Abstract
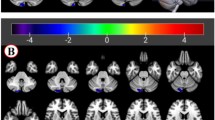
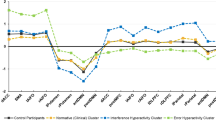
Predicting selective serotonin reuptake inhibitor (SSRI) response in obsessive–compulsive disorder (OCD) remains a clinical challenge. Converging evidence implicated that the sensorimotor circuit is linked to OCD-related sensory phenomena and repetitive motor rituals, and it is densely innervated by serotonergic projections, making it a plausible substrate of SSRI effects. We therefore hypothesized that baseline functional connectivity (FC) of this circuit could serve as a candidate neural marker of SSRI treatment response. In this exploratory single-site resting-state fMRI study, 54 drug-naïve patients with OCD and 39 matched healthy controls (HCs) underwent scanning. Patients received sertraline for 12 weeks and were classified as responders (rOCD, n = 33) or non-responders (nOCD, n = 21) based on Yale-Brown Obsessive Compulsive Scale score reductions. Seed-based FC analysis of the sensorimotor circuit was conducted across the three groups. We observed that OCD patients exhibited abnormal FC primarily within the sensorimotor circuit and in its connections with the cerebellum. The rOCD group showed generally higher FC within the sensorimotor circuit than HCs, whereas the nOCD group showed lower FC values. Cerebellar regions with altered connectivity included areas involved in sensorimotor processing and higher-level functions. In prediction analyses, the connectivity between right thalamus and cerebellar Crus I region achieved an AUC of 0.854 for distinguishing responders from non-responders under leave-one-out cross-validation. Moreover, FC-based models showed better predictive performance than clinical models. These findings suggest that baseline sensorimotor-network FC may serve as a candidate biomarker of sertraline response in OCD, pending validation in large, independent samples.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 13 print issues and online access
$259.00 per year
only $19.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
Individual-level clinical and imaging data derive from an ongoing longitudinal study, and therefore they cannot be publicly shared until the cohort’s primary results are published. Derived measures and analysis code are available from the corresponding author upon reasonable request.
References
Goodman WK, Storch EA, Sheth SA. Harmonizing the neurobiology and treatment of obsessive-compulsive disorder. Am J Psychiatry. 2021;178:17–29.
Ruscio AM, Stein DJ, Chiu WT, Kessler RC. The epidemiology of obsessive-compulsive disorder in the National Comorbidity Survey Replication. Mol Psychiatry. 2010;15:53–63.
Huang Y, Wang Y, Wang H, Liu Z, Yu X, Yan J, et al. Prevalence of mental disorders in China: a cross-sectional epidemiological study. Lancet Psychiatry. 2019;6:211–24.
Adam Y, Meinlschmidt G, Gloster AT, Lieb R. Obsessive-compulsive disorder in the community: 12-month prevalence, comorbidity and impairment. Soc Psychiatry Psychiatr Epidemiol. 2012;47:339–49.
Yang W, Tang Z, Wang X, Ma X, Cheng Y, Wang B, et al. The cost of obsessive-compulsive disorder (OCD) in China: a multi-center cross-sectional survey based on hospitals. Gen Psychiatr. 2021;34:e100632.
Romanelli RJ, Wu FM, Gamba R, Mojtabai R, Segal JB. Behavioral therapy and serotonin reuptake inhibitor pharmacotherapy in the treatment of obsessive-compulsive disorder: a systematic review and meta-analysis of head-to-head randomized controlled trials. Depress Anxiety. 2014;31:641–52.
Simpson HB, Huppert JD, Petkova E, Foa EB, Liebowitz MR. Response versus remission in obsessive-compulsive disorder. J Clin Psychiatry. 2006;67:269–76.
Hazari N, Narayanaswamy JC, Arumugham SS. Predictors of response to serotonin reuptake inhibitors in obsessive-compulsive disorder. Expert Rev Neurother. 2016;16:1175–91.
Stein DJ, Costa DLC, Lochner C, Miguel EC, Reddy YCJ, Shavitt RG, et al. Obsessive-compulsive disorder. Nat Rev Dis Prim. 2019;5:52.
Nakao T, Okada K, Kanba S. Neurobiological model of obsessive-compulsive disorder: evidence from recent neuropsychological and neuroimaging findings. Psychiatry Clin Neurosci. 2014;68:587–605.
van den Heuvel OA, van Wingen G, Soriano-Mas C, Alonso P, Chamberlain SR, Nakamae T, et al. Brain circuitry of compulsivity. Eur Neuropsychopharmacol. 2016;26:810–27.
Shephard E, Stern ER, van den Heuvel OA, Costa DLC, Batistuzzo MC, Godoy PBG, et al. Toward a neurocircuit-based taxonomy to guide treatment of obsessive-compulsive disorder. Mol Psychiatry. 2021;26:4583–604.
Bruin WB, Abe Y, Alonso P, Anticevic A, Backhausen LL, Balachander S, et al. The functional connectome in obsessive-compulsive disorder: resting-state mega-analysis and machine learning classification for the ENIGMA-OCD consortium. Mol Psychiatry. 2023;28:4307–19.
Abramovitch A, Abramowitz JS, Mittelman A. The neuropsychology of adult obsessive-compulsive disorder: a meta-analysis. Clin Psychol Rev. 2013;33:1163–71.
Lipszyc J, Schachar R. Inhibitory control and psychopathology: a meta-analysis of studies using the stop signal task. J Int Neuropsychol Soc. 2010;16:1064–76.
Zandbelt BB, Bloemendaal M, Hoogendam JM, Kahn RS, Vink M. Transcranial magnetic stimulation and functional MRI reveal cortical and subcortical interactions during stop-signal response inhibition. J Cogn Neurosci. 2013;25:157–74.
Rodriguez-Manrique D, Ruan H, Winkelmann C, Haun J, Gigl S, Berberich G, et al. Investigating the effects of brain stimulation on the neural substrates of inhibition in patients with OCD: a simultaneous tDCS - fMRI study. Transl Psychiatry. 2025;15:173.
Bär KJ, de la Cruz F, Schumann A, Koehler S, Sauer H, Critchley H, et al. Functional connectivity and network analysis of midbrain and brainstem nuclei. Neuroimage. 2016;134:53–63.
Beliveau V, Svarer C, Frokjaer VG, Knudsen GM, Greve DN, Fisher PM. Functional connectivity of the dorsal and median raphe nuclei at rest. Neuroimage. 2015;116:187–95.
Shanahan NA, Holick Pierz KA, Masten VL, Waeber C, Ansorge M, Gingrich JA, et al. Chronic reductions in serotonin transporter function prevent 5-HT1B-induced behavioral effects in mice. Biol Psychiatry. 2009;65:401–8.
Woehrle NS, Klenotich SJ, Jamnia N, Ho EV, Dulawa SC. Effects of chronic fluoxetine treatment on serotonin 1B receptor-induced deficits in delayed alternation. Psychopharmacology. 2013;227:545–51.
Pittenger C, Adams TG Jr., Gallezot JD, Crowley MJ, Nabulsi N, James R, et al. OCD is associated with an altered association between sensorimotor gating and cortical and subcortical 5-HT1b receptor binding. J Affect Disord. 2016;196:87–96.
Melo L, Beaupain MC, Ghanavati E, Kuo MF, Nitsche MA. Neurochemical mechanisms underlying serotonergic modulation of neuroplasticity in humans. Brain Stimul. 2024;17:421–30.
Kim M, Kwak S, Yoon YB, Kwak YB, Kim T, Cho KIK, et al. Functional connectivity of the raphe nucleus as a predictor of the response to selective serotonin reuptake inhibitors in obsessive-compulsive disorder. Neuropsychopharmacology. 2019;44:2073–81.
Yan H, Shan X, Li H, Liu F, Xie G, Li P, et al. Cerebellar functional connectivity and its associated genes: a longitudinal study in drug-naive patients with obsessive-compulsive disorder. J Psychiatr Res. 2024;177:378–91.
Yan H, Zhang Y, Shan X, Li H, Liu F, Xie G, et al. Altered interhemispheric functional connectivity in patients with obsessive-compulsive disorder and its potential in therapeutic response prediction. J Neurosci Res. 2024;102:25272.
Wang H, Teng C, Zhang D, Zhang W, Zhang H, Wu X, et al. Abnormal intrinsic brain activity of the sensory-motor area as a predictor of the response to selective serotonin reuptake inhibitors in treatment-naïve obsessive-compulsive disorder. J Affect Disord. 2025;388:119457.
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. Arlington, VA: American Psychiatric Association; 2013.
Goodman WK, Price LH, Rasmussen SA, Mazure C, Fleischmann RL, Hill CL, et al. The Yale-Brown obsessive compulsive scale. I. Development, use, and reliability. Arch Gen Psychiatry. 1989;46:1006–11.
Sheehan DV, Lecrubier Y, Sheehan KH, Amorim P, Janavs J, Weiller E, et al. The Mini-International Neuropsychiatric Interview (M.I.N.I.): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J Clin Psychiatry. 1998;59:22–33. quiz 34-57.
Wang P, Gu W, Gao J, Wang C, Fang J, Hu M, et al. Protocol for a pragmatic trial of pharmacotherapy options following unsatisfactory initial treatment in OCD (PROCEED). Front Psychiatry. 2022;13:822976.
Zheng YP, Wei LA, Goa LG, Zhang GC, Wong CG. Applicability of the Chinese beck depression inventory. Compr Psychiatry. 1988;29:484–9.
Che H-H, Lu M-L, Chen H-C, Chang S-W, Lee Y-J. Validation of the Chinese version of the beck anxiety inventory. J Med. 2006;10:447–54.
Farris SG, McLean CP, Van Meter PE, Simpson HB, Foa EB. Treatment response, symptom remission, and wellness in obsessive-compulsive disorder. J Clin Psychiatry. 2013;74:685–90.
Whitfield-Gabrieli S, Nieto-Castanon A. Conn: a functional connectivity toolbox for correlated and anticorrelated brain networks. Brain Connect. 2012;2:125–41.
Friston KJ, Holmes AP, Worsley KJ, Poline JP, Frith CD, Frackowiak RS. Statistical parametric maps in functional imaging: a general linear approach. Human brain mapping. 1994;2:189–210.
Kong R, Yang Q, Gordon E, Xue A, Yan X, Orban C, et al. Individual-specific areal-level parcellations improve functional connectivity prediction of behavior. Cereb Cortex. 2021;31:4477–500.
Desikan RS, Ségonne F, Fischl B, Quinn BT, Dickerson BC, Blacker D, et al. An automated labeling system for subdividing the human cerebral cortex on MRI scans into gyral based regions of interest. Neuroimage. 2006;31:968–80.
Tzourio-Mazoyer N, Landeau B, Papathanassiou D, Crivello F, Etard O, Delcroix N, et al. Automated anatomical labeling of activations in SPM using a macroscopic anatomical parcellation of the MNI MRI single-subject brain. Neuroimage. 2002;15:273–89.
Marek S, Laumann TO. Replicability and generalizability in population psychiatric neuroimaging. Neuropsychopharmacology. 2024;50:52–57.
van Wijk BCM, Alkemade A, Forstmann BU. Functional segregation and integration within the human subthalamic nucleus from a micro- and meso-level perspective. Cortex. 2020;131:103–13.
Grahn JA, Parkinson JA, Owen AM. The cognitive functions of the caudate nucleus. Prog Neurobiol. 2008;86:141–55.
Li Z, Luo J, Li C, Zhu H. Upregulation of Nav1.6 expression in the ventral posterolateral nucleus of thalamus contributes to hyperalgesia in a model of Parkinson’s disease. Exp Neurol. 2025;383:115032.
Lv D, Ou Y, Chen Y, Ding Z, Ma J, Zhan C, et al. Anatomical distance affects functional connectivity at rest in medicine-free obsessive-compulsive disorder. BMC Psychiatry. 2022;22:462.
Xu Y, Zhang Y. Abnormal voxel-mirrored homotopic connectivity in first-episode, drug-naïve patients with obsessive-compulsive disorder. Eur J Neurosci. 2023;58:3531–39.
Stern ER, Eng GK, De Nadai AS, Iosifescu DV, Tobe RH, Collins KA. Imbalance between default mode and sensorimotor connectivity is associated with perseverative thinking in obsessive-compulsive disorder. Transl Psychiatry. 2022;12:19.
Chen Y, Meng Z, Zhang Z, Zhu Y, Gao R, Cao X, et al. The right thalamic glutamate level correlates with functional connectivity with right dorsal anterior cingulate cortex/middle occipital gyrus in unmedicated obsessive-compulsive disorder: a combined fMRI and (1)H-MRS study. Aust N Z J Psychiatry. 2019;53:207–18.
Tikoo S, Cardona F, Tommasin S, Giannì C, Conte G, Upadhyay N, et al. Resting-state functional connectivity in drug-naive pediatric patients with Tourette syndrome and obsessive-compulsive disorder. J Psychiatr Res. 2020;129:129–40.
Cano M, Alonso P, Martínez-Zalacaín I, Subirà M, Real E, Segalàs C, et al. Altered functional connectivity of the subthalamus and the bed nucleus of the stria terminalis in obsessive-compulsive disorder. Psychological Med. 2018;48:919–28.
de Wit SJ, de Vries FE, van der Werf YD, Cath DC, Heslenfeld DJ, Veltman EM, et al. Presupplementary motor area hyperactivity during response inhibition: a candidate endophenotype of obsessive-compulsive disorder. Am J Psychiatry. 2012;169:1100–8.
Klaassens BL, van Gorsel HC, Khalili-Mahani N, van der Grond J, Wyman BT, Whitcher B, et al. Single-dose serotonergic stimulation shows widespread effects on functional brain connectivity. Neuroimage. 2015;122:440–50.
Chau DT, Fogelman P, Nordanskog P, Drevets WC, Hamilton JP. Distinct neural-functional effects of treatments with selective serotonin reuptake inhibitors, electroconvulsive therapy, and transcranial magnetic stimulation and their relations to regional brain function in major depression: a meta-analysis. Biol Psychiatry Cogn Neurosci Neuroimaging. 2017;2:318–26.
Lesch KP, Waider J. Serotonin in the modulation of neural plasticity and networks: implications for neurodevelopmental disorders. Neuron. 2012;76:175–91.
De Zeeuw CI, Lisberger SG, Raymond JL. Diversity and dynamism in the cerebellum. Nat Neurosci. 2021;24:160–67.
Van Overwalle F. Social and emotional learning in the cerebellum. Nat Rev Neurosci. 2024;25:776–91.
Ashida R, Cerminara NL, Edwards RJ, Apps R, Brooks JCWSensorimotor. language, and working memory representation within the human cerebellum. Hum Brain Mapp. 2019;40:4732–47.
Moberget T, Ivry RB. Prediction, psychosis, and the cerebellum. Biol Psychiatry Cogn Neurosci Neuroimaging. 2019;4:820–31.
Tonna M, Ottoni R, Ossola P, De Panfilis C, Marchesi C. Late-onset obsessive-compulsive disorder associated with left cerebellar lesion. Cerebellum. 2014;13:531–5.
Yang Z, Xiao S, Su T, Gong J, Qi Z, Chen G, et al. A multimodal meta-analysis of regional functional and structural brain abnormalities in obsessive-compulsive disorder. Eur Arch Psychiatry Clin Neurosci. 2024;274:165–80.
Tao Q, Dang J, Niu X, Gao X, Zhang M, Yang Z, et al. White matter microstructural abnormalities and gray matter volume alterations in obsessive-compulsive disorder: a coordinate-based meta-analysis. J Affect Disord. 2023;320:751–61.
Li H, Wang Y, Xi H, Zhang J, Zhao M, Jia X. Alterations of regional spontaneous brain activity in obsessive-compulsive disorders: a meta-analysis. J Psychiatr Res. 2023;165:325–35.
Tanaka H, Ishikawa T, Lee J, Kakei S. The cerebro-cerebellum as a locus of forward model: a review. Front Syst Neurosci. 2020;14:19.
Popa LS, Ebner TJ. Cerebellum, predictions and errors. Front Cell Neurosci. 2018;12:524.
Acknowledgements
This study was supported by the National Natural Science Foundation of China (82230045、82071518) and granted from the Shanghai Science and Technology Committee (25Y22800100). We acknowledge Dr. Michael William Simpson for useful discussions during the paper preparation.
Author information
Authors and Affiliations
Contributions
Tingyue Bai: Writing – original draft, Writing – review & editing, Formal analysis, Investigation, Data curation; Yang Wang: Writing – review & editing, Conceptualization, Methodology, Validation; Hanyang Ruan: Writing – review & editing, Conceptualization, Methodology, Validation; Gui Zhang: Conceptualization; Jian Gao: Investigation; Zifeng Zheng: Investigation; Di Li: Investigation; Qing Zhao: Investigation; Huiqin Han: Resources; Qing Fan: Resources; Dongdong Shi: Resources; Zhen Wang: Supervision, Resources, Funding acquisition, Conceptualization.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bai, T., Wang, Y., Ruan, H. et al. Sensorimotor circuit connectivity as a candidate biomarker for responsiveness to sertraline in obsessive–compulsive disorder. Neuropsychopharmacol. (2026). https://doi.org/10.1038/s41386-026-02375-5
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41386-026-02375-5