Abstract
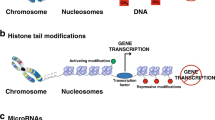
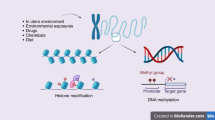
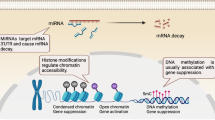
The 2020 Annual Review Issue, “Preventing Disease in the 21st Century” was selected by the Editors-in-Chief of Pediatric Research to include a variety of disease entities that confront health-care practitioners entrusted to the care of infants and children. In keeping with this mandate, this article reviews the subject of epigenetics, which impacts pediatric research from bench to bedside. Epigenetic mechanisms exert their effects through the interaction of environment, various susceptibility genes, and immunologic development and include: (1) DNA methylation; (2) posttranslational modifications of histone proteins through acetylation and methylation, and (3) RNA-mediated gene silencing by microRNA (miRNA) regulation. The effects of epigenetics during fetal life and early periods of development are first reviewed together with clinical applications of cardiovascular and metabolic disorders in later life. The relationships of epigenetics to the allergic and autoimmune diseases and cancer are next reviewed. A specific focus of the article is directed to the recent recognition that many of these disorders are driven by aberrant immune responses in which immunoregulatory events are often poorly functioning and where through interventive epigenetic measures prevention may be possible by alterations in programming of DNA during fetal and early periods as well as in later life.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Devaskar, S. U. & Raychaudhuri, S. Epigenetics-a science of heritable biological adaptation. Pediatr. Res. 61(Pt 2), 1R–4R (2007).
Slack, J. M. Conrad Hal Waddington: the last Renaissance biologist? Nat. Rev. Genet. 3, 889–895 (2002).
Bellanti, J. A. (ed.) Immunology IV: Clinical Applications in Health and Disease (I Care Press, Bethesda, MD, 2012).
Waddington, C. The Study of the Genes (Allen and Unwin, London, 1957).
Rodenhiser, D. & Mann, M. Epigenetics and human disease: translating basic biology into clinical applications. CMAJ 174, 341–348 (2006).
Bellanti, J. A. Genetics/epigenetics/allergy: the gun is loaded … but what pulls the trigger? Allergy Asthma Proc. 40, 76–83 (2019).
Kwon, C. S. & Wagner, D. Unwinding chromatin for development and growth: a few genes at a time. Trends Genet. 23, 403–412 (2007).
Ambros, V. The functions of animal microRNAs. Nature 431, 350–355 (2004).
Cutfield, W. S. et al. Could epigenetics play a role in the developmental origins of health and disease? Pediatr. Res. 61(Pt 2), 68R–75R (2007).
Yeh, T. F. et al. Outcomes at school age after postnatal dexamethasone therapy for lung disease of prematurity. N. Engl. J. Med. 350, 1304–1313 (2004).
Niemann, H. & Wrenzycki, C. Alterations of expression of developmentally important genes in preimplantation bovine embryos by in vitro culture conditions: implications for subsequent development. Theriogenology 53, 21–34 (2000).
Eppig, J. J. & O’Brien, M. J. Comparison of preimplantation developmental competence after mouse oocyte growth and development in vitro and in vivo. Theriogenology 49, 415–422 (1998).
Doherty, A. S. et al. Differential effects of culture on imprinted H19 expression in the preimplantation mouse embryo. Biol. Reprod. 62, 1526–1535 (2000).
Fahrner, J. A. & Bjornsson, H. T. Mendelian disorders of the epigenetic machinery: tipping the balance of chromatin states. Annu. Rev. Genomics Hum. Genet. 15, 269–293 (2014).
Bjornsson, H. T. The Mendelian disorders of the epigenetic machinery. Genome Res. 25, 1473–1481 (2015).
Sadikovic, B., Aref-Eshghi, E., Levy, M. A. & Rodenhiser, D. DNA methylation signatures in mendelian developmental disorders as a diagnostic bridge between genotype and phenotype. Epigenomics 11, 563–575 (2019).
Strong, E. et al. Symmetrical dose-dependent DNA-methylation profiles in children with deletion or duplication of 7q11.23. Am. J. Hum. Genet. 97, 216–227 (2015).
Aref-Eshghi, E. et al. BAFopathies’ DNA methylation epi-signatures demonstrate diagnostic utility and functional continuum of Coffin-Siris and Nicolaides-Baraitser syndromes. Nat. Commun. 9, 4885 (2018).
Schenkel, L. C. et al. Identification of epigenetic signature associated with alpha thalassemia/mental retardation X-linked syndrome. Epigenet. Chromatin 10, 10 (2017).
Aref-Eshghi, E. et al. Genomic DNA methylation signatures enable concurrent diagnosis and clinical genetic variant classification in neurodevelopmental syndromes. Am. J. Hum. Genet. 102, 156–174 (2018). **Describes the identification and use of DNA methylation episignatures in 14 developmental disorders.
Butcher, D. T. et al. CHARGE and Kabuki syndromes: gene-specific DNA methylation signatures identify epigenetic mechanisms linking these clinically overlapping conditions. Am. J. Hum. Genet. 100, 773–788 (2017).
Kernohan, K. D. et al. Identification of a methylation profile for DNMT1-associated autosomal dominant cerebellar ataxia, deafness, and narcolepsy. Clin. Epigenet. 8, 91 (2016).
Schenkel, L. C. et al. Clinical validation of Fragile X syndrome screening by DNA methylation array. J. Mol. Diagn. 18, 834–841 (2016).
Aref-Eshghi, E. et al. Clinical validation of a genome-wide dna methylation assay for molecular diagnosis of imprinting disorders. J. Mol. Diagn. 19, 848–856 (2017).
Prickett, A. R. et al. Genome-wide methylation analysis in Silver-Russell syndrome patients. Hum. Genet. 134, 317–332 (2015).
Wu, D., Gong, C. & Su, C. Genome-wide analysis of differential DNA methylation in Silver-Russell syndrome. Sci. China Life Sci. 60, 692–699 (2017).
Schenkel, L. C. et al. Peripheral blood epi-signature of Claes-Jensen syndrome enables sensitive and specific identification of patients and healthy carriers with pathogenic mutations in KDM5C. Clin. Epigenet. 10, 21 (2018).
Aref-Eshghi, E. et al. The defining DNA methylation signature of Kabuki syndrome enables functional assessment of genetic variants of unknown clinical significance. Epigenetics 12, 923–933 (2017).
Li, Y. et al. An epigenetic signature in peripheral blood associated with the haplotype on 17q21.31, a risk factor for neurodegenerative tauopathy. PLoS Genet. 10, e1004211 (2014).
Choufani, S. et al. NSD1 mutations generate a genome-wide DNA methylation signature. Nat. Commun. 6, 10207 (2015).
Hood, R. L. et al. The defining DNA methylation signature of Floating-Harbor syndrome. Sci. Rep. 6, 38803 (2016).
Bacalini, M. G. et al. Identification of a DNA methylation signature in blood cells from persons with Down Syndrome. Aging 7, 82–96 (2015).
Guastafierro, T. et al. Genome-wide DNA methylation analysis in blood cells from patients with Werner syndrome. Clin. Epigenet. 92, 28–31 (2017).
Campisano, S., La Colla, A., Echarte, S. M. & Chisari, A. N. Interplay between early-life malnutrition, epigenetic modulation of the immune function and liver diseases. Nutr. Res. Rev. 32, 128–145 (2019).
Lesseur, C. & Chen, J. Adverse maternal metabolic intrauterine environment and placental epigenetics: implications for fetal metabolic programming. Curr. Environ. Health Rep. 5, 531–543 (2018).
Tiffon, C. The impact of nutrition and environmental epigenetics on human health and disease. Int. J. Mol. Sci. 19, E3425 (2018).
Goyal, D., Limesand, S. W. & Goyal, R. Epigenetic responses and the developmental origins of health and disease. J. Endocrinol. 242, T105–T119 (2019).
Picascia, A. et al. Epigenetic control of autoimmune diseases: from bench to bedside. Clin. Immunol. 157, 1–15 (2015).
Sabounchi, S., Bollyky, J. & Nadeau, K. Review of Environmental impact on the epigenetic regulation of atopic diseases. Curr. Allergy Asthma Rep. 15, 1–33 (2015).
Teruel, M. & Alarcón-Riquelme, M. E. The genetic basis of systemic lupus erythematosus: what are the risk factors and what have we learned. J. Autoimmun. 74, 161–175 (2016).
Richardson, B. The interaction between environmental triggers and epigenetics in autoimmunity. Clin. Immunol. 192, 1–5 (2018).
Chen, W., Liu, D., Li, Q. Z. & Zhu, H. The function of ncRNAs in rheumatic diseases. Epigenomics 11, 821–833 (2019)
Selmi, C. Autoimmunity in 2018. Clin. Rev. Allergy Immunol. 56, 375–384 (2019).
Selmi, C. & Gershwin, M. E. Sex and autoimmunity: proposed mechanisms of disease onset and severity. Expert Rev. Clin. Immunol. 15, 607–615 (2019).
Charras, A. & Hedrich, C. M. The role of epigenetics in paediatric rheumatic disease. Curr. Opin. Rheumatol. 31, 450–463 (2019).
Potaczek, D. P. et al. Epigenetics and allergy: from basic mechanisms to clinical applications. Epigenomics 9, 539–571 (2017).
North, M. L. & Ellis, A. K. The role of epigenetics in the developmental origins of allergic disease. Ann. Allergy Asthma Immunol. 106, 355–361 (2011).
Tost, J. A translational perspective on epigenetics in allergic diseases. J. Allergy Clin. Immunol. 142, 715–726 (2018).
Peng, C. et al. Epigenome-wide association study of total serum immunoglobulin E in children: a life course. Clin. Epigenet. 10, 55 (2018).
DeVries, A. & Vercelli, D. The neonatal methylome as a gatekeeper in the trajectory to childhood asthma. Epigenomics 9, 585–593 (2017).
DeVries, A. & Vercelli, D. Early predictors of asthma and allergy in children: the role of epigenetics. Curr. Opin. Allergy Clin. Immunol. 15, 435–439 (2015).
DeVries, A. & Vercelli, D. Epigenetics in allergic diseases. Curr. Opin. Pediatr. 27, 719–723 (2015).
Bunning, B. J., DeKruyff, R. H. & Nadeau, K. C. Epigenetic changes during food-specific immunotherapy. Curr. Allergy Asthma Rep. 16, 87 (2016).
Reese, S. E. et al. Epigenome-wide meta-analysis of DNA methylation and childhood asthma. J. Allergy Clin. Immunol. 143, 2062–2074 (2019).
Hughes, R. L. et al. The role of the gut microbiome in predicting response to diet and the development of precision nutrition models-Part I. Overview of current methods. Adv. Nutr. https://doi.org/10.1093/advances/nmz022 (2019).
Walker, M. M., Talley, N. J. & Keely, S. Follow up on atopy and the gastrointestinal tract - a review of a common association 2018. Expert Rev. Gastroenterol. Hepatol. 13, 437–445 (2019).
Cait, A. et al. Reduced genetic potential for butyrate fermentation in the gut microbiome of infants who develop allergic sensitization. J. Allergy Clin. Immunol. https://doi.org/10.1016/j.jaci.2019.06.029 (2019).
Thio, C. L. et al. Regulation of type 2 innate lymphoid cell-dependent airway hyperreactivity by butyrate. J. Allergy Clin. Immunol. 142, 1867–1883 (2018).
Wang, J., Wen, L., Wang, Y. & Chen, F. Therapeutic effect of histone deacetylase inhibitor, sodium butyrate, on allergic rhinitis in vivo. DNA Cell Biol. 35, 203–208 (2016).
Zhu, Z. et al. Short-chain fatty acids as a target for prevention against food allergy by regulatory T cells. JGH Open 3, 190–195 (2019).
Ehrlich, M. DNA hypermethylation in disease: mechanisms and clinical relevance. Epigenetics 8, 1–23 (2019).
Paska, A. V. & Hudler, P. Aberrant methylation patterns in cancer: a clinical view. Biochem. Med. (Zagreb) 25, 161–176 (2015).
Robertson, K. D. DNA methylation and human disease. Nat. Rev. Genet. 6, 597–610 (2005).
Martino, D. et al. Epigenetics in immune development and in allergic and autoimmune diseases. J. Reprod. Immunol. 104-105, 43–48 (2014).
Chavez-Valencia, R. A. et al. The DNA methylation landscape of CD4(+) T cells in oligoarticular juvenile idiopathic arthritis. J. Autoimmun. 86, 29–38 (2018).
Phillips, J. E., Couper, J. J., Penno, M. A. S., Harrison, L. C. & ENDIA Study Group. Type 1 diabetes: a disease of developmental origins. Pediatr. Diabetes 18, 417–421 (2017).
Langie, S. A. et al. DNA methylation and the hygiene hypothesis: connecting respiratory allergy and childhood acute lymphoblastic leukemia. Epigenomics 11, 1519–1537 (2019).
Gentner, M. B. & Leppert, M. L. O. Environmental influences on health and development: nutrition, substance exposure, and adverse childhood experiences. Dev. Med. Child Neurol. 61, 1008–1014 (2019).
McCann, T. S. et al. Biology and targeting of the Jumonji-domain histone demethylase family in childhood neoplasia: a preclinical overview. Expert Opin. Ther. Targets 23, 267–280 (2019).
Mika, A. et al. Epigenetic changes at the Birc5 promoter induced by YM155 in synovial sarcoma. J. Clin. Med. 8, E408 (2019).
Flotho, C. Gene mutations do not operate in a vacuum: the increasing importance of epigenetics in juvenile myelomonocytic leukemia. Epigenetics 14, 236–244 (2019).
Lawless, O. J. et al. In vitro induction of T regulatory cells by a methylated CpG DNA sequence in humans: potential therapeutic applications in allergic and autoimmune diseases. Allergy Asthma Proc. 39, 143–152 (2018).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The author declares no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bellanti, J.A. Epigenetic studies and pediatric research. Pediatr Res 87, 378–384 (2020). https://doi.org/10.1038/s41390-019-0644-9
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41390-019-0644-9
This article is cited by
-
A comprehensive analysis of minimally differentially methylated regions common to pediatric and adult solid tumors
npj Precision Oncology (2024)
-
Epigenetics of pregnancy: looking beyond the DNA code
Journal of Assisted Reproduction and Genetics (2022)
-
Precision medicine in the era of artificial intelligence: implications in chronic disease management
Journal of Translational Medicine (2020)