolescents with major depression based on brain scans from 20 cohorts worldwide in the ENIGMA Major Depressive Disorder Working Group. Mol Psychiatry. 2017;22:900–9.
Boes AD, McCormick LM, Coryell WH, Nopoulos P. Rostral anterior cingulate cortex volume correlates with depressed mood in normal healthy children. Biol Psychiatry. 2008;63:391–7.
Boes AD, Uitermarkt BD, Albazron FM, Lan MJ, Liston C, Pascual-Leone A, et al. Rostral anterior cingulate cortex is a structural correlate of repetitive TMS treatment response in depression. Brain Stimul. 2018;11:575–81.
De Bellis MD, Zisk A. The biological effects of childhood trauma. Child Adolesc Psychiatr Clin N Am. 2014;23:185–222.
Saxbe D, Khoddam H, Piero LD, Stoycos SA, Gimbel SI, Margolin G, et al. Community violence exposure in early adolescence: Longitudinal associations with hippocampal and amygdala volume and resting state connectivity. Dev Sci. 2018;21:e12686.
Whittle S, Simmons JG, Hendriksma S, Vijayakumar N, Byrne ML, Dennison M, et al. Childhood maltreatment, psychopathology, and the development of hippocampal subregions during adolescence. Brain Behav. 2017;7:e00607.
Woon FL, Hedges DW. Hippocampal and amygdala volumes in children and adults with childhood maltreatment-related posttraumatic stress disorder: a meta-analysis. Hippocampus. 2008;18:729–36.
Samplin E, Ikuta T, Malhotra AK, Szeszko PR, DeRosse P. Sex differences in resilience to childhood maltreatment: effects of trauma history on hippocampal volume, general cognition and subclinical psychosis in healthy adults. J Psychiatr Res. 2013;47:1174–9.
Rinne-Albers MA, Pannekoek JN, van Hoof M-J, van Lang ND, Lamers-Winkelman F, Rombouts SA, et al. Anterior cingulate cortex grey matter volume abnormalities in adolescents with PTSD after childhood sexual abuse. Eur Neuropsychopharmacol. 2017;27:1163–71.
Glover EM, Jovanovic T, Mercer KB, Kerley K, Bradley B, Ressler KJ, et al. Estrogen levels are associated with extinction deficits in women with posttraumatic stress disorder. Biol Psychiatry. 2012;72:19–24.
Milad MR, Zeidan MA, Contero A, Pitman RK, Klibanski A, Rauch SL, et al. The influence of gonadal hormones on conditioned fear extinction in healthy humans. Neuroscience. 2010;168:652–8.
Ravi M, Stevens JS, Michopoulos V. Neuroendocrine pathways underlying risk and resilience to PTSD in women. Front Neuroendocrinol. 2019;55:100790.
Zeidan MA, Igoe SA, Linnman C, Vitalo A, Levine JB, Klibanski A, et al. Estradiol modulates medial prefrontal cortex and amygdala activity during fear extinction in women and female rats. Biol Psychiatry. 2011;70:920–7.
Marek S, Tervo-Clemmens B, Calabro FJ, Montez DF, Kay BP, Hatoum AS, et al. Reproducible brain-wide association studies require thousands of individuals. Nature. 2022;603:654–60.
Galatzer-Levy IR, Nickerson A, Litz BT, Marmar CR. Patterns of lifetime PTSD comorbidity: a latent class analysis. Depress Anxiety. 2013;30:489–96.
Funding
This research was supported by the National Institute of Mental Health (R01 MH094757 [to KJR], R21 MH106902 [to TJ], R01 MH117009 [to JSS], F31 MH126623 [to ARR]), and a NARSAD award from the Brain & Behavior Research Foundation to BOR.
Author information
Authors and Affiliations
Contributions
Design and conceptualization of the study: ARR, SS, VM, BOR, TJ, KJR, JSS. Data collection and recruitment: VM, RH, SJHVR, JSS. Data processing and statistical analyses: ARR, SS, VM, JSS. Initial drafting of the paper: ARR, SS, JSS. All authors revised the paper critically for important intellectual context and agree to be accountable for all aspects of the work, and ensure the accuracy and integrity of the findings.
Corresponding author
Ethics declarations
Competing interests
KJR has received consulting fees or sponsored research support from Alkermes, BrainsWay, and Genomind, and he serves on scientific advisory boards for Janssen, Takeda, and Verily. KJR and JSS are on the NPP Editorial Board. The remaining authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Roeckner, A.R., Sogani, S., Michopoulos, V. et al. Sex-dependent risk factors for PTSD: a prospective structural MRI study. Neuropsychopharmacol. 47, 2213–2220 (2022). https://doi.org/10.1038/s41386-022-01452-9
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/s41386-022-01452-9
This article is cited by
-
Relationships among peri-traumatic circulating endocannabinoids and long-term, negative outcomes following traumatic injury
Psychopharmacology (2025)
-
Poor sleep and decreased cortical thickness in veterans with mild traumatic brain injury and post-traumatic stress disorder
Military Medical Research (2024)
-
Chemogenetic activation of CRF neurons as a model of chronic stress produces sex-specific physiological and behavioral effects
Neuropsychopharmacology (2024)
-
Sex-Based Contributors to and Consequences of Post-traumatic Stress Disorder
Current Psychiatry Reports (2023)
-
Post-traumatic Stress Disorder: Focus on Neuroinflammation
Molecular Neurobiology (2023)