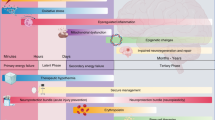
Abstract
The next phase of clinical trials in neonatal encephalopathy (NE) focuses on hypothermia adjuvant therapies targeting alternative recovery mechanisms during the process of hypoxic brain injury. Identifying infants eligible for neuroprotective therapies begins with the clinical detection of brain injury and classification of severity. Combining a variety of biomarkers (serum, clinical exam, EEG, movement patterns) with innovative clinical trial design and analyses will help target infants with the most appropriate and timely treatments. The timing of magnetic resonance imaging (MRI) and MR spectroscopy after NE both assists in identifying the acute perinatal nature of the injury (days 3–7) and evaluates the full extent and evolution of the injury (days 10–21). Early, intermediate outcome of neuroprotective interventions may be best defined by the 21-day neuroimaging, with recognition that the full neurodevelopmental trajectory is not yet defined. An initial evaluation of each new therapy at this time point may allow higher-throughput selection of promising therapies for more extensive investigation. Functional recovery can be assessed using a trajectory of neurodevelopmental evaluations targeted to a prespecified and mechanistically derived hypothesis of drug action. As precision medicine revolutionizes healthcare, it should also include the redesign of NE clinical trials to allow safe, efficient, and targeted therapeutics.
Impact
-
As precision medicine revolutionizes healthcare, it should also include the redesign of NE clinical trials to allow faster development of safe, effective, and targeted therapeutics.
-
This article provides a multidisciplinary perspective on the future of clinical trials in NE; novel trial design; study management and oversight; biostatistical methods; and a combination of serum, imaging, and neurodevelopmental biomarkers can advance the field and improve outcomes for infants affected by NE.
-
Innovative clinical trial designs, new intermediate trial end points, and a trajectory of neurodevelopmental evaluations targeted to a prespecified and mechanistically derived hypothesis of drug action can help address common challenges in NE clinical trials and allow for faster selection and validation of promising therapies for more extensive investigation.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Executive Summary: Neonatal Encephalopathy and Neurologic Outcome, Second Edition. Report of the American College of Obstetricians and Gynecologists’ Task Force on Neonatal Encephalopathy. Obstet. Gynecol. 123, 896–901 (2014).
Ferriero, D. M. Neonatal brain injury. N. Engl. J. Med. 351, 1985–1995 (2004).
Gunn, A. J., Bennet, L. & Gunn, A. J. Fetal hypoxia insults and patterns of brain injury: insights from animal models. Clin. Perinatol. 36, 579–593 (2009).
Tagin, M. A., Woolcott, C. G., Vincer, M. J., Whyte, R. K. & Stinson, D. A. Hypothermia for neonatal hypoxic ischemic encephalopathy: an updated systematic review and meta-analysis. Arch. Pediatr. Adolesc. Med. 166, 558–566 (2012).
Shankaran, S. et al. Whole-body hypothermia for neonates with hypoxic–ischemic encephalopathy. N. Engl. J. Med. 353, 1574–1584 (2005).
Shankaran, S. et al. Childhood outcomes after hypothermia for neonatal encephalopathy. N. Engl. J. Med. 366, 2085–2092 (2012).
Jacobs, S. E. et al. Cooling for newborns with hypoxic ischaemic encephalopathy. Cochrane Database Syst. Rev. CD003311 (2013).
Lally, P. J. et al. Magnetic resonance spectroscopy assessment of brain injury after moderate hypothermia in neonatal encephalopathy: a prospective multicentre cohort study. Lancet Neurol. 18, 35–45 (2019).
Shankaran, S. et al. Effect of depth and duration of cooling on death or disability at age 18 months among neonates with hypoxic-ischemic encephalopathy a randomized clinical trial. JAMA 318, 57–67 (2017).
Gunn, A. J. & Battin, M. Towards faster studies of neonatal encephalopathy. Lancet Neurol. 18, 21–22 (2019).
Chalak, L., Ferriero, D. M., Gressens, P., Molloy, E. & Bearer, C. A 20 years conundrum of neonatal encephalopathy and hypoxic ischemic encephalopathy: are we closer to a consensus guideline? Pediatr. Res. 86, 548–549 (2019).
Molloy, E. J. & Bearer, C. Neonatal encephalopathy versus hypoxic-ischemic encephalopathy. Pediatr. Res. 84, 574 (2018).
Jones, J. & Hunter, D. Consensus methods for medical and health services research. BMJ 311, 376–380 (1995).
Davidson, J. O., Wassink, G., van den Heuij, L. G., Bennet, L. & Gunn, A. J. Therapeutic hypothermia for neonatal hypoxic-ischemic encephalopathy - where to from here? Front. Neurol. 6, 198 (2015).
Massaro, A. N. et al. Plasma biomarkers of brain injury in neonatal hypoxic-ischemic encephalopathy. J. Pediatr. 194, 67.e1–75.e1 (2018).
Wu, Y. W. et al. High-dose erythropoietin and hypothermia for hypoxic-ischemic encephalopathy: a phase II trial. Pediatrics 137, e20160191 (2016).
Juul, S. E. et al. High-dose erythropoietin for asphyxia and encephalopathy (HEAL): a randomized controlled trial-background, aims, and study protocol. Neonatology 113, 331–338 (2018).
Cotten, C. M. et al. Feasibility of autologous cord blood cells for infants with hypoxic-ischemic encephalopathy. J. Pediatr. 164, 973.e1–979.e1 (2014).
Cotten, C. M. A multi-site study of autologous cord blood cells for hypoxic ischemic encephalopathy. https://clinicaltrials.gov/ct2/show/NCT02612155?cond=Hypoxic-Ischemic+Encephalopathy&rank=38 (2008). Cited 13 Feb 2019.
Robertson, N. J. et al. Melatonin as an adjunct to therapeutic hypothermia in a piglet model of neonatal encephalopathy: a translational study. Neurobiol. Dis. 121, 240–251 (2019).
Robertson, N. J. et al. Melatonin augments hypothermic neuroprotection in a perinatal asphyxia model. Brain 136, 90–105 (2013).
Weiss, M. D. Melatonin as a neuroprotective therapy in neonates with HIE undergoing hypothermia. https://clinicaltrials.gov/ct2/show/NCT02621944 (2019). Cited 13 Feb 2019.
Young, L., Berg, M. & Soll, R. Prophylactic barbiturate use for the prevention of morbidity and mortality following perinatal asphyxia. Cochrane Database Syst. Rev. CD001240 (2016).
Filippi, L. et al. Safety and efficacy of topiramate in neonates with hypoxic ischemic encephalopathy treated with hypothermia (NeoNATI): a feasibility study. J. Matern Neonatal Med. 31, 973–980 (2018).
Hoffman, K. R. Topiramate in neonates receiving whole body cooling for hypoxic ischemic encephalopathy. https://clinicaltrials.gov/ct2/show/NCT01765218?cond=Hypoxic-Ischemic+Encephalopathy&rank=5 (2018). Cited 14 Feb 2019.
Inder, T. E. & Volpe, J. J. in Volpe’s Neurology of the Newborn 510.e15–563.e15 (Elsevier Inc., 2018).
Laptook, A. R. et al. Effect of therapeutic hypothermia initiated after 6 hours of age on death or disability among newborns with hypoxic-ischemic encephalopathy a randomized clinical trial. JAMA 318, 1550–1560 (2017).
Natarajan, G., Laptook, A. & Shankaran, S. Therapeutic hypothermia: how can we optimize this therapy to further improve outcomes? Clin. Perinatol. 45, 241–255 (2018).
Van Laerhoven, H., De Haan, T. R., Offringa, M., Post, B. & Van Der Lee, J. H. Prognostic tests in term neonates with hypoxic-ischemic encephalopathy: a systematic review. Pediatrics 131, 88–98 (2013).
Merchant, N. & Azzopardi, D. Early predictors of outcome in infants treated with hypothermia for hypoxic-ischaemic encephalopathy. Dev. Med. Child Neurol. 57, 8–16 (2015).
Shankaran, S. et al. Evolution of encephalopathy during whole body hypothermia for neonatal hypoxic-ischemic encephalopathy. J. Pediatr. 160, 567–572.e3 (2012).
Graham, E. M., Burd, I., Everett, A. D. & Northington, F. J. Blood biomarkers for evaluation of perinatal encephalopathy. Front. Pharm. 7, 1–12 (2016).
Jenkins, D. D. et al. Serum cytokines in a clinical trial of hypothermia for neonatal hypoxic-ischemic encephalopathy. J. Cereb. Blood Flow. Metab. 32, 1888–1896 (2012).
Orrock, J. E. et al. Association of brain injury and neonatal cytokine response during therapeutic hypothermia in newborns with hypoxic-ischemic encephalopathy. Pediatr. Res. 79, 742–747 (2016).
Chalak, L. F. et al. Biomarkers for severity of neonatal hypoxic-ischemic encephalopathy and outcomes in newborns receiving hypothermia therapy. J. Pediatr. 164, 468.e1–474.e1 (2014).
Mizrahi, E. M. & Hrachovy, R. A. Atlas of Neonatal Electroencephalography, 4th edn. (Demos Medical Publishing, 2016).
Massey, S. L. et al. Interrater and intrarater agreement in neonatal electroencephalogram background scoring. J. Clin. Neurophysiol. 36, 1–8 (2019).
Weeke, L. C. et al. Role of EEG background activity, seizure burden and MRI in predicting neurodevelopmental outcome in full-term infants with hypoxic-ischaemic encephalopathy in the era of therapeutic hypothermia. Eur. J. Paediatr. Neurol. 20, 855–864 (2016).
Murray, D. M., O’Connor, C. M., Anthony Ryan, C., Korotchikova, I. & Boylan, G. B. Early EEG grade and outcome at 5 years after mild neonatal hypoxic ischemic encephalopathy. Pediatrics 138, e20160659 (2016).
Nash, K. B. et al. Video-EEG monitoring in newborns with hypoxic-ischemic encephalopathy treated with hypothermia. Neurology 76, 556–562 (2011).
Chandrasekaran, M., Chaban, B., Montaldo, P. & Thayyil, S. Predictive value of amplitude-integrated EEG (aEEG) after rescue hypothermic neuroprotection for hypoxic ischemic encephalopathy: a meta-analysis. J. Perinatol. 37, 684–689 (2017).
Ter Horst, H. J. et al. Prognostic significance of amplitude-integrated EEG during the first 72 hours after birth in severely asphyxiated neonates. Pediatr. Res. 55, 1026–1033 (2004).
Thoresen, M., Hellström-Westas, L., Liu, X. & De Vries, L. S. Effect of hypothermia on amplitude-integrated electroencephalogram in infants with asphyxia. Pediatrics 126, 131–141 (2010).
Shalak, L. F. L. A. Amplitude-integrated electroencephalography coupled with an early persistent encephalopathy. Pediatrics 111, 351–357 (2003).
Skranes, J. H. et al. Amplitude-integrated electroencephalography improves the identification of infants with encephalopathy for therapeutic hypothermia and predicts neurodevelopmental outcomes at 2 years of age. J. Pediatr. 187, 34–42 (2017).
Carrasco, M. et al. Cerebral autoregulation and conventional and diffusion tensor imaging magnetic resonance imaging in neonatal hypoxic-ischemic encephalopathy. Pediatr. Neurol. 82, 36–43 (2018).
Massaro, A. N. et al. Impaired cerebral autoregulation and brain injury in newborns with hypoxic-ischemic encephalopathy treated with hypothermia. J. Neurophysiol. 114, 818–824 (2015).
Chalak, L. F., Tarumi, T. & Zhang, R. The “neurovascular unit approach” to evaluate mechanisms of dysfunctional autoregulation in asphyxiated newborns in the era of hypothermia therapy. Early Hum. Dev. 90, 687–694 (2014).
Thewissen, L. et al. Measuring near-infrared spectroscopy derived cerebral autoregulation in neonates: from research tool toward bedside multimodal monitoring. Front. Pediatr. 6, 1–21 (2018).
Lee, J. K. et al. Optimizing cerebral autoregulation may decrease neonatal regional hypoxic-ischemic brain injury. Dev. Neurosci. 39, 248–256 (2017).
Tian, F., Tarumi, T., Liu, H., Zhang, R. & Chalak, L. Wavelet coherence analysis of dynamic cerebral autoregulation in neonatal hypoxic-ischemic encephalopathy. NeuroImage Clin. 11, 124–132 (2016).
Tekes, A. et al. Apparent diffusion coefficient scalars correlate with near-infrared spectroscopy markers of cerebrovascular autoregulation in neonates cooled for perinatal hypoxic-ischemic injury. Am. J. Neuroradiol. 36, 188–193 (2015).
Howlett, J. A. et al. Cerebrovascular autoregulation and neurologic injury in neonatal hypoxic-ischemic encephalopathy. Pediatr. Res. 74, 525–535 (2013).
Burton, V. J. et al. A pilot cohort study of cerebral autoregulation and 2-year neurodevelopmental outcomes in neonates with hypoxic-ischemic encephalopathy who received therapeutic hypothermia. BMC Neurol. 15, 1–13 (2015).
Chalak, L. F. et al. Novel wavelet real time analysis of neurovascular coupling in neonatal encephalopathy. Sci. Rep. 7, 45958 (2017).
Chalak, L. F. & Zhang, R. New wavelet neurovascular bundle for bedside evaluation of cerebral autoregulation and neurovascular coupling in newborns with hypoxic-ischemic encephalopathy. Dev. Neurosci. 39, 89–96 (2017).
Groenendaal, F. & de Vries, L. S. Fifty years of brain imaging in neonatal encephalopathy following perinatal asphyxia. Pediatr. Res. 81, 150–155 (2017).
Weeke, L. C. et al. A novel magnetic resonance imaging score predicts neurodevelopmental outcome after perinatal asphyxia and therapeutic hypothermia. J. Pediatr. 192, 33.e2–40.e2 (2018).
McKinstry, R. et al. A prospective, longitudinal diffusion tensor imaging study of brain injury in newborns. Neurology 59, 824 (2002).
Barkovich, A. J. et al. MR imaging, MR spectroscopy, and diffusion tensor imaging of sequential studies in neonates with encephalopathy. AJNR 27, 533–547 (2006).
Ou, Y. et al. Using clinically acquired MRI to construct age-specific ADC atlases: Quantifying spatiotemporal ADC changes from birth to 6-year old. Hum. Brain Mapp. 38, 3052–3068 (2017).
Robertson, N. J., Thayyil, S., B. Cady, E. & Raivich, G. Magnetic resonance spectroscopy biomarkers in term perinatal asphyxial encephalopathy: from neuropathological correlates to future clinical applications. Curr. Pediatr. Rev. 10, 37–47 (2014).
Thayyil, S. et al. Cerebral magnetic resonance biomarkers in neonatal encephalopathy: a meta-analysis. Pediatrics 125, e382–e395 (2010).
Thayyil, S. et al. Hypothermia for encephalopathy in low and middle-income countries (HELIX): study protocol for a randomised controlled trial. Trials 18, 1–14 (2017).
Azzopardi, D. et al. Moderate hypothermia within 6 h of birth plus inhaled xenon versus moderate hypothermia alone after birth asphyxia (TOBY-Xe): a proof-of-concept, open-label, randomised controlled trial. Lancet Neurol. 15, 145–153 (2016).
NICHD. NICHD Neonatal Research Network. https://neonatal.rti.org/ (1986). Cited 9 Apr 2019.
Schulz, K. F., Altman, D. G., Moher, D. & CONSORT Group. CONSORT 2010 statement: updated guidelines for reporting parallel group randomised trials. BMJ 340, c332 (2010).
Lean Six Sigma Institute, LLC. Lean Six Sigma Institute. www.leansixsigmainstitute.org/ (2019). Cited 9 Apr 2019.
Lilford, R. J., Thornton, J. G. & Braunholtz, D. Clinical trials and rare diseases: a way out of a conundrum. BMJ 311, 1621 (1995).
Quintana, M., Viele, K. & Lewis, R. Bayesian analysis: using prior information to interpret the results of clinical trials. JAMA 318, 1605–1606 (2017).
FDA. FDA Guidance. Adaptive designs for clinical trials of drugs and biologics guidance for industry. https://www.fda.gov/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/default.htm (2018).
Chow, S.-C. Adaptive clinical trial design. Annu Rev. Med. 65, 405–415 (2014).
Maitre, N. L. Neurorehabilitation after neonatal intensive care: evidence and challenges. Arch. Dis. Child. Fetal Neonatal Ed. 100, F534–F540 (2015).
Anderson, P. J., Luca, C. R. De, Hutchinson, E., Roberts, G. & Doyle, L. W. Underestimation of developmental delay by the new Bayley-III Scale. Arch. Pediatr. Adolesc. Med. 164, 352 (2010).
Chalak, L. F. et al. Neurodevelopmental outcomes after hypothermia therapy in the era of Bayley-III. J. Perinatol. 34, 629–633 (2014).
Hack, M. et al. Poor predictive validity of the Bayley Scales of infant development for cognitive function of extremely low birth weight children at school age. Pediatrics 116, 333–341 (2005).
Romeo, D. M. et al. Neonatal neurological examination during the first 6 h after birth. Early Hum. Dev. 108, 41–44 (2017).
Dubowitz, L., Mercuri, E. & Dubowitz, V. An optimality score for the neurologic examination of the term newborn. J. Pediatr. 133, 406–416 (1999).
George, J. M. et al. Relationship between very early brain structure and neuromotor, neurological and neurobehavioral function in infants born <31 weeks gestational age. Early Hum. Dev. 117, 74–82 (2018).
Haataja, L. et al. Neurologic examination in infants with hypoxicischemic encephalopathy at age 9 to 14 months: use of optimality scores and correlation with magnetic resonance imaging finding. J. Pediatr. 138, 332–337 (2001).
Cainelli, E., Trevisanuto, D., Cavallin, F., Manara, R. & Suppiej, A. Evoked potentials predict psychomotor development in neonates with normal MRI after hypothermia for hypoxic-ischemic encephalopathy. Clin. Neurophysiol. 129, 1300–1306 (2018).
Key, A. P. F., Lambert, E. W., Aschner, J. L. & Maitre, N. L. Influence of gestational age and postnatal age on speech sound processing in NICU infants. Psychophysiology 49, 720–731 (2012).
Papadelis, C. et al. Reorganization of the somatosensory cortex in hemiplegic cerebral palsy associated with impaired sensory tracts. Neuroimage Clin. 17, 198–212 (2018)
Murray, M. M., Brunet, D. & Michel, C. M. Topographic ERP analyses: a step-by-step tutorial review. Brain Topogr. 20, 249–264 (2008).
Morgan, C., Honan, I., Allsop, A., Novak, I. & Badawi, N. Psychometric properties of assessments of cognition in infants with cerebral palsy or motor impairment: a systematic review. J. Pediatr. Psychol. 44, 238–252 (2019).
Griffiths, A., Toovey, R., Morgan, P. E. & Spittle, A. J. Psychometric properties of gross motor assessment tools for children: a systematic review. BMJ Open 8, e021734 (2018).
Kjølbye, C. B., Bo Drivsholm, T., Ertmann, R. K., Lykke, K. & Køster-Rasmussen, R. Motor function tests for 0-2-year-old children – a systematic review. Dan. Med. J. 65, 1–8 (2018).
Eeles, A. L. et al. Assessments of sensory processing in infants: a systematic review. Dev. Med. Child Neurol. 55, 314–326 (2013).
Nelson, H. D., Nygren, P., Walker, M. & Panoscha, R. Screening for speech and language delay in preschool children: systematic evidence review for the US Preventive Services Task Force. Pediatrics 117, e298–e319 (2006).
Hanratty, J. et al. Systematic review of the measurement properties of tools used to measure behaviour problems in young children with autism. PLoS ONE 10, 1–21 (2015).
Halle, T. G. & Darling-Churchill, K. E. Review of measures of social and emotional development. J. Appl. Dev. Psychol. 45, 8–18 (2016).
McCrae, J. S. & Brown, S. M. Systematic review of social–emotional screening instruments for young children in child welfare. Res. Soc. Work Pract. 28, 767–788 (2018).
Brydges, C. R. et al. Cognitive outcomes in children and adolescents born very preterm: a meta-analysis. Dev. Med. Child Neurol. 60, 452–468 (2018).
Wong, H. S., Santhakumaran, S., Cowan, F. M. & Modi, N. Developmental assessments in preterm children: a meta-analysis. Pediatrics 138, e20160251 (2016).
Hack, M. Poor predictive validity of the Bayley Scales of infant development for cognitive function of extremely low birth weight children at school age. Pediatrics 116, 333–341 (2005).
Maitre, N. L., Slaughter, J. C. & Aschner, J. L. Early prediction of cerebral palsy after neonatal intensive care using motor development trajectories in infancy. Early Hum. Dev. 89, 781–786 (2013).
Einspieler, C., Prechtl, H. F., Bos, A., Ferrari, F. & Cioni, G. Prechtl’s Method on the Qualitative Assessment of General Movements in Preterm, Term and Young Infants, 1st edn. (Mac Keith Press, 2008).
Kwong, A. K. L., Fitzgerald, T. L., Doyle, L. W., Cheong, J. L. Y. & Spittle, A. J. Predictive validity of spontaneous early infant movement for later cerebral palsy: a systematic review. Dev. Med. Child Neurol. 60, 480–489 (2018).
Romeo, D. M., Ricci, D., Brogna, C. & Mercuri, E. Use of the Hammersmith Infant Neurological Examination in infants with cerebral palsy: a critical review of the literature. Dev. Med. Child Neurol. 58, 240–245 (2016).
Palisano, R. J., Avery, L., Gorter, J. W., Galuppi, B. & Mccoy, S. W. Stability of the gross motor function classification system, manual ability classification system, and communication function classification system. Dev. Med. Child Neurol. 60, 1026–1032 (2018).
Gorter, J. W., Ketelaar, M., Rosenbaum, P., Helders, P. J. M. & Palisano, R. Use of the GMFCS in infants with CP: the need for reclassification at age 2 years or older. Dev. Med. Child Neurol. 51, 46–52 (2009).
Abbasi, H. & Unsworth, C. P. Applications of advanced signal processing and machine learning in the neonatal hypoxic-ischemic electroencephalography. Neural Regen. Res. 15, 222–231 (2020).
Jackson, T. C., Kotermanski, S. E. & Kochanek, P. M. Infants uniquely express high levels of RBM3 and other cold-adaptive neuroprotectant proteins in the human brain. Dev. Neurosci. 40, 325–336 (2018).
Jackson, T. C. & Kochanek, P. M. A new vision for therapeutic hypothermia in the era of targeted temperature management: a speculative synthesis. Ther. Hypothermia Temp. Manag. 9, 13–47 (2019).
Jenkins, D. D. et al. Altered circulating leukocytes and their chemokines in a clinical trial of therapeutic hypothermia for neonatal hypoxic ischemic encephalopathy. Pediatr. Crit. Care Med. 14, 786–795 (2013).
Vik, S. D., Torp, H., Follestad, T., Støen, R. & Nyrnes, S. A. NeoDoppler: new ultrasound technology for continous cerebral circulation monitoring in neonates. Pediatr. Res. 87, 95–103 (2019).
Smyser, C. D., Wheelock, M. D., Limbrick, D. D. & Neil, J. J. Neonatal brain injury and aberrant connectivity. Neuroimage 185, 609–623 (2019).
De Vis, J. B. et al. Arterial spin-labelling perfusion MRI and outcome in neonates with hypoxic-ischemic encephalopathy. Eur. Radiol. 25, 113–121 (2015).
Mohammadi-Nejad, A.-R. et al. Neonatal brain resting-state functional connectivity imaging modalities. Photoacoustics 10, 1–19 (2018).
Li, H. X., Yu, M. & Zheng, A. Bin et al. Resting-state network complexity and magnitude changes in neonates with severe hypoxic ischemic encephalopathy. Neural Regen. Res. 14, 642–648 (2019).
The Nobel Prize in Economic Sciences Committee, Banerjee, A., Duflo, E. & Kremer, M. Understanding development and poverty alleviation. https://www.nobelprize.org/uploads/2019/10/advanced-economicsciencesprize2019.pdf (2019).
Denihan, N. M., Boylan, G. B. & Murray, D. M. Metabolomic profiling in perinatal asphyxia: a promising new field. Biomed. Res. Int. 2015, 254076 (2015).
AAP and ACOG Task Force on Neonatal Encephalopathy. Neonatal Encephalopathy and Neurologic Outcome, Second Edition. Pediatrics 133, e1482–e1488 (2014).
U.S. Department of Health and Human Services Food and Drug Administration. Oversight of clinical investigations—a risk-based approach to monitoring. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/oversight-clinical-investigations-risk-based-approach-monitoring (2013). Cited 7 May 2019.
Acknowledgements
Thank you to Kenji M. Cunnion and Neel K. Krishna for hosting the 2019 HIE Symposium: Developing the Future. The symposium was made possible through support from ReAlta Life Sciences, Children’s Specialty Group, Eastern Virginia Medical School, and the Children’s Hospital of The King’s Daughters. This work was supported by 1R01HD081120-01A1 from the Eunice Kennedy Shriver National Institute of Child Health and Human Development to N.L.M. The content is solely the responsibility of the authors and does not necessarily represent the official views of the funding organizations.
Author information
Authors and Affiliations
Contributions
K.L.B., T.E.I., A.M.G., and N.L.M. participated in the conception and design, drafted the initial manuscript, revised the manuscript critically for important intellectual content, and approved the final version for publication. C.M.C., D.R.N., T.A.S., and J.C.S. participated in the conception and design, revised the manuscript critically for important intellectual content, and approved the final version for publication.
Corresponding author
Ethics declarations
Competing interests
A.M.G. is a consultant for Radial Medical, Autonomic Technologies, and Element Science. All other authors have disclosed that they do not have any relevant relationships with commercial companies.
Informed consent
Patient consent was not required for this manuscript.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Benninger, K.L., Inder, T.E., Goodman, A.M. et al. Perspectives from the Society for Pediatric Research. Neonatal encephalopathy clinical trials: developing the future. Pediatr Res 89, 74–84 (2021). https://doi.org/10.1038/s41390-020-0859-9
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/s41390-020-0859-9
This article is cited by
-
Bayley trajectories predict school readiness better than single assessments in formerly very preterm preschoolers
Pediatric Research (2023)
-
Advanced Neuromonitoring Modalities on the Horizon: Detection and Management of Acute Brain Injury in Children
Neurocritical Care (2023)
-
Hypoxia–ischemia-mediated effects on neurodevelopmentally regulated cold-shock proteins in neonatal mice under strict temperature control
Pediatric Research (2022)
-
A neonatal neuroNICU collaborative approach to neuromonitoring of posthemorrhagic ventricular dilation in preterm infants
Pediatric Research (2022)
-
Biomarker und Neuromonitoring zur Entwicklungsprognose nach perinataler Hirnschädigung
Monatsschrift Kinderheilkunde (2022)