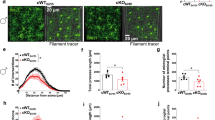
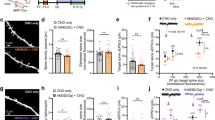
Abstract
Serotonin (5-hydroxytryptamine, 5-HT) is a powerful modulator of neuronal activity within the central nervous system and dysfunctions of the serotonergic system have been linked to several neuropsychiatric disorders such as major depressive disorders or schizophrenia. The anterior cingulate cortex (aCC) plays an important role in cognitive capture of stimuli and valence processing and it is densely innervated by serotonergic fibers from the nucleus raphe. In order to understand how pathophysiological 5-HT signalling can lead to neuropsychiatric diseases, it is important to understand the physiological actions of 5-HT on cortical circuits. Therefore, we combined electrophysiological recordings with pharmacology and immunocytochemistry to investigate the effects of 5-HT on Somatostatin-positive interneurons (SOM-INs) and compared these to supragranular pyramidal cells (PCs). This comparison allowed us to identify common and contrasting effects of 5-HT on SOM-INs and PCs of the aCC resulting in a specific modulation of the excitation-to-inhibition balance in PCs but not in SOM-INs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 13 print issues and online access
$259.00 per year
only $19.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data analyzed in this study are included in this published article.
References
Groenewegen HJ, Uylings HB. The prefrontal cortex and the integration of sensory, limbic and autonomic information. Prog Brain Res. 2000;126:3–28. https://doi.org/10.1016/S0079-6123(00)26003-2.
Rikhye RV, Gilra A, Halassa MM. Thalamic regulation of switching between cortical representations enables cognitive flexibility. Nat Neurosci. 2018;21:1753–63. https://doi.org/10.1038/s41593-018-0269-z.
Ren J, Friedmann D, Xiong J, Liu CD, Ferguson BR, Weerakkody T, et al. Anatomically Defined and Functionally Distinct Dorsal Raphe Serotonin Sub-systems. Cell. 2018;175:472–87.e20. https://doi.org/10.1016/j.cell.2018.07.043.
Clarke HF, Dalley JW, Crofts HS, Robbins TW, Roberts AC. Cognitive inflexibility after prefrontal serotonin depletion. Science. 2004;304:878–80. https://doi.org/10.1126/science.1094987.
Hamani C, Mayberg H, Stone S, Laxton A, Haber S, Lozano AM. The subcallosal cingulate gyrus in the context of major depression. Biol Psychiatry. 2011;69:301–8. https://doi.org/10.1016/j.biopsych.2010.09.034.
Miranda L. Antidepressant and anxiolytic effects of activating 5HT2A receptors in the anterior cingulate cortex and the theoretical mechanisms underlying them - A scoping review of available literature. Brain Res. 2024;1846:149226. https://doi.org/10.1016/j.brainres.2024.149226.
Ghaderi A, Brown EC, Clark DL, Ramasubbu R, Kiss ZHT, Protzner AB. Role of the serotonergic system in subcallosal DBS for treatment-resistant depression. Brain Stimul. 2022;15:211–3. https://doi.org/10.1016/j.brs.2021.12.009.
Rojas PS, Fiedler JL. What Do We Really Know About 5-HT1A Receptor Signaling in Neuronal Cells? Front Cell Neurosci. 2016;10:272. https://doi.org/10.3389/fncel.2016.00272.
Puig MV, Gulledge AT. Serotonin and prefrontal cortex function: neurons, networks, and circuits. Mol Neurobiol. 2011;44:449–64. https://doi.org/10.1007/s12035-011-8214-0.
Czyrak A, Czepiel K, Mackowiak M, Chocyk A, Wedzony K. Serotonin 5-HT1A receptors might control the output of cortical glutamatergic neurons in rat cingulate cortex. Brain Res. 2003;989:42–51. https://doi.org/10.1016/s0006-8993(03)03352-3.
DeFelipe J, Arellano JI, Gomez A, Azmitia EC, Munoz A. Pyramidal cell axons show a local specialization for GABA and 5-HT inputs in monkey and human cerebral cortex. J Comp Neurol. 2001;433:148–55. https://doi.org/10.1002/cne.1132.
Jakab RL, Goldman-Rakic PS. 5-Hydroxytryptamine2A serotonin receptors in the primate cerebral cortex: possible site of action of hallucinogenic and antipsychotic drugs in pyramidal cell apical dendrites. Proc Natl Acad Sci USA. 1998;95:735–40. https://doi.org/10.1073/pnas.95.2.735.
Hagberg GB, Blomstrand F, Nilsson M, Tamir H, Hansson E. Stimulation of 5-HT2A receptors on astrocytes in primary culture opens voltage-independent Ca2+ channels. Neurochem Int. 1998;32:153–62. https://doi.org/10.1016/s0197-0186(97)00087-9.
Stephens EK, Baker AL, Gulledge AT. Mechanisms Underlying Serotonergic Excitation of Callosal Projection Neurons in the Mouse Medial Prefrontal Cortex. Front Neural Circuits. 2018;12:2. https://doi.org/10.3389/fncir.2018.00002.
Santello M, Bisco A, Nevian NE, Lacivita E, Leopoldo M, Nevian T. The brain-penetrant 5-HT(7) receptor agonist LP-211 reduces the sensory and affective components of neuropathic pain. Neurobiol Dis. 2017;106:214–21. https://doi.org/10.1016/j.nbd.2017.07.005.
Puig MV, Watakabe A, Ushimaru M, Yamamori T, Kawaguchi Y. Serotonin modulates fast-spiking interneuron and synchronous activity in the rat prefrontal cortex through 5-HT1A and 5-HT2A receptors. J Neurosci. 2010;30:2211–22. https://doi.org/10.1523/JNEUROSCI.3335-09.2010.
Goodfellow NM, Benekareddy M, Vaidya VA, Lambe EK. Layer II/III of the prefrontal cortex: Inhibition by the serotonin 5-HT1A receptor in development and stress. J Neurosci. 2009;29:10094–103. https://doi.org/10.1523/JNEUROSCI.1960-09.2009.
Araneda R, Andrade R. 5-Hydroxytryptamine2 and 5-hydroxytryptamine 1A receptors mediate opposing responses on membrane excitability in rat association cortex. Neuroscience. 1991;40:399–412. https://doi.org/10.1016/0306-4522(91)90128-b.
Amargos-Bosch M, Bortolozzi A, Puig MV, Serrats J, Adell A, Celada P, et al. Co-expression and in vivo interaction of serotonin1A and serotonin2A receptors in pyramidal neurons of prefrontal cortex. Cereb Cortex. 2004;14:281–99. https://doi.org/10.1093/cercor/bhg128.
Avesar D, Gulledge AT. Selective serotonergic excitation of callosal projection neurons. Front Neural Circuits. 2012;6:12.
Elliott MC, Tanaka PM, Schwark RW, Andrade R Serotonin Differentially Regulates L5 Pyramidal Cell Classes of the Medial Prefrontal Cortex in Rats and Mice. eNeuro. 2018;5. https://doi.org/10.1523/ENEURO.0305-17.2018.
Stephens EK, Avesar D, Gulledge AT. Activity-dependent serotonergic excitation of callosal projection neurons in the mouse prefrontal cortex. Front Neural Circuits. 2014;8:97 https://doi.org/10.3389/fncir.2014.00097.
Hostetler RE, Hu H, Agmon A Genetically Defined Subtypes of Somatostatin-Containing Cortical Interneurons. eNeuro. 2023;10. https://doi.org/10.1523/ENEURO.0204-23.2023.
Tremblay R, Lee S, Rudy B. GABAergic Interneurons in the Neocortex: From Cellular Properties to Circuits. Neuron. 2016;91:260–92. https://doi.org/10.1016/j.neuron.2016.06.033.
de Filippo R, Rost BR, Stumpf A, Cooper C, Tukker JJ, Harms C, et al. Somatostatin interneurons activated by 5-HT(2A) receptor suppress slow oscillations in medial entorhinal cortex. Elife. 2021;10. https://doi.org/10.7554/eLife.66960.
Kim D, Jeong H, Lee J, Ghim JW, Her ES, Lee SH, et al. Distinct Roles of Parvalbumin- and Somatostatin-Expressing Interneurons in Working Memory. Neuron. 2016;92:902–15. https://doi.org/10.1016/j.neuron.2016.09.023.
Williams GV, Rao SG, Goldman-Rakic PS. The physiological role of 5-HT2A receptors in working memory. J Neurosci. 2002;22:2843–54. https://doi.org/10.1523/JNEUROSCI.22-07-02843.2002.
Seney ML, Tripp A, McCune S, Lewis DA, Sibille E. Laminar and cellular analyses of reduced somatostatin gene expression in the subgenual anterior cingulate cortex in major depression. Neurobiol Dis. 2015;73:213–9. https://doi.org/10.1016/j.nbd.2014.10.005.
Tripp A, Kota RS, Lewis DA, Sibille E. Reduced somatostatin in subgenual anterior cingulate cortex in major depression. Neurobiol Dis. 2011;42:116–24. https://doi.org/10.1016/j.nbd.2011.01.014.
Liguz-Lecznar M, Dobrzanski G, Kossut M. Somatostatin and Somatostatin-Containing Interneurons-From Plasticity to Pathology. Biomolecules. 2022;12. https://doi.org/10.3390/biom12020312.
Scheggia D, Manago F, Maltese F, Bruni S, Nigro M, Dautan D, et al. Somatostatin interneurons in the prefrontal cortex control affective state discrimination in mice. Nat Neurosci. 2020;23:47–60. https://doi.org/10.1038/s41593-019-0551-8.
Teissier A, Chemiakine A, Inbar B, Bagchi S, Ray RS, Palmiter RD, et al. Activity of Raphe Serotonergic Neurons Controls Emotional Behaviors. Cell Rep. 2015;13:1965–76. https://doi.org/10.1016/j.celrep.2015.10.061.
Oliva AA Jr, Jiang M, Lam T, Smith KL, Swann JW. Novel hippocampal interneuronal subtypes identified using transgenic mice that express green fluorescent protein in GABAergic interneurons. J Neurosci. 2000;20:3354–68. https://doi.org/10.1523/JNEUROSCI.20-09-03354.2000.
Riedemann T, Schmitz C, Sutor B. Immunocytochemical heterogeneity of somatostatin-expressing GABAergic interneurons in layers II and III of the mouse cingulate cortex: A combined immunofluorescence/design-based stereologic study. J Comp Neurol. 2016;524:2281–99. https://doi.org/10.1002/cne.23948.
Riedemann T, Straub T, Sutor B. Two types of somatostatin-expressing GABAergic interneurons in the superficial layers of the mouse cingulate cortex. PLoS One. 2018;13:e0200567. https://doi.org/10.1371/journal.pone.0200567.
Riedemann T, Sutor B Cell-Type-Specific Effects of Somatostatin on Synaptic Transmission in the Anterior Cingulate Cortex. J Neurosci. 2024;44. https://doi.org/10.1523/JNEUROSCI.0598-23.2024.
Riedemann T, Polder HR, Sutor B Determination and compensation of series resistances during whole-cell patch-clamp recordings using an active bridge circuit and the phase-sensitive technique. Pflugers Arch. 2016;468:1725-40. https://doi.org/10.1007/s00424-016-1868-8.
Rothman JS, Silver RA. NeuroMatic: An Integrated Open-Source Software Toolkit for Acquisition, Analysis and Simulation of Electrophysiological Data. Front Neuroinform. 2018;12:14 https://doi.org/10.3389/fninf.2018.00014.
Santana N, Artigas F. Laminar and Cellular Distribution of Monoamine Receptors in Rat Medial Prefrontal Cortex. Front Neuroanat. 2017;11:87. https://doi.org/10.3389/fnana.2017.00087.
Athilingam JC, Ben-Shalom R, Keeshen CM, Sohal VS, Bender KJ Serotonin enhances excitability and gamma frequency temporal integration in mouse prefrontal fast-spiking interneurons. Elife. 2017;6. https://doi.org/10.7554/eLife.31991.
Ferezou I, Cauli B, Hill EL, Rossier J, Hamel E, Lambolez B. 5-HT3 receptors mediate serotonergic fast synaptic excitation of neocortical vasoactive intestinal peptide/cholecystokinin interneurons. J Neurosci. 2002;22:7389–97. https://doi.org/10.1523/JNEUROSCI.22-17-07389.2002.
Higa GSV, Francis-Oliveira J, Carlos-Lima E, Tamais AM, Borges FDS, Kihara AH, et al. 5-HT-dependent synaptic plasticity of the prefrontal cortex in postnatal development. Sci Rep. 2022;12:21015. https://doi.org/10.1038/s41598-022-23767-9.
Schmitz D, Gloveli T, Empson RM, Heinemann U. Potent depression of stimulus evoked field potential responses in the medial entorhinal cortex by serotonin. Br J Pharm. 1999;128:248–54. https://doi.org/10.1038/sj.bjp.0702788.
Torres-Escalante JL, Barral JA, Ibarra-Villa MD, Perez-Burgos A, Gongora-Alfaro JL, Pineda JC. 5-HT1A, 5-HT2, and GABAB receptors interact to modulate neurotransmitter release probability in layer 2/3 somatosensory rat cortex as evaluated by the paired pulse protocol. J Neurosci Res. 2004;78:268–78. https://doi.org/10.1002/jnr.20247.
Tian Z, Yamanaka M, Bernabucci M, Zhao MG, Zhuo M. Characterization of serotonin-induced inhibition of excitatory synaptic transmission in the anterior cingulate cortex. Mol Brain. 2017;10:21. https://doi.org/10.1186/s13041-017-0303-1.
Zhou FM, Hablitz JJ. Activation of serotonin receptors modulates synaptic transmission in rat cerebral cortex. J Neurophysiol. 1999;82:2989–99. https://doi.org/10.1152/jn.1999.82.6.2989.
Beique JC, Imad M, Mladenovic L, Gingrich JA, Andrade R. Mechanism of the 5-hydroxytryptamine 2A receptor-mediated facilitation of synaptic activity in prefrontal cortex. Proc Natl Acad Sci USA. 2007;104:9870–5. https://doi.org/10.1073/pnas.0700436104.
Aghajanian GK, Marek GJ. Serotonin induces excitatory postsynaptic potentials in apical dendrites of neocortical pyramidal cells. Neuropharmacology. 1997;36:589–99. https://doi.org/10.1016/s0028-3908(97)00051-8.
Aghajanian GK, Marek GJ. Serotonin, via 5-HT2A receptors, increases EPSCs in layer V pyramidal cells of prefrontal cortex by an asynchronous mode of glutamate release. Brain Res. 1999;825:161–71. https://doi.org/10.1016/s0006-8993(99)01224-x.
Barre A, Berthoux C, De Bundel D, Valjent E, Bockaert J, Marin P, et al. Presynaptic serotonin 2A receptors modulate thalamocortical plasticity and associative learning. Proc Natl Acad Sci USA. 2016;113:E1382–91. https://doi.org/10.1073/pnas.1525586113.
Beique JC, Campbell B, Perring P, Hamblin MW, Walker P, Mladenovic L, et al. Serotonergic regulation of membrane potential in developing rat prefrontal cortex: coordinated expression of 5-hydroxytryptamine (5-HT)1A, 5-HT2A, and 5-HT7 receptors. J Neurosci. 2004;24:4807–17. https://doi.org/10.1523/JNEUROSCI.5113-03.2004.
Liu Q, Wong-Riley MT. Postnatal changes in cytochrome oxidase expressions in brain stem nuclei of rats: implications for sensitive periods. J Appl Physiol (1985). 2003;95:2285–91. https://doi.org/10.1152/japplphysiol.00638.2003.
Schwaller F, Kanellopoulos AH, Fitzgerald M. The developmental emergence of differential brainstem serotonergic control of the sensory spinal cord. Sci Rep. 2017;7:2215. https://doi.org/10.1038/s41598-017-02509-2.
Zhang ZW. Serotonin induces tonic firing in layer V pyramidal neurons of rat prefrontal cortex during postnatal development. J Neurosci. 2003;23:3373–84. https://doi.org/10.1523/JNEUROSCI.23-08-03373.2003.
Hu H, Cavendish JZ, Agmon A. Not all that glitters is gold: off-target recombination in the somatostatin-IRES-Cre mouse line labels a subset of fast-spiking interneurons. Front Neural Circuits. 2013;7:195. https://doi.org/10.3389/fncir.2013.00195.
Large AM, Kunz NA, Mielo SL, Oswald AM. Inhibition by Somatostatin Interneurons in Olfactory Cortex. Front Neural Circuits. 2016;10:62 https://doi.org/10.3389/fncir.2016.00062.
Nassar M, Simonnet J, Lofredi R, Cohen I, Savary E, Yanagawa Y, et al. Diversity and overlap of parvalbumin and somatostatin expressing interneurons in mouse presubiculum. Front Neural Circuits. 2015;9:20. https://doi.org/10.3389/fncir.2015.00020.
Nigro MJ, Hashikawa-Yamasaki Y, Rudy B. Diversity and Connectivity of Layer 5 Somatostatin-Expressing Interneurons in the Mouse Barrel Cortex. J Neurosci. 2018;38:1622–33. https://doi.org/10.1523/JNEUROSCI.2415-17.2017.
Andrade R, Nicoll RA. Novel anxiolytics discriminate between postsynaptic serotonin receptors mediating different physiological responses on single neurons of the rat hippocampus. Naunyn Schmiedebergs Arch Pharm. 1987;336:5–10. https://doi.org/10.1007/BF00177743.
Colino A, Halliwell JV. Differential modulation of three separate K-conductances in hippocampal CA1 neurons by serotonin. Nature. 1987;328:73–7. https://doi.org/10.1038/328073a0.
De Vivo M, Maayani S. Characterization of the 5-hydroxytryptamine1a receptor-mediated inhibition of forskolin-stimulated adenylate cyclase activity in guinea pig and rat hippocampal membranes. J Pharm Exp Ther. 1986;238:248–53.
Weiss S, Sebben M, Kemp DE, Bockaert J. Serotonin 5-HT1 receptors mediate inhibition of cyclic AMP production in neurons. Eur J Pharm. 1986;120:227–30. https://doi.org/10.1016/0014-2999(86)90544-3.
Zgombick JM, Beck SG, Mahle CD, Craddock-Royal B, Maayani S. Pertussis toxin-sensitive guanine nucleotide-binding protein(S) couple adenosine A1 and 5-hydroxytryptamine1A receptors to the same effector systems in rat hippocampus: biochemical and electrophysiological studies. Mol Pharm. 1989;35:484–94.
Grunschlag CR, Haas HL, Stevens DR. 5-HT inhibits lateral entorhinal cortical neurons of the rat in vitro by activation of potassium channel-coupled 5-HT1A receptors. Brain Res. 1997;770:10–7. https://doi.org/10.1016/s0006-8993(97)00738-5.
Deng PY, Poudel SK, Rojanathammanee L, Porter JE, Lei S. Serotonin inhibits neuronal excitability by activating two-pore domain k+ channels in the entorhinal cortex. Mol Pharm. 2007;72:208–18. https://doi.org/10.1124/mol.107.034389.
Berg KA, Maayani S, Goldfarb J, Scaramellini C, Leff P, Clarke WP. Effector pathway-dependent relative efficacy at serotonin type 2A and 2C receptors: evidence for agonist-directed trafficking of receptor stimulus. Mol Pharm. 1998;54:94–104.
Nakaki T, Roth BL, Chuang DM, Costa E. Phasic and tonic components in 5-HT2 receptor-mediated rat aorta contraction: participation of Ca++ channels and phospholipase C. J Pharm Exp Ther. 1985;234:442–6.
Okoro EO. Overlap in the pharmacology of L-type Ca2+-channel blockers and 5-HT2 receptor antagonists in rat aorta. J Pharm Pharm. 1999;51:953–7. https://doi.org/10.1211/0022357991773221.
Baker A, Kalmbach B, Morishima M, Kim J, Juavinett A, Li N, et al. Specialized Subpopulations of Deep-Layer Pyramidal Neurons in the Neocortex: Bridging Cellular Properties to Functional Consequences. J Neurosci. 2018;38:5441–55. https://doi.org/10.1523/JNEUROSCI.0150-18.2018.
Avesar D, Stephens EK, Gulledge AT. Serotonergic Regulation of Corticoamygdalar Neurons in the Mouse Prelimbic Cortex. Front Neural Circuits. 2018;12:63. https://doi.org/10.3389/fncir.2018.00063.
van Aerde KI, Feldmeyer D. Morphological and physiological characterization of pyramidal neuron subtypes in rat medial prefrontal cortex. Cereb Cortex. 2015;25:788–805. https://doi.org/10.1093/cercor/bht278.
Weiler S, Guggiana Nilo D, Bonhoeffer T, Hubener M, Rose T, Scheuss V. Functional and structural features of L2/3 pyramidal cells continuously covary with pial depth in mouse visual cortex. Cereb Cortex. 2023;33:3715–33. https://doi.org/10.1093/cercor/bhac303.
Gouwens NW, Sorensen SA, Berg J, Lee C, Jarsky T, Ting J, et al. Classification of electrophysiological and morphological neuron types in the mouse visual cortex. Nat Neurosci. 2019;22:1182–95. https://doi.org/10.1038/s41593-019-0417-0.
Song C, Moyer JR Jr. Layer- and subregion-specific differences in the neurophysiological properties of rat medial prefrontal cortex pyramidal neurons. J Neurophysiol. 2018;119:177–91. https://doi.org/10.1152/jn.00146.2017.
Xiang Z, Prince DA. Heterogeneous actions of serotonin on interneurons in rat visual cortex. J Neurophysiol. 2003;89:1278–87. https://doi.org/10.1152/jn.00533.2002.
Karnani MM, Jackson J, Ayzenshtat I, Hamzehei Sichani A, Manoocheri K, Kim S, et al. Opening Holes in the Blanket of Inhibition: Localized Lateral Disinhibition by VIP Interneurons. J Neurosci. 2016;36:3471–80. https://doi.org/10.1523/JNEUROSCI.3646-15.2016.
Pfeffer CK, Xue M, He M, Huang ZJ, Scanziani M. Inhibition of inhibition in visual cortex: the logic of connections between molecularly distinct interneurons. Nat Neurosci. 2013;16:1068–76. https://doi.org/10.1038/nn.3446.
Tokarski K, Kusek M, Hess G. 5-HT7 receptors modulate GABAergic transmission in rat hippocampal CA1 area. J Physiol Pharm. 2011;62:535–40.
Brys I, Barrientos SA, Ward JE, Wallander J, Petersson P, Halje P. 5-HT2AR and NMDAR psychedelics induce similar hyper-synchronous states in the rat cognitive-limbic cortex-basal ganglia system. Commun Biol. 2023;6:737. https://doi.org/10.1038/s42003-023-05093-6.
Wood J, Kim Y, Moghaddam B. Disruption of prefrontal cortex large scale neuronal activity by different classes of psychotomimetic drugs. J Neurosci. 2012;32:3022–31. https://doi.org/10.1523/JNEUROSCI.6377-11.2012.
McGinty DJ, Harper RM. Dorsal raphe neurons: depression of firing during sleep in cats. Brain Res. 1976;101:569–75. https://doi.org/10.1016/0006-8993(76)90480-7.
Menon JML, Nolten C, Achterberg EJM, Joosten R, Dematteis M, Feenstra MGP, et al. Brain Microdialysate Monoamines in Relation to Circadian Rhythms, Sleep, and Sleep Deprivation - a Systematic Review, Network Meta-analysis, and New Primary Data. J Circadian Rhythms. 2019;17:1. https://doi.org/10.5334/jcr.174.
Oikonomou G, Altermatt M, Zhang RW, Coughlin GM, Montz C, Gradinaru V, et al. The Serotonergic Raphe Promote Sleep in Zebrafish and Mice. Neuron. 2019;103:686–701.e8. https://doi.org/10.1016/j.neuron.2019.05.038.
Sanchez S, Sanchez C, Paredes SD, Cubero J, Rodriguez AB, Barriga C. Circadian variations of serotonin in plasma and different brain regions of rats. Mol Cell Biochem. 2008;317:105–11. https://doi.org/10.1007/s11010-008-9836-z.
Unger EK, Keller JP, Altermatt M, Liang R, Matsui A, Dong C, et al. Directed Evolution of a Selective and Sensitive Serotonin Sensor via Machine Learning. Cell. 2020;183:1986–2002.e26. https://doi.org/10.1016/j.cell.2020.11.040.
Bridi MCD, Zong FJ, Min X, Luo N, Tran T, Qiu J, et al. Daily Oscillation of the Excitation-Inhibition Balance in Visual Cortical Circuits. Neuron. 2020;105:621–9.e4. https://doi.org/10.1016/j.neuron.2019.11.011.
Acknowledgements
We would like to thank Gabi Horn for excellent technical assistance. Friedrich-Baur Stiftung (03/21) and FöFoLe (21/2020) provided funding for this project.
Author information
Authors and Affiliations
Contributions
SH: Data collection, analysis, revision of manuscript; NS: Conceptualization, data collection, analysis, writing of initial draft; TR: Funding acquisition, conceptualization, supervision, data collection, writing of initial draft, revision of manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Schmitz, N., Hodzic, S. & Riedemann, T. Common and contrasting effects of 5-HTergic signaling in pyramidal cells and SOM interneurons of the mouse cortex. Neuropsychopharmacol. 50, 783–797 (2025). https://doi.org/10.1038/s41386-024-02022-x
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41386-024-02022-x