Abstract
Background
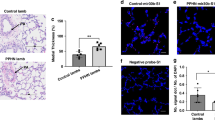
Persistent pulmonary hypertension of the newborn (PPHN) occurs when pulmonary vascular resistance (PVR) fails to decrease at birth. Decreased angiogenesis in the lung contributes to the persistence of high PVR at birth. MicroRNAs (miRNAs) regulate gene expression through transcript binding and degradation. They were implicated in dysregulated angiogenesis in cancer and cardiovascular disease.
Methods
We investigated whether altered miRNA levels contribute to impaired angiogenesis in PPHN. We used a fetal lamb model of PPHN induced by prenatal ductus arteriosus constriction and sham ligation as controls. We performed RNA sequencing of pulmonary artery endothelial cells (PAECs) isolated from control and PPHN lambs.
Results
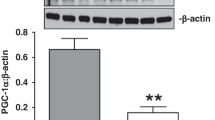
We observed a differentially expressed miRNA profile in PPHN for organ development, cell–cell signaling, and cardiovascular function. MiR-34c was upregulated in PPHN PAECs compared to controls. Exogenous miR34c mimics decreased angiogenesis by control PAEC and anti-miR34c improved angiogenesis of PPHN PAEC in vitro. Notch1, a predicted target for miR-34c by bioinformatics, was decreased in PPHN PAECs, along with Notch1 downstream targets, Hey1 and Hes1. Exogenous miR-34c decreased Notch1 expression in control PAECs and anti-miR-34c restored Notch1 and Hes1 expression in PPHN PAECs.
Conclusion
We conclude that increased miR-34c in PPHN contributes to impaired angiogenesis by decreasing Notch1 expression in PAECs.
Impact
-
Adds a novel mechanism for the regulation of angiogenesis in persistent pulmonary hypertension of the newborn.
-
Identifies non-coding RNAs that are involved in the altered angiogenesis in PPHN and thus the potential for future studies to identify links between known pathways regulating angiogenesis.
-
Provides preliminary data to conduct studies targeting miR34c expression in vivo in animal models of pulmonary hypertension to identify the mechanistic role of miR34c in angiogenesis in the lung vasculature.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Data availability
The original sequencing output will be available for free download from the NCBI Sequence Read Archive after manuscript online publishing date under “Ovine PPHN PAEC smallRNA-seq” file name.
References
Gien, J. et al. Chronic intrauterine pulmonary hypertension increases endothelial cell rho kinase activity and impairs angiogenesis in vitro. Am. J. Physiol. Lung Cell Mol. Physiol. 295, L680–L687 (2008).
Teng, R. J., Eis, A., Bakhutashvili, I., Arul, N. & Konduri, G. G. Increased superoxide production contributes to the impaired angiogenesis of fetal pulmonary arteries with in utero pulmonary hypertension. Am. J. Physiol. Lung Cell Mol. Physiol. 297, L184–L195 (2009).
Mahajan, C. N., Afolayan, A. J., Eis, A., Teng, R. J. & Konduri, G. G. Altered prostanoid metabolism contributes to impaired angiogenesis in persistent pulmonary hypertension in a fetal lamb model. Pediatr. Res. 77, 455–462 (2015).
Rana, U. et al. Amp-kinase dysfunction alters Notch ligands to impair angiogenesis in neonatal pulmonary hypertension. Am. J. Respir. Cell Mol. Biol. 62, 719–731 (2020).
Risau, W. Mechanisms of angiogenesis. Nature 386, 671–674 (1997).
Gien, J., Seedorf, G. J., Balasubramaniam, V., Markham, N. & Abman, S. H. Intrauterine pulmonary hypertension impairs angiogenesis in vitro: role of vascular endothelial growth factor nitric oxide signaling. Am. J. Respir. Crit. Care Med. 176, 1146–1153 (2007).
Teng, R. J. et al. Amp kinase activation improves angiogenesis in pulmonary artery endothelial cells with in utero pulmonary hypertension. Am. J. Physiol. Lung Cell Mol. Physiol. 304, L29–L42 (2013).
Macfarlane, L. A. & Murphy, P. R. MicroRAN: biogenesis, function and role in cancer. Curr. Genomics 11, 537–561 (2010).
Tetreault, N. & De Guire, V. miRNAs: their discovery, biogenesis and mechanism of action. Clin. Biochem. 46, 842–845 (2013).
Poliseno, L. et al. MicroRNAs modulate the angiogenic properties of HUVECs. Blood 108, 3068–3071 (2006).
Caporali, A. & Emanueli, C. MicroRNA regulation in angiogenesis. Vasc. Pharm. 55, 79–86 (2011).
Pullamsetti, S. S. et al. Inhibition of microRNA-17 improves lung and heart function in experimental pulmonary hypertension. Am. J. Respir. Crit. Care Med. 185, 409–419 (2012).
Boucherat, O., Potus, F. & Bonnet, S. MicroRNA and pulmonary hypertension. Adv. Exp. Med. Biol. 888, 237–252 (2015).
Doebele, C. et al. Members of the microRNA-17-92 cluster exhibit a cell-intrinsic antiangiogenic function in endothelial cells. Blood 115, 4944–4950 (2010).
Bonauer, A. et al. MicroRNA-92a controls angiogenesis and functional recovery of ischemic tissues in mice. Science 324, 1710–1713 (2009).
Xu, Y. P. et al. miR-126a-5p is involved in the hypoxia-induced endothelial-to-mesenchymal transition of neonatal pulmonary hypertension. Hypertens. Res. 40, 552–561 (2017).
Nicoli, S. et al. MicroRNA-mediated integration of haemodynamics and VEGF signalling during angiogenesis. Nature 464, 1196–1200 (2010).
Qin, W. et al. BMPRII is a direct target of miR-21. Acta Biochim. Biophys. Sin. (Shanghai) 41, 618–623 (2009).
Sood, B. et al. Microrna Biomarkers in Persistent Pulmonary Hypertension of the Newborn. https://pvrinstitute.org/media/2534/074-beena-sood.pdf (2017).
Konduri, G. G. & Mital, S. Adenosine and ATP cause nitric oxide-dependent pulmonary vasodilation in fetal lambs. Biol. Neonate 78, 220–229 (2000).
Konduri, G. G. & Mattei, J. Role of oxidative phosphorylation and ATP release in mediating birth-related pulmonary vasodilation in fetal lambs. Am. J. Physiol. Heart Circ. Physiol. 283, H1600–H1608 (2002).
Hoyer, L. W., De los Santos, R. P. & Hoyer, J. R. Antihemophilic factor antigen. Localization in endothelial cells by immunofluorescent microscopy. J. Clin. Invest. 52, 2737–2744 (1973).
Voyta, J. C., Via, D. P., Butterfield, C. E. & Zetter, B. R. Identification and isolation of endothelial cells based on their increased uptake of acetylated-low density lipoprotein. J. Cell Biol. 99, 2034–2040 (1984).
Rio, D. C., Ares, M. Jr., Hannon, G. J. & Nilsen, T. W. Purification of RNA using TRIzol (TRI reagent). Cold Spring Harb. Protoc. 2010, pdb.prot5439 (2010).
Gao, F. et al. Changes in miRNA in the lung and whole blood after whole thorax irradiation in rats. Sci. Rep. 7, 44132 (2017).
Kriegel, A. J. et al. Characteristics of microRNAs enriched in specific cell types and primary tissue types in solid organs. Physiol. Genomics 45, 1144–1156 (2013).
Hackenberg, M., Sturm, M., Langenberger, D., Falcon-Perez, J. M. & Aransay, A. M. Miranalyzer: a microRNA detection and analysis tool for next-generation sequencing experiments. Nucleic Acids Res. 37, W68–W76 (2009).
Agarwal, V., Bell, G. W., Nam, J. W. & Bartel, D. P. Predicting effective microRAN target sites in mammalian mRNAs. Elife 4, e05005 (2015).
Benjamini, Y. & Hochberg, Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J. R. Stat. Soc. Ser. B (Methodol.) 57, 289–300 (1995).
Bray, S. J. Notch signalling in context. Nat. Rev. Mol. Cell Biol. 17, 722–735 (2016).
Limbourg, F. P. et al. Essential role of endothelial Notch1 in angiogenesis. Circulation 111, 1826–1832 (2005).
Bae, Y. et al. miRNA-34c regulates Notch signaling during bone development. Hum. Mol. Genet 21, 2991–3000 (2012).
Luo, Y., Wang, D., Chen, S. & Yang, Q. The role of miR-34c-5p/Notch in epithelial-mesenchymal transition (EMT) in endometriosis. Cell Signal 72, 109666 (2020).
Hou, L. et al. miR-34c represses muscle development by forming a regulatory loop with Notch1. Sci. Rep. 7, 9346 (2017).
Hellstrom, M. et al. Dll4 signalling through Notch1 regulates formation of tip cells during angiogenesis. Nature 445, 776–780 (2007).
Takeshita, K. et al. Critical role of endothelial Notch1 signaling in postnatal angiogenesis. Circ. Res. 100, 70–78 (2007).
Fischer, A., Schumacher, N., Maier, M., Sendtner, M. & Gessler, M. The Notch target genes Hey1 and Hey2 are required for embryonic vascular development. Genes Dev. 18, 901–911 (2004).
Kitagawa, M. et al. Hes1 and Hes5 regulate vascular remodeling and arterial specification of endothelial cells in brain vascular development. Mech. Dev. 130, 458–466 (2013).
Wei, H. et al. miR34c5p targets Notch1 and suppresses the metastasis and invasion of cervical cancer. Mol. Med. Rep. 23, 120 (2021).
Wan, W. F. et al. miR-34c inhibits PDGF-BB-induced HAVSMCs phenotypic transformation and proliferation via PDGFR-BETA/SIRT1 pathway. Mol. Biol. Rep. 48, 4137–4151 (2021).
Funding
This work was supported by grants 1R01 HL136597–01 from NHLBI, Multiyear Innovation Research grant, and Muma Endowed Chair in Neonatology from Children’s Research Institute of Children’s Wisconsin (G.G.K.).
Author information
Authors and Affiliations
Contributions
Substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data: D.M., U.R., A.J.K., P.L., T.M., and G.G.K. Drafting the article or revising it critically for important intellectual content: D.M. and G.G.K. Final approval of the version to be published: D.M. and G.G.K.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Mukherjee, D., Rana, U., Kriegel, A.J. et al. Fetal pulmonary hypertension: dysregulated microRNA-34c-Notch1 axis contributes to impaired angiogenesis in an ovine model. Pediatr Res 93, 551–558 (2023). https://doi.org/10.1038/s41390-022-02151-3
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41390-022-02151-3
This article is cited by
-
PIEZO2 in tumors: from mechanobiological switches to activity-targeted therapies
Journal of Experimental & Clinical Cancer Research (2025)
-
Deciphering epigenetic control of Notch signaling in persistent pulmonary hypertension of the newborn
Pediatric Research (2025)
-
Decreased endothelial micro-RNA-30b-5p impairs angiogenesis in fetal lambs with persistent pulmonary hypertension
Pediatric Research (2025)
-
The vascular phenotype of BPD: new basic science insights—new precision medicine approaches
Pediatric Research (2024)